Growing Negative Pressure in Dissolved Solutes: Raman - HAL-Insu
... wavenumber of the line center. Only this latter differs from mono- to biphasic states. No Bose-Einstein (BE) thermal correction was done27, since Raman shifts are compared at identical temperatures in two different pressure conditions (monophasic and biphasic regimes). Moreover this correction would ...
... wavenumber of the line center. Only this latter differs from mono- to biphasic states. No Bose-Einstein (BE) thermal correction was done27, since Raman shifts are compared at identical temperatures in two different pressure conditions (monophasic and biphasic regimes). Moreover this correction would ...
Pervaporation overview
... used with an inside–out configuration to avoid increase in permeate pressure within the fibers, but the outside–in configuration can be used with short fibers. Another advantage of the inside-out configuration is that the thin top layer is better protected but higher membrane area can be achieved wi ...
... used with an inside–out configuration to avoid increase in permeate pressure within the fibers, but the outside–in configuration can be used with short fibers. Another advantage of the inside-out configuration is that the thin top layer is better protected but higher membrane area can be achieved wi ...
Charging of Oil-Water Interfaces Due to Spontaneous Adsorption of
... magnitude in negative surface potential was registered at high pH. The observed decrease in the magnitude of UE with the increase of the NaCl concentration (Figure 2) is usually explained with the shrinkage of the electrical double layer at high ionic strength. At lower electrolyte concentrations, U ...
... magnitude in negative surface potential was registered at high pH. The observed decrease in the magnitude of UE with the increase of the NaCl concentration (Figure 2) is usually explained with the shrinkage of the electrical double layer at high ionic strength. At lower electrolyte concentrations, U ...
Reasoning about Fluids Via Molecular Collections
... Our goal is to construct a history - for MC, describing the sequence of places it is in and what is happening to it in those places . For our purposes, MC is uniquely defined by the place it is in, the substance of which it is composed,' and its current phase (i .e ., solid, liquid or gas) . The pla ...
... Our goal is to construct a history - for MC, describing the sequence of places it is in and what is happening to it in those places . For our purposes, MC is uniquely defined by the place it is in, the substance of which it is composed,' and its current phase (i .e ., solid, liquid or gas) . The pla ...
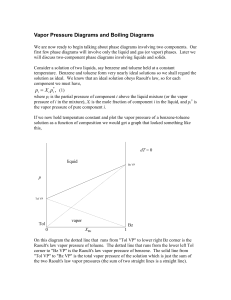
Vapor Pressure Diagrams and Boiling Diagrams
... As we heat the liquid it will begin to boil when the temperature reaches the temperature of point "a." The first vapor to come off has the composition shown at point "b." Capture the vapor, condense it, and heat it up. The new liquid will boil at point "c" giving a vapor with composition at point "d ...
... As we heat the liquid it will begin to boil when the temperature reaches the temperature of point "a." The first vapor to come off has the composition shown at point "b." Capture the vapor, condense it, and heat it up. The new liquid will boil at point "c" giving a vapor with composition at point "d ...
Contact Angle Goniometry as a Tool for Surface Tension
... Measurement of Young’s Contact Angle The technique of measuring Young’s contact angles was described previously (7) in detail, but its main facets will be presented for the benefit of novices to the method. To find Young’s contact angle for a liquid in contact with a surface, a drop of the liquid is ...
... Measurement of Young’s Contact Angle The technique of measuring Young’s contact angles was described previously (7) in detail, but its main facets will be presented for the benefit of novices to the method. To find Young’s contact angle for a liquid in contact with a surface, a drop of the liquid is ...
Calculating the conductivity of natural waters
... that must be determined empirically for each ion, but having this term in the denominator gives it a slightly wider range of validity than a form like Eq. 4. This form is simpler than the 4-term expansions generally recommended for analysis of binary electrolytes (Barthel et al. 1998) and thus canno ...
... that must be determined empirically for each ion, but having this term in the denominator gives it a slightly wider range of validity than a form like Eq. 4. This form is simpler than the 4-term expansions generally recommended for analysis of binary electrolytes (Barthel et al. 1998) and thus canno ...
High pressure differential scanning calorimetry of the hydrothermal
... This signal is attributed to the appearance of a solid phase (G–L / G–L–S). Comparison of the heat ow curves with those obtained from experiments in the cooling mode showed that the precipitation of the solid salt is kinetically hindered when the measurement is performed in the heating mode and tha ...
... This signal is attributed to the appearance of a solid phase (G–L / G–L–S). Comparison of the heat ow curves with those obtained from experiments in the cooling mode showed that the precipitation of the solid salt is kinetically hindered when the measurement is performed in the heating mode and tha ...
The Solubility of Potassium Sulfate in Thermodynamic view
... independent from each other and the electrolyte behaves almost as an ideal solution. As the concentration increases, the average distance between the ions decreases, so interactions between them become considerable and the solution becomes progressively less ideal. In addition, the ion- solvent inte ...
... independent from each other and the electrolyte behaves almost as an ideal solution. As the concentration increases, the average distance between the ions decreases, so interactions between them become considerable and the solution becomes progressively less ideal. In addition, the ion- solvent inte ...
An assessment of excess carbon dioxide partial pressures in natural
... The ion activity coefficient varies as a function of the salt content of the solution and this needs to be allowed for. For the calculation a typical background ‘sea salt’ concentration of 10 mgrl of chloride is used, the charge balance being matched by monovalent ions. Within the calculation, an al ...
... The ion activity coefficient varies as a function of the salt content of the solution and this needs to be allowed for. For the calculation a typical background ‘sea salt’ concentration of 10 mgrl of chloride is used, the charge balance being matched by monovalent ions. Within the calculation, an al ...
Experiment 7
... SOLUTION AT 30°C Background The solubility of potassium hydrogen tartarate (KHT) in distilled water and in aqueous sodium chloride solutions at concentrations 0.05, 0.10, 0.15 and 0.20 mol dm-3 are determined at 30 oC by the following method. For the equilibrium dissociation reaction KHT (s) ↔ K+ (a ...
... SOLUTION AT 30°C Background The solubility of potassium hydrogen tartarate (KHT) in distilled water and in aqueous sodium chloride solutions at concentrations 0.05, 0.10, 0.15 and 0.20 mol dm-3 are determined at 30 oC by the following method. For the equilibrium dissociation reaction KHT (s) ↔ K+ (a ...
chemistry — released items - North Carolina Public Schools
... CHEMISTRY — RELEASED ITEMS How does a single covalent bond between two carbon atoms compare to a double covalent bond between two carbon atoms? A ...
... CHEMISTRY — RELEASED ITEMS How does a single covalent bond between two carbon atoms compare to a double covalent bond between two carbon atoms? A ...
Ions in crystals: The topology of the electron density in ionic
... We present a systematic analysis of the topological properties of the electronic density in a family of ionic crystals: the halide perovskites AM X 3 . Our analysis lies within the framework of the atoms in molecules ~AIM! theory developed by Bader and co-workers.1,2 AIM theory provides a rigorous q ...
... We present a systematic analysis of the topological properties of the electronic density in a family of ionic crystals: the halide perovskites AM X 3 . Our analysis lies within the framework of the atoms in molecules ~AIM! theory developed by Bader and co-workers.1,2 AIM theory provides a rigorous q ...
AP syllabus
... 2. Kinetic theory of ideal gases 3. Pressure units and conversions between units 4. Solving problems using Dalton’s Law of Partial Pressures 5. Solving problems using Boyle’s, Charles’, Gay-Lussac’s and combined gas laws 6. Solving gas law problems when the gas is collected over water 7. Solving pro ...
... 2. Kinetic theory of ideal gases 3. Pressure units and conversions between units 4. Solving problems using Dalton’s Law of Partial Pressures 5. Solving problems using Boyle’s, Charles’, Gay-Lussac’s and combined gas laws 6. Solving gas law problems when the gas is collected over water 7. Solving pro ...
AP Lab - MW of Volatile Liquid - North Allegheny School District
... applicable to any substance, provided the chemist has sufficient skill and sophisticated gas-handling equipment to obtain the sample under controlled conditions. However, if the sample is a liquid at room temperature, with only a modest vapor pressure, and if it can be completely vaporized at some r ...
... applicable to any substance, provided the chemist has sufficient skill and sophisticated gas-handling equipment to obtain the sample under controlled conditions. However, if the sample is a liquid at room temperature, with only a modest vapor pressure, and if it can be completely vaporized at some r ...
Qualitative Analysis Test for Ions
... chloride ions. In this test, dilute nitric acid is added to the solution, followed by the silver nitrate solution. A white precipitate shows the presence of chloride ions. Why must the dilute nitric acid be added to make this a reliable test? Put a cross ( ) in the box next to your answer. ...
... chloride ions. In this test, dilute nitric acid is added to the solution, followed by the silver nitrate solution. A white precipitate shows the presence of chloride ions. Why must the dilute nitric acid be added to make this a reliable test? Put a cross ( ) in the box next to your answer. ...
Packet #6- Ionic and Covalent Bonding
... highest occupied energy levels - or outer shells - of the atoms. An atom that shares one or more of its electrons will complete its highest occupied energy level. Covalent bonds are strong - a lot of energy is needed to break them. Substances with covalent bonds often form molecules with low melting ...
... highest occupied energy levels - or outer shells - of the atoms. An atom that shares one or more of its electrons will complete its highest occupied energy level. Covalent bonds are strong - a lot of energy is needed to break them. Substances with covalent bonds often form molecules with low melting ...
Thermodynamics of Ion Association in the Saturated Solution of
... The concentration Gibbs free energies of solvation ∆G c and modeled Gibbs free energies of solvation ∆Gm for barium diphenylamine sulfonate in mixed EtOH-H2O solvents at different temperature are tabulated in Table (4). The transfer Concentration Gibbs free energies of solvation ∆Gt.c and transfer m ...
... The concentration Gibbs free energies of solvation ∆G c and modeled Gibbs free energies of solvation ∆Gm for barium diphenylamine sulfonate in mixed EtOH-H2O solvents at different temperature are tabulated in Table (4). The transfer Concentration Gibbs free energies of solvation ∆Gt.c and transfer m ...
DEFINATION - PharmaQuesT
... Polymorphic behavior of drugs and excipients is an important part of the preformulation work ...
... Polymorphic behavior of drugs and excipients is an important part of the preformulation work ...
ExamView - Chap_Test_v2.tst
... compound may be used more than once. a. ionic compound b. covalent compound ____ 23. this compound forms when metals react with non-metals ____ 24. molecules exist in this compound ____ 25. the name of this compound may include Roman numerals ____ 26. electrons are transferred when this compound for ...
... compound may be used more than once. a. ionic compound b. covalent compound ____ 23. this compound forms when metals react with non-metals ____ 24. molecules exist in this compound ____ 25. the name of this compound may include Roman numerals ____ 26. electrons are transferred when this compound for ...
Ionic Conductivity in the Metal–Organic Framework UiO
... be occupied by tBuO anions. Interestingly, metals analysis consistently yielded a Li:Zr ratio too high for the observed tBuO content, indicating that Li + and tBuO are not introduced in a stoichiometric ratio. Depending on the UiO-66 synthesis batch and preparation method, an excess of up to one Li ...
... be occupied by tBuO anions. Interestingly, metals analysis consistently yielded a Li:Zr ratio too high for the observed tBuO content, indicating that Li + and tBuO are not introduced in a stoichiometric ratio. Depending on the UiO-66 synthesis batch and preparation method, an excess of up to one Li ...
Document
... Thermodynamical description of mixtures, partial molar quantities; partial molar volume and Gibbs free energy. Phases, components and degree of freedom, the phase rule, phase diagrams; interpretation, lever rule. Liquid-liquid phase diagrams; phase separation, critical solution temperatures. Tempera ...
... Thermodynamical description of mixtures, partial molar quantities; partial molar volume and Gibbs free energy. Phases, components and degree of freedom, the phase rule, phase diagrams; interpretation, lever rule. Liquid-liquid phase diagrams; phase separation, critical solution temperatures. Tempera ...
AP CHEMISTRY COURSE SYLLABUS
... Vaporization of liquid Nitrogen Lab: Melting/Freezing of water. ...
... Vaporization of liquid Nitrogen Lab: Melting/Freezing of water. ...
plumbum thiogallate optical properties
... introduction of Ln dopants into cationic sublattice in typical "laser" concentrations 10 20 cm-3 without changing optical properties of the crystal itself. Such crystalline hosts must contain at least cations with coordination number 6 or 8 and ionic radii close to the radius of the doping ion. Sulf ...
... introduction of Ln dopants into cationic sublattice in typical "laser" concentrations 10 20 cm-3 without changing optical properties of the crystal itself. Such crystalline hosts must contain at least cations with coordination number 6 or 8 and ionic radii close to the radius of the doping ion. Sulf ...
Ionic liquid
An ionic liquid (IL) is a salt in the liquid state. In some contexts, the term has been restricted to salts whose melting point is below some arbitrary temperature, such as 100 °C (212 °F). While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions and short-lived ion pairs. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid salts, or ionic glasses. Ionic liquids have many applications, such as powerful solvents and electrically conducting fluids (electrolytes). Salts that are liquid at near-ambient temperature are important for electric battery applications, and have been used as sealants due to their very low vapor pressure.Any salt that melts without decomposing or vaporizing usually yields an ionic liquid. Sodium chloride (NaCl), for example, melts at 801 °C (1,474 °F) into a liquid that consists largely of sodium cations (Na+) and chloride anions (Cl−). Conversely, when an ionic liquid is cooled, it often forms an ionic solid—which may be either crystalline or glassy.The ionic bond is usually stronger than the Van der Waals forces between the molecules of ordinary liquids. For that reason, common salts tend to melt at higher temperatures than other solid molecules. Some salts are liquid at or below room temperature. Examples include compounds based on the 1-Ethyl-3-methylimidazolium (EMIM) cation and include: EMIM:Cl, EMIM dicyanamide, (C2H5)(CH3)C3H3N+2·N(CN)−2, that melts at −21 °C (−6 °F); and 1-butyl-3,5-dimethylpyridinium bromide which becomes a glass below −24 °C (−11 °F).Low-temperature ionic liquids can be compared to ionic solutions, liquids that contain both ions and neutral molecules, and in particular to the so-called deep eutectic solvents, mixtures of ionic and non-ionic solid substances which have much lower melting points than the pure compounds. Certain mixtures of nitrate salts can have melting points below 100 °C.The term ""ionic liquid"" in the general sense was used as early as 1943.