
Syllabus Cambridge IGCSE Chemistry (US) Syllabus Code 0439 For examination in 2013
... 2. scientific vocabulary, terminology, and conventions (including symbols, quantities, and units) 3. scientific instruments and apparatus, including techniques of operation and aspects of safety 4. scientific quantities and their determination 5. scientific and technological applications with their ...
... 2. scientific vocabulary, terminology, and conventions (including symbols, quantities, and units) 3. scientific instruments and apparatus, including techniques of operation and aspects of safety 4. scientific quantities and their determination 5. scientific and technological applications with their ...
Chapter 3 Stoichiometry: Calculations with Chemical
... • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
... • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
Calculations with Chemical Formulas and Equations
... • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
... • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... • One mole of atoms, ions, or molecules contains Avogadro s number of those particles • One mole of molecules or formula units contains Avogadro s number times the number of atoms or ions of each element in the compound Stoichiometry ...
... • One mole of atoms, ions, or molecules contains Avogadro s number of those particles • One mole of molecules or formula units contains Avogadro s number times the number of atoms or ions of each element in the compound Stoichiometry ...
Unit 4
... a gas? How many molecules are in 1 mole of any compound? For the reaction below, what mass of water can be produced from 1.5 moles of hydrogen? (27g) 2H2 + O2 --> 2H2O ...
... a gas? How many molecules are in 1 mole of any compound? For the reaction below, what mass of water can be produced from 1.5 moles of hydrogen? (27g) 2H2 + O2 --> 2H2O ...
Chemical Vapor Deposition (CVD)
... • This technique is suitable for the manufacture of coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total p ...
... • This technique is suitable for the manufacture of coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total p ...
CHAPTER 9
... 4. RELATING IDEAS What step must be performed before any stoichiometry problem is solved? Explain. ...
... 4. RELATING IDEAS What step must be performed before any stoichiometry problem is solved? Explain. ...
Stoichiometry
... Chemistry - A Molecular Science (CAMS), the first half of this two-volume sequence, stressed bonding, structure, and reactivity. The material was qualitative and stressed several types of reactions and the factors that affected their relative extents of reaction. However, as the title of this text s ...
... Chemistry - A Molecular Science (CAMS), the first half of this two-volume sequence, stressed bonding, structure, and reactivity. The material was qualitative and stressed several types of reactions and the factors that affected their relative extents of reaction. However, as the title of this text s ...
Spontaneous Change: Entropy and Gibbs Energy
... without changing the length of the box. Such an increase in U can be achieved, for example, by raising the temperature of the system. Figure 13.1(b) shows that, for U = 8 * (h2/8mL2), there are five possible microstates (W = 5) and an increase in the number of energy levels that are accessible to th ...
... without changing the length of the box. Such an increase in U can be achieved, for example, by raising the temperature of the system. Figure 13.1(b) shows that, for U = 8 * (h2/8mL2), there are five possible microstates (W = 5) and an increase in the number of energy levels that are accessible to th ...
guess paper class xii
... 15 How many ml of a 0.1M HCl are required to react completely with1 gm mixture of Na 2CO3 and NaHCO3 containing equimolar amounts of two? ...
... 15 How many ml of a 0.1M HCl are required to react completely with1 gm mixture of Na 2CO3 and NaHCO3 containing equimolar amounts of two? ...
EQUILIBRIUM - SCH4U1-CCVI
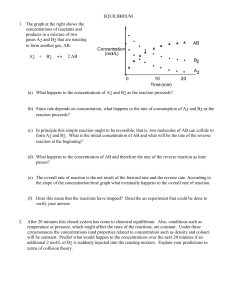
... concentrations may be used to calculate the value of the equilibrium constant for this reaction. Then predictions may be made about the equilibrium concentrations in other solutions. To prepare a solution with a known concentration of the coloured complex, a dilute solution of thiocyanate ion is rea ...
... concentrations may be used to calculate the value of the equilibrium constant for this reaction. Then predictions may be made about the equilibrium concentrations in other solutions. To prepare a solution with a known concentration of the coloured complex, a dilute solution of thiocyanate ion is rea ...
幻灯片 1
... Isotopes are atoms with the same atomic number but different numbers of the neutrons for one element, so their mass numbers will be different. They have the same amount of protons. Potential application: NMR, dating and origin of rocks, the studies for the reaction mechanism, etc ...
... Isotopes are atoms with the same atomic number but different numbers of the neutrons for one element, so their mass numbers will be different. They have the same amount of protons. Potential application: NMR, dating and origin of rocks, the studies for the reaction mechanism, etc ...
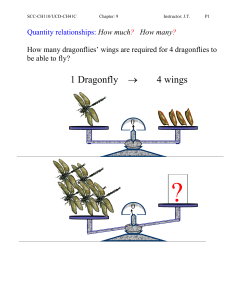
Quantity relationships: How much
... Percent Yield: The calculated amount of product if is based on the assumption that all of the reactant is converted into product is called the theoretical yield. In laboratory or in industrial production, the actual amount of product isolated from a reaction is usually less than the theoretical yiel ...
... Percent Yield: The calculated amount of product if is based on the assumption that all of the reactant is converted into product is called the theoretical yield. In laboratory or in industrial production, the actual amount of product isolated from a reaction is usually less than the theoretical yiel ...
Topic 7.2 Equilibrium The Position of Equilibrium
... High pressure is favourable due to 4 moles on left and 2 moles on right. Increased pressure causes a shift to the left, favouring product formation. This is expensive to due and most production plants will resist compressing gases in terms of operating costs. Compromise will be met. ...
... High pressure is favourable due to 4 moles on left and 2 moles on right. Increased pressure causes a shift to the left, favouring product formation. This is expensive to due and most production plants will resist compressing gases in terms of operating costs. Compromise will be met. ...
mclintock.ch6 [Compatibility Mode]
... ► The numbers and kinds of atoms must be the same on both sides of the reaction arrow. ► Numbers in front of formulas are called coefficients; they multiply all the atoms in a formula. ► The symbol 2 NaHCO3 indicates two units of sodium bicarbonate, which contains 2 Na, 2 H, 2 C, and 6 O. ► Substanc ...
... ► The numbers and kinds of atoms must be the same on both sides of the reaction arrow. ► Numbers in front of formulas are called coefficients; they multiply all the atoms in a formula. ► The symbol 2 NaHCO3 indicates two units of sodium bicarbonate, which contains 2 Na, 2 H, 2 C, and 6 O. ► Substanc ...
Stoichiometry
... The compound para-aminobenzoic acid (you may have seen it listed as PABA on your bottle of sunscreen) is composed of carbon (61.31%), hydrogen (5.14%), nitrogen (10.21%), and oxygen (23.33%). Find the empirical formula of PABA. ...
... The compound para-aminobenzoic acid (you may have seen it listed as PABA on your bottle of sunscreen) is composed of carbon (61.31%), hydrogen (5.14%), nitrogen (10.21%), and oxygen (23.33%). Find the empirical formula of PABA. ...
Chapter 4 Classifying Reactions: Chemicals in Balance
... The name and state of each reactant and product are given. Plan Your Strategy A skeleton equation lists the chemical formula of each reactant on the left, separated by a + sign if more than one reactant is present, followed by →. The chemical formula of each product is listed on the right, separated ...
... The name and state of each reactant and product are given. Plan Your Strategy A skeleton equation lists the chemical formula of each reactant on the left, separated by a + sign if more than one reactant is present, followed by →. The chemical formula of each product is listed on the right, separated ...
1. Blood cholesterol levels are generally expressed as milligrams of
... (called Mylar). Your friend knows you are taking chemistry and asks you why the helium balloon is made of a different material than regular balloons blown up with air. (For this example, simplify by assuming air used in regular balloons is 100% N2.) Give your friend an answer in words supported by 1 ...
... (called Mylar). Your friend knows you are taking chemistry and asks you why the helium balloon is made of a different material than regular balloons blown up with air. (For this example, simplify by assuming air used in regular balloons is 100% N2.) Give your friend an answer in words supported by 1 ...
Document
... container holding the equilibrium mixture of N2, O2, and NO is decreased to 0.5 L without changing the quantities of the gases present, how will their concentrations change? A) The concentration of NO will increase; the concentrations of N2 and O2 will decrease. B) The concentrations of N2 and O2 wi ...
... container holding the equilibrium mixture of N2, O2, and NO is decreased to 0.5 L without changing the quantities of the gases present, how will their concentrations change? A) The concentration of NO will increase; the concentrations of N2 and O2 will decrease. B) The concentrations of N2 and O2 wi ...
Chapter 3 - Educator
... Once we know the formulas of the reactants and products in a reaction, we can write the unbalanced equation. We then balance the equation by determining the coefficients that provide equal numbers of each type of atom on each side of the equation. For most purposes, a balanced equation should contai ...
... Once we know the formulas of the reactants and products in a reaction, we can write the unbalanced equation. We then balance the equation by determining the coefficients that provide equal numbers of each type of atom on each side of the equation. For most purposes, a balanced equation should contai ...
Chemical Quantities
... To understand the molecular and mass information given in a balanced equation. Reactions are what chemistry is really all about. Recall from Chapter 6 that chemical changes are actually rearrangements of atom groupings that can be described by chemical equations. These chemical equations tell us the ...
... To understand the molecular and mass information given in a balanced equation. Reactions are what chemistry is really all about. Recall from Chapter 6 that chemical changes are actually rearrangements of atom groupings that can be described by chemical equations. These chemical equations tell us the ...












![mclintock.ch6 [Compatibility Mode]](http://s1.studyres.com/store/data/003971396_1-780a12aa3165c9221aca3ac594a06674-300x300.png)







