Print Activity - Let`s Talk Science
... Is it a liquid or a solid? they do in a normal solid. This makes the goop feel and act as a solid. When the pressure is released the molecules form the random arrangement of a liquid and the goop suddenly flows. Why does it matter? All fluids have a measurable viscosity. Viscosity is the thickness ...
... Is it a liquid or a solid? they do in a normal solid. This makes the goop feel and act as a solid. When the pressure is released the molecules form the random arrangement of a liquid and the goop suddenly flows. Why does it matter? All fluids have a measurable viscosity. Viscosity is the thickness ...
Book 2,Part 7 - GEOCITIES.ws
... pure component at the same temperature, multiplied by the mole fraction of it in the liquid mixture. Raoult’s law is obeyed when the intermolecular forces between liquid molecules in the mixture are the same as in the pure liquids. ...
... pure component at the same temperature, multiplied by the mole fraction of it in the liquid mixture. Raoult’s law is obeyed when the intermolecular forces between liquid molecules in the mixture are the same as in the pure liquids. ...
Redox reactions in deep eutectic solvents
... Ionic liquids are a field of intense chemical research activity due to their potential to facilitate a wide range of chemical innovations. Due to the cost of ionic liquids alternative materials which exhibit similar properties, but are less expensive, are also the subject of interest and deep eutecti ...
... Ionic liquids are a field of intense chemical research activity due to their potential to facilitate a wide range of chemical innovations. Due to the cost of ionic liquids alternative materials which exhibit similar properties, but are less expensive, are also the subject of interest and deep eutecti ...
11-16 States of Matter
... Very large amounts of energy (more than solids, liquids, or regular gases) Is a super-heated gas, with lots & lots of movement Examples: Stars, fire, and lightning Lightning Stars ...
... Very large amounts of energy (more than solids, liquids, or regular gases) Is a super-heated gas, with lots & lots of movement Examples: Stars, fire, and lightning Lightning Stars ...
Solutions
... Parts per million (ppm) is similar to percent by mass because it compares masses. It represents the ratio between the mass of a solute and the total mass of a solution. Instead of multiply by 100 you multiply by 1,000,000. ppm is often used to measure concentrations of solutes that are present in ve ...
... Parts per million (ppm) is similar to percent by mass because it compares masses. It represents the ratio between the mass of a solute and the total mass of a solution. Instead of multiply by 100 you multiply by 1,000,000. ppm is often used to measure concentrations of solutes that are present in ve ...
Preparation of Reducing Sugar Hydrolyzed from High
... crystalline orientation of cellulose, thus improving the yield of fermentable sugar in enzymatic hydrolysis [2-4]. Chemical methods have been employed to carry out pretreatment on grape stalks to regain both hemicellulose and cellulose [5]. However, the wastewater, which contained chemical substance ...
... crystalline orientation of cellulose, thus improving the yield of fermentable sugar in enzymatic hydrolysis [2-4]. Chemical methods have been employed to carry out pretreatment on grape stalks to regain both hemicellulose and cellulose [5]. However, the wastewater, which contained chemical substance ...
Chemistry in Focus: Tiny Thermometers
... It turns out that the tiny thermometers were produced by accident. The Japanese scientists were actually trying to make tiny (nanoscale) gallium nitride wires. However, when they examined the results of their experiment, they discovered tiny tubes of carbon atoms that were filled with elemental gall ...
... It turns out that the tiny thermometers were produced by accident. The Japanese scientists were actually trying to make tiny (nanoscale) gallium nitride wires. However, when they examined the results of their experiment, they discovered tiny tubes of carbon atoms that were filled with elemental gall ...
Ch13ov1
... L Temperature increases may result in greater or lesser solubility. • Temperature effect is not related to ∆Hsoln. • Rate of dissolving is usually enhanced at higher temperature, regardless of the effect on solubility limit. Non-reactive Gas Solutes L Gases are more soluble at higher pressure. L Gas ...
... L Temperature increases may result in greater or lesser solubility. • Temperature effect is not related to ∆Hsoln. • Rate of dissolving is usually enhanced at higher temperature, regardless of the effect on solubility limit. Non-reactive Gas Solutes L Gases are more soluble at higher pressure. L Gas ...
Ion exchange chromatography
... • Flow rate determines the amount of time in which proteins can interact with the column resin, which is called the residence time of a particular column at a given flow rate • Flow rate affects both resolution and capacity ...
... • Flow rate determines the amount of time in which proteins can interact with the column resin, which is called the residence time of a particular column at a given flow rate • Flow rate affects both resolution and capacity ...
CHM – 124 Principles of Chemistry
... State Boyle’s, Charles’, and Gay-Lussac’s laws. Use all of them in problems. State the combined gas law. Indicate when it is used. State the ideal gas law. Solve problems involving its use. State the molar volume concept. Calculate the molar mass of a gas from its density at STP and the molar volume ...
... State Boyle’s, Charles’, and Gay-Lussac’s laws. Use all of them in problems. State the combined gas law. Indicate when it is used. State the ideal gas law. Solve problems involving its use. State the molar volume concept. Calculate the molar mass of a gas from its density at STP and the molar volume ...
General Chemistry
... LIKE DISSOLVES LIKE • polar solvents dissolve polar solutes. • Non-polar solvents dissolve non-polar solutes. Water is polar (because it’s bent). It will therefore tend to dissolve other polar molecules or ions. For example, most salts, alcohols and sugars dissolve in water. Alcohols and sugars all ...
... LIKE DISSOLVES LIKE • polar solvents dissolve polar solutes. • Non-polar solvents dissolve non-polar solutes. Water is polar (because it’s bent). It will therefore tend to dissolve other polar molecules or ions. For example, most salts, alcohols and sugars dissolve in water. Alcohols and sugars all ...
Lecture 10 Activity of chemical components
... modified and it is lesser than the one measured in very dilute solution. For example in 0.1 molar solutions it could differ by factor of three. Similarly, if we were to consider the effect of added electrolyte like Na Acetate, we can recalculate the activity coefficient using Debye model. ...
... modified and it is lesser than the one measured in very dilute solution. For example in 0.1 molar solutions it could differ by factor of three. Similarly, if we were to consider the effect of added electrolyte like Na Acetate, we can recalculate the activity coefficient using Debye model. ...
Experiment 4 Separation of a Mixture
... We will at the beginning of lab talked extensively about the different separation techniques and the above table, after which time you will be asked to design a flow chart that will be used to separate the different components of an unknown mixture. Your flow chart will be rather general to begin w ...
... We will at the beginning of lab talked extensively about the different separation techniques and the above table, after which time you will be asked to design a flow chart that will be used to separate the different components of an unknown mixture. Your flow chart will be rather general to begin w ...
Chemistry Lecture *34". Ionic. Compounds I-P one atom trans
... I-P one atom trans-Pers its electrons to another, they will stick together because one atom will have a positive charge and the other will have a negative charge. Electrostatic -Porce is the -Porce o£ attraction between opposite charges. Thus, anions and cations will stick together due to the electr ...
... I-P one atom trans-Pers its electrons to another, they will stick together because one atom will have a positive charge and the other will have a negative charge. Electrostatic -Porce is the -Porce o£ attraction between opposite charges. Thus, anions and cations will stick together due to the electr ...
Chapter 2 What Is Matter
... Actually, solutions do not have to be a solid dissolved in a liquid. Since there are three common phases of matter, there are nine possible types of solutions. Such as: Air is a gas dissolved in a gas. Soft drinks are a gas dissolved in a liquid. Humid air is a liquid dissolved in a gas. Antifreeze ...
... Actually, solutions do not have to be a solid dissolved in a liquid. Since there are three common phases of matter, there are nine possible types of solutions. Such as: Air is a gas dissolved in a gas. Soft drinks are a gas dissolved in a liquid. Humid air is a liquid dissolved in a gas. Antifreeze ...
Miami-Dade College
... substances to physical properties such as vapor pressure, melting point, boiling point, and viscosity. ...
... substances to physical properties such as vapor pressure, melting point, boiling point, and viscosity. ...
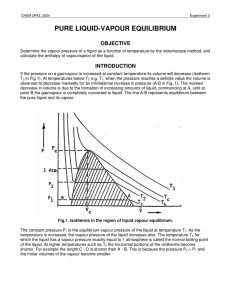
pure liquid-vapour equilibrium - Theoretical and Computational
... Part of the shaded area, up to the normal boiling point, Tb, of the liquid, is the subject of this experiment in which the vapour pressure dependence on temperature will be investigated. This dependence can be related to the enthalpy of vapourisation Hvap in the following way. For equilibrium betwee ...
... Part of the shaded area, up to the normal boiling point, Tb, of the liquid, is the subject of this experiment in which the vapour pressure dependence on temperature will be investigated. This dependence can be related to the enthalpy of vapourisation Hvap in the following way. For equilibrium betwee ...
Module 4 : Organoelement compounds of Group 15 Lecture 1
... The importance given to organoarsenic compounds earlier due to their medicinal values was waded out after antibiotics were discovered and also their carcinogenic and toxic properties were revealed. Also, the synthetically important organometallic compounds of group 13 and 14 masked the growth of gro ...
... The importance given to organoarsenic compounds earlier due to their medicinal values was waded out after antibiotics were discovered and also their carcinogenic and toxic properties were revealed. Also, the synthetically important organometallic compounds of group 13 and 14 masked the growth of gro ...
NH 4 1+
... Here the possible cross-products are Pb with I and Na with NO3. Let’s check whether either of these meets one of the three criteria: something insoluble, a weak acid, or something that decomposes. NaNO3 is not insoluble (by rule #1); it is not a weak acid (since it doesn’t start with H, it’s not an ...
... Here the possible cross-products are Pb with I and Na with NO3. Let’s check whether either of these meets one of the three criteria: something insoluble, a weak acid, or something that decomposes. NaNO3 is not insoluble (by rule #1); it is not a weak acid (since it doesn’t start with H, it’s not an ...
[HMIM][Br9]: a Room-temperature Ionic Liquid Based on a
... According to Spange et al. [21] the chemical shift of the hydrogen atom at the C-2 position of the imidazolium ring is dependent on the interaction of the cation with the anion. In diluted solutions of different imidazolium salts in dichloromethane they found the resonance of this hydrogen atom shif ...
... According to Spange et al. [21] the chemical shift of the hydrogen atom at the C-2 position of the imidazolium ring is dependent on the interaction of the cation with the anion. In diluted solutions of different imidazolium salts in dichloromethane they found the resonance of this hydrogen atom shif ...
Chapter 6 - Department of Chemical Engineering
... Safety Issues: In this experiment acetone and chloroform will be used as chemicals. Due to their hazardous and corrosive nature, avoid skin and eye contact while handling chemicals. Avoid wearing contact lenses. The experiment will be performed under a hood. Do not inhale any chemical vapors. In cas ...
... Safety Issues: In this experiment acetone and chloroform will be used as chemicals. Due to their hazardous and corrosive nature, avoid skin and eye contact while handling chemicals. Avoid wearing contact lenses. The experiment will be performed under a hood. Do not inhale any chemical vapors. In cas ...
Diapositivo 1
... need to calculate the chemical potential of a liquid. To do this, we will use the fact that the chemical potential of a substance present as a dilute vapor must be equal to the chemical potential of the liquid, at equilibrium. Remember also that it is usual, in order to characterize a given solution ...
... need to calculate the chemical potential of a liquid. To do this, we will use the fact that the chemical potential of a substance present as a dilute vapor must be equal to the chemical potential of the liquid, at equilibrium. Remember also that it is usual, in order to characterize a given solution ...
Export To Word
... Exploring the states (of Matter): Super Solids, the attached PowerPoint could be shown to as a preview to this exploration. This is the second lesson of three lessons designed for second grade students to understand and appreciate what matter is and Explore the States (of Matter): which properties ...
... Exploring the states (of Matter): Super Solids, the attached PowerPoint could be shown to as a preview to this exploration. This is the second lesson of three lessons designed for second grade students to understand and appreciate what matter is and Explore the States (of Matter): which properties ...
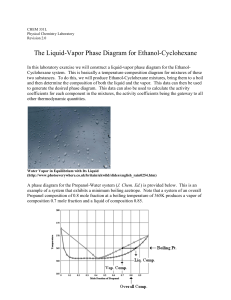
The Liquid-Vapor Phase Diagram for Ethanol
... Set-up the Still as pictured in the background material above. Use a heating block on a hot plate as a heat source. To begin the equilibrium measurements, introduce about 50 mL of Ethanol into the clean, dry distilling flask. Add a boiling stone to the liquid. Adjust the heat source so that the liqu ...
... Set-up the Still as pictured in the background material above. Use a heating block on a hot plate as a heat source. To begin the equilibrium measurements, introduce about 50 mL of Ethanol into the clean, dry distilling flask. Add a boiling stone to the liquid. Adjust the heat source so that the liqu ...
Chapter 3: Calculations with Chemical Formulas
... The alkaline earth metals (Be, Mg, Ca, Sr, Ba, and Ra) and also Zn and Cd in compounds are always assigned an oxidation state of +2. Hydrogen in compounds is assigned an oxidation state of +1. Oxygen in compounds is assigned an oxidation state of -2. Halogen in compounds is assigned an oxidation sta ...
... The alkaline earth metals (Be, Mg, Ca, Sr, Ba, and Ra) and also Zn and Cd in compounds are always assigned an oxidation state of +2. Hydrogen in compounds is assigned an oxidation state of +1. Oxygen in compounds is assigned an oxidation state of -2. Halogen in compounds is assigned an oxidation sta ...
Ionic liquid
An ionic liquid (IL) is a salt in the liquid state. In some contexts, the term has been restricted to salts whose melting point is below some arbitrary temperature, such as 100 °C (212 °F). While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions and short-lived ion pairs. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid salts, or ionic glasses. Ionic liquids have many applications, such as powerful solvents and electrically conducting fluids (electrolytes). Salts that are liquid at near-ambient temperature are important for electric battery applications, and have been used as sealants due to their very low vapor pressure.Any salt that melts without decomposing or vaporizing usually yields an ionic liquid. Sodium chloride (NaCl), for example, melts at 801 °C (1,474 °F) into a liquid that consists largely of sodium cations (Na+) and chloride anions (Cl−). Conversely, when an ionic liquid is cooled, it often forms an ionic solid—which may be either crystalline or glassy.The ionic bond is usually stronger than the Van der Waals forces between the molecules of ordinary liquids. For that reason, common salts tend to melt at higher temperatures than other solid molecules. Some salts are liquid at or below room temperature. Examples include compounds based on the 1-Ethyl-3-methylimidazolium (EMIM) cation and include: EMIM:Cl, EMIM dicyanamide, (C2H5)(CH3)C3H3N+2·N(CN)−2, that melts at −21 °C (−6 °F); and 1-butyl-3,5-dimethylpyridinium bromide which becomes a glass below −24 °C (−11 °F).Low-temperature ionic liquids can be compared to ionic solutions, liquids that contain both ions and neutral molecules, and in particular to the so-called deep eutectic solvents, mixtures of ionic and non-ionic solid substances which have much lower melting points than the pure compounds. Certain mixtures of nitrate salts can have melting points below 100 °C.The term ""ionic liquid"" in the general sense was used as early as 1943.
















![[HMIM][Br9]: a Room-temperature Ionic Liquid Based on a](http://s1.studyres.com/store/data/016911324_1-ac5688316a1e3a6c1ba364df016e5832-300x300.png)