Name - WordPress.com
... The process of dissolving a solute in a solvent is known generally as solvation. When the solvent is water, the dissolving process is also referred to as hydration. Ionic Compounds in Water When ionic compounds dissolve in water, each ion separates from the solid structure and disperses throughout t ...
... The process of dissolving a solute in a solvent is known generally as solvation. When the solvent is water, the dissolving process is also referred to as hydration. Ionic Compounds in Water When ionic compounds dissolve in water, each ion separates from the solid structure and disperses throughout t ...
Class Presentation – Naming and Formula Writing
... 5) sodium iodide 6) strontium fluoride 7) lithium sulfide 8) radium chloride 9) calcium oxide 10) aluminum phosphide ...
... 5) sodium iodide 6) strontium fluoride 7) lithium sulfide 8) radium chloride 9) calcium oxide 10) aluminum phosphide ...
solute
... between solute and solvent particles particle size – smaller dissolves faster because more surface area temperature – solutes generally dissolve faster at higher temps. – higher kinetic energy at higher temps. – stronger and more frequent collisions between solute and solvent particles - Excepti ...
... between solute and solvent particles particle size – smaller dissolves faster because more surface area temperature – solutes generally dissolve faster at higher temps. – higher kinetic energy at higher temps. – stronger and more frequent collisions between solute and solvent particles - Excepti ...
Chapter 2 Name___________________________________
... A) Covalent bonds involve the sharing of protons between atoms, and ionic bonds involve the sharing of neutrons between atoms. B) Covalent bonds involve the sharing of protons between atoms, and ionic bonds involve the sharing of electrons between atoms. C) Covalent bonds involve the sharing of neut ...
... A) Covalent bonds involve the sharing of protons between atoms, and ionic bonds involve the sharing of neutrons between atoms. B) Covalent bonds involve the sharing of protons between atoms, and ionic bonds involve the sharing of electrons between atoms. C) Covalent bonds involve the sharing of neut ...
Unit 4 - Dorman High School
... V. Ionic Bonding and Structures of Ionic Compounds When an ionic compound is formed the bond is extremely strong. We write the formulas for these compounds, but they are empirical formulas because the compound is composed of a very tightly packed and ordered arrangement of ions. Ionic compounds can ...
... V. Ionic Bonding and Structures of Ionic Compounds When an ionic compound is formed the bond is extremely strong. We write the formulas for these compounds, but they are empirical formulas because the compound is composed of a very tightly packed and ordered arrangement of ions. Ionic compounds can ...
Honors Chemistry Final Essay Questions 2007
... 1. 200. mL of 6.00 M HCl and 100.0 mL of 5.00 M Sr(OH)2 are mixed and diluted to 500. mL with distilled water. What are the values of pH, pOH, [H3O+1], and [OH -1] for the resulting ...
... 1. 200. mL of 6.00 M HCl and 100.0 mL of 5.00 M Sr(OH)2 are mixed and diluted to 500. mL with distilled water. What are the values of pH, pOH, [H3O+1], and [OH -1] for the resulting ...
Chemical Bonding
... compounds from one another, but not change them. The polar regions of these molecules are attracted to polar regions of the cellulose chains (which help to hold the fibers together in paper). Not surprisingly, water molecules, being polar, are also attracted to these regions and when paper is wet it ...
... compounds from one another, but not change them. The polar regions of these molecules are attracted to polar regions of the cellulose chains (which help to hold the fibers together in paper). Not surprisingly, water molecules, being polar, are also attracted to these regions and when paper is wet it ...
General College Chemistry
... describe matter and energy; classify states of matter and describe phase changes using the kinetic molecular theory; distinguish between elements/compounds/mixtures; physical/chemical, intensive/extensive, endothermic/exothermic changes and/or properties; solve conversion problems, including metric ...
... describe matter and energy; classify states of matter and describe phase changes using the kinetic molecular theory; distinguish between elements/compounds/mixtures; physical/chemical, intensive/extensive, endothermic/exothermic changes and/or properties; solve conversion problems, including metric ...
Test 2
... 11. (10 points) I have discovered a new material called zehfusoid. It is a solid at 25oC and 1 atm of pressure, but becomes liquid either when the pressure is raised to 100 atm or when the temperature is raised to 150o C. If the temperature drops to -50o C the solid turns to a gas. From this informa ...
... 11. (10 points) I have discovered a new material called zehfusoid. It is a solid at 25oC and 1 atm of pressure, but becomes liquid either when the pressure is raised to 100 atm or when the temperature is raised to 150o C. If the temperature drops to -50o C the solid turns to a gas. From this informa ...
Properties of Ionic Compounds
... The coordination number of an ion is the number of ions of opposite charge that surround the ion in a crystal. • In NaCℓ, each ion has a coordination number of 6. – The coordination number of Na+ is 6 because each Na+ ion is surrounded by six Cℓ– ions. – The coordination number of Cℓ– is also 6 beca ...
... The coordination number of an ion is the number of ions of opposite charge that surround the ion in a crystal. • In NaCℓ, each ion has a coordination number of 6. – The coordination number of Na+ is 6 because each Na+ ion is surrounded by six Cℓ– ions. – The coordination number of Cℓ– is also 6 beca ...
Science 10 (4.2) Names and formulas of
... 1. 1st part: ______________ ion, which is always a __________ in a compound containing ____ elements. 2. 2nd part: _____________ ion, which is always a __________________ in a compound containing ____ elements. 3. non-metal names end in "-____". ...
... 1. 1st part: ______________ ion, which is always a __________ in a compound containing ____ elements. 2. 2nd part: _____________ ion, which is always a __________________ in a compound containing ____ elements. 3. non-metal names end in "-____". ...
Carrying Charges
... 1. Participant must wear a pair of safety goggles before beginning! 2. Put a small amount of each liquid in separate wells in the well plate. 3. Dip the ends of both wires (electrodes) into each liquid and observe what happens. If the buzzer sounds, then the liquid conducts electricity and has ions ...
... 1. Participant must wear a pair of safety goggles before beginning! 2. Put a small amount of each liquid in separate wells in the well plate. 3. Dip the ends of both wires (electrodes) into each liquid and observe what happens. If the buzzer sounds, then the liquid conducts electricity and has ions ...
Chem Unit 3 Vocabulary
... pressure; 18 evaporation; 19 fluid; 20 freezing point; 21 ideal gas; 22 kinetic-molecular theory; 23 melting; 24 melting point; 25 molar enthalpy of fusion; 26 molar enthalpy of vaporization; etc… 1 solids in which the particles are arranged randomly 2 a change of a liquid to bubbles of vapor that a ...
... pressure; 18 evaporation; 19 fluid; 20 freezing point; 21 ideal gas; 22 kinetic-molecular theory; 23 melting; 24 melting point; 25 molar enthalpy of fusion; 26 molar enthalpy of vaporization; etc… 1 solids in which the particles are arranged randomly 2 a change of a liquid to bubbles of vapor that a ...
Chemical Bonding
... It is generally assumed that covalent and ionic bonds are entirely distinct but this is probably not a totally valid assumption. Bonds intermediate between ionic and covalent do occur through a process of deformation or polarization. ...
... It is generally assumed that covalent and ionic bonds are entirely distinct but this is probably not a totally valid assumption. Bonds intermediate between ionic and covalent do occur through a process of deformation or polarization. ...
NOMENCLATURE OF IONIC COMPOUNDS CHEMISTRY 1405
... (II) and this is incorrect. Oxidation number is expressed in parenthesis only for transistion metal ions or metal ions which show variable oxidation numbers. Barium belongs to group 2 and all elements in group 2 have a fixed oxidation number of +2. ...
... (II) and this is incorrect. Oxidation number is expressed in parenthesis only for transistion metal ions or metal ions which show variable oxidation numbers. Barium belongs to group 2 and all elements in group 2 have a fixed oxidation number of +2. ...
Unit 3 Review Questions - Unit #1-0
... 31. In the correct Lewis structure for water, how many unshared pairs of electrons will oxygen have? ...
... 31. In the correct Lewis structure for water, how many unshared pairs of electrons will oxygen have? ...
Nugget
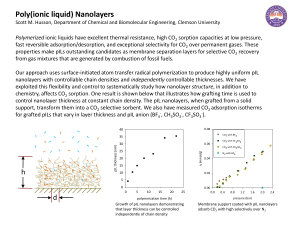
... Scott M. Husson, Department of Chemical and Biomolecular Engineering, Clemson University ...
... Scott M. Husson, Department of Chemical and Biomolecular Engineering, Clemson University ...
Ch 11 Review - mvhs
... forces. The magnitude of such forces is determined by the number of electrons in the atom. A Xe atom has more electrons than a neon atom has. (Size of the atom was accepted but mass was not.) (b) The electrical conductivity of copper metal is based on mobile valence electrons (partially filled bands ...
... forces. The magnitude of such forces is determined by the number of electrons in the atom. A Xe atom has more electrons than a neon atom has. (Size of the atom was accepted but mass was not.) (b) The electrical conductivity of copper metal is based on mobile valence electrons (partially filled bands ...
PowerPoint Chapter 14 - Preparatory Chemistry
... • Three questions will help you predict whether substances are composed of polar or nonpolar molecules. – Is the substance molecular? – If the substance is molecular, do the molecules contain polar covalent bonds? – If the molecules contain polar covalent bonds, are these bonds asymmetrically arrang ...
... • Three questions will help you predict whether substances are composed of polar or nonpolar molecules. – Is the substance molecular? – If the substance is molecular, do the molecules contain polar covalent bonds? – If the molecules contain polar covalent bonds, are these bonds asymmetrically arrang ...
CHEM1405 2005-J-3 June 2005 • Ammonia (NH 3) has a boiling
... much larger atom that H or C and hence has more electrons and these are held further from the nucleus. The electron cloud in I2 is, therefore, much more polarisable leading to stronger dispersion forces in I2. and a higher melting point. NaCl has relatively strong ionic bonds between all of the Na+ ...
... much larger atom that H or C and hence has more electrons and these are held further from the nucleus. The electron cloud in I2 is, therefore, much more polarisable leading to stronger dispersion forces in I2. and a higher melting point. NaCl has relatively strong ionic bonds between all of the Na+ ...
Spring 2001 - TAMU Chemistry
... Point 5 is called the triple point. At Point 3, the liquid phase is in equilibrium with gas phase. When the substance moves from the conditions at Point 1 to the conditions at Point 2, the substance boils. When the conditions change from Point 2 to Point 4, the temperature changes and the pressure s ...
... Point 5 is called the triple point. At Point 3, the liquid phase is in equilibrium with gas phase. When the substance moves from the conditions at Point 1 to the conditions at Point 2, the substance boils. When the conditions change from Point 2 to Point 4, the temperature changes and the pressure s ...
Chapter 2
... ❷All atoms of a given element are identical, both in mass and in chemical properties. However, they are different from atoms of other elements. ❸Atoms are not created or destroyed in chemical reactions. ❹Atoms combine in simple, fixed, whole-number ratios to form compounds. What evidence is there fo ...
... ❷All atoms of a given element are identical, both in mass and in chemical properties. However, they are different from atoms of other elements. ❸Atoms are not created or destroyed in chemical reactions. ❹Atoms combine in simple, fixed, whole-number ratios to form compounds. What evidence is there fo ...
This podcast will discuss two related misconceptions about solubility
... atoms, the more polar the molecule is. When two of the same atom bond, such as in O2 or N2, there is zero difference between the electronegativities and the molecule is completely non-polar. When a molecule is in between the two, it dissolves compounds a little more polar and a little less polar tha ...
... atoms, the more polar the molecule is. When two of the same atom bond, such as in O2 or N2, there is zero difference between the electronegativities and the molecule is completely non-polar. When a molecule is in between the two, it dissolves compounds a little more polar and a little less polar tha ...
Working with solutions
... O Ionic solids in water- positive and negative ions are attracted to polar water molecules. O Molecular solids in water- break up into individual neutral molecules and are surrounded by water. O Ionic compounds conducted electricity molecular compounds do not. ...
... O Ionic solids in water- positive and negative ions are attracted to polar water molecules. O Molecular solids in water- break up into individual neutral molecules and are surrounded by water. O Ionic compounds conducted electricity molecular compounds do not. ...
Find your NEW seats Bellringer: Please complete Ms - Parkway C-2
... Evaporation • Liquids may change to a vapor at temperatures below their boiling points through the process of evaporation. Evaporation is a surface phenomenon in which molecules located near the liquid's edge, escape into the surroundings as vapor (a gas). On the other hand, boiling is a process in ...
... Evaporation • Liquids may change to a vapor at temperatures below their boiling points through the process of evaporation. Evaporation is a surface phenomenon in which molecules located near the liquid's edge, escape into the surroundings as vapor (a gas). On the other hand, boiling is a process in ...
Ionic liquid
An ionic liquid (IL) is a salt in the liquid state. In some contexts, the term has been restricted to salts whose melting point is below some arbitrary temperature, such as 100 °C (212 °F). While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions and short-lived ion pairs. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid salts, or ionic glasses. Ionic liquids have many applications, such as powerful solvents and electrically conducting fluids (electrolytes). Salts that are liquid at near-ambient temperature are important for electric battery applications, and have been used as sealants due to their very low vapor pressure.Any salt that melts without decomposing or vaporizing usually yields an ionic liquid. Sodium chloride (NaCl), for example, melts at 801 °C (1,474 °F) into a liquid that consists largely of sodium cations (Na+) and chloride anions (Cl−). Conversely, when an ionic liquid is cooled, it often forms an ionic solid—which may be either crystalline or glassy.The ionic bond is usually stronger than the Van der Waals forces between the molecules of ordinary liquids. For that reason, common salts tend to melt at higher temperatures than other solid molecules. Some salts are liquid at or below room temperature. Examples include compounds based on the 1-Ethyl-3-methylimidazolium (EMIM) cation and include: EMIM:Cl, EMIM dicyanamide, (C2H5)(CH3)C3H3N+2·N(CN)−2, that melts at −21 °C (−6 °F); and 1-butyl-3,5-dimethylpyridinium bromide which becomes a glass below −24 °C (−11 °F).Low-temperature ionic liquids can be compared to ionic solutions, liquids that contain both ions and neutral molecules, and in particular to the so-called deep eutectic solvents, mixtures of ionic and non-ionic solid substances which have much lower melting points than the pure compounds. Certain mixtures of nitrate salts can have melting points below 100 °C.The term ""ionic liquid"" in the general sense was used as early as 1943.