Preparation of Reducing Sugar Hydrolyzed from High
... crystalline orientation of cellulose, thus improving the yield of fermentable sugar in enzymatic hydrolysis [2-4]. Chemical methods have been employed to carry out pretreatment on grape stalks to regain both hemicellulose and cellulose [5]. However, the wastewater, which contained chemical substance ...
... crystalline orientation of cellulose, thus improving the yield of fermentable sugar in enzymatic hydrolysis [2-4]. Chemical methods have been employed to carry out pretreatment on grape stalks to regain both hemicellulose and cellulose [5]. However, the wastewater, which contained chemical substance ...
purdue university - IUPUI ScholarWorks
... Teaching, Research, and Outreach Policy on Research Misconduct (VIII.3.1), October 1, 2008.* Further, I certify that this work is free of plagiarism and all materials appearing in this thesis/dissertation have been properly quoted and attributed. I certify that all copyrighted material incorporated ...
... Teaching, Research, and Outreach Policy on Research Misconduct (VIII.3.1), October 1, 2008.* Further, I certify that this work is free of plagiarism and all materials appearing in this thesis/dissertation have been properly quoted and attributed. I certify that all copyrighted material incorporated ...
GCSE Chemistry Sample Assessment Materials
... Crossed out responses not replaced should be marked. Credit will be given for correct and relevant alternative responses which are not recorded in the mark scheme. Extended response question A level of response mark scheme is used. Before applying the mark scheme please read through the whole answer ...
... Crossed out responses not replaced should be marked. Credit will be given for correct and relevant alternative responses which are not recorded in the mark scheme. Extended response question A level of response mark scheme is used. Before applying the mark scheme please read through the whole answer ...
Spring 2016 CHEM Handbook - Chemistry | Virginia Tech
... Chemistry is central to the sciences, the understanding of the physical world, and the study of biological systems. Chemistry is the science of transformations and energetics of materials at the molecular level. Chemistry has applications from the nanoscale to the macroscopic. Chemists use their tra ...
... Chemistry is central to the sciences, the understanding of the physical world, and the study of biological systems. Chemistry is the science of transformations and energetics of materials at the molecular level. Chemistry has applications from the nanoscale to the macroscopic. Chemists use their tra ...
study guide spring 2012
... A chemical equation is balanced when the a. coefficients of the reactants equal the coefficients of the products. b. same number of each kind of atom appears in the reactants and in the products. c. products and reactants are the same chemicals. d. subscripts of the reactants equal the subscripts of ...
... A chemical equation is balanced when the a. coefficients of the reactants equal the coefficients of the products. b. same number of each kind of atom appears in the reactants and in the products. c. products and reactants are the same chemicals. d. subscripts of the reactants equal the subscripts of ...
Stoichiometery
... A more typical problem 2 H2 + O2 → 2 H2O What masses of oxygen and hydrogen are required to create 5.0 g H2O? 5.0 g H2O * 1 mol H2O * 1 mol O2 * 32.0 g O2 = 4.44 g O2 18.016 g H2O 2 mol H2O 1 mol O2 5.0 g H2O * 1 mol H2O * 2 mol H2 * 2.016 g H2 = 0.56 g H2 18.016 g H2O 2 mol H2O 1 mol H2 ...
... A more typical problem 2 H2 + O2 → 2 H2O What masses of oxygen and hydrogen are required to create 5.0 g H2O? 5.0 g H2O * 1 mol H2O * 1 mol O2 * 32.0 g O2 = 4.44 g O2 18.016 g H2O 2 mol H2O 1 mol O2 5.0 g H2O * 1 mol H2O * 2 mol H2 * 2.016 g H2 = 0.56 g H2 18.016 g H2O 2 mol H2O 1 mol H2 ...
Amines - ncert
... Due to the electron releasing nature of alkyl group, it (R) pushes electrons towards nitrogen and thus makes the unshared electron pair more available for sharing with the proton of the acid. Moreover, the substituted ammonium ion formed from the amine gets stabilised due to dispersal of the positiv ...
... Due to the electron releasing nature of alkyl group, it (R) pushes electrons towards nitrogen and thus makes the unshared electron pair more available for sharing with the proton of the acid. Moreover, the substituted ammonium ion formed from the amine gets stabilised due to dispersal of the positiv ...
Atmospheric Formation_TELTEK
... predicted that unsubstituted alkylamines will primarily be oxidized in the gas phase, and that alkanolamines will primarily be oxidized in the atmospheric aqueous phase. Existing theoretical gas phase degradation schemes have been updated for methylamine, dimethylamine and trimethylamine, and the th ...
... predicted that unsubstituted alkylamines will primarily be oxidized in the gas phase, and that alkanolamines will primarily be oxidized in the atmospheric aqueous phase. Existing theoretical gas phase degradation schemes have been updated for methylamine, dimethylamine and trimethylamine, and the th ...
Chapter 6
... row as reactants, and those on the right side of the equilibrium arrow as products. As Berthollet discovered, writing a reaction in this fashion does not guarantee that the reaction of A and B to produce C and D is favorable. Depending on initial conditions, the reaction may move to the left, move t ...
... row as reactants, and those on the right side of the equilibrium arrow as products. As Berthollet discovered, writing a reaction in this fashion does not guarantee that the reaction of A and B to produce C and D is favorable. Depending on initial conditions, the reaction may move to the left, move t ...
Slide 1
... requires the addition of heat, which shifts the equilibrium to the left and reduces the concentration of hydrogen chloride. • Thus, the value of Keq decreases. • Lowering the temperature of the system means that heat is removed, so the equilibrium relieves the stress by shifting to the right, increa ...
... requires the addition of heat, which shifts the equilibrium to the left and reduces the concentration of hydrogen chloride. • Thus, the value of Keq decreases. • Lowering the temperature of the system means that heat is removed, so the equilibrium relieves the stress by shifting to the right, increa ...
SQA Advanced Higher Chemistry Unit 2 Principles of Chemical
... there in this sample of iron ore? How pure is this sample of anti-viral drug?) It is also critical for industrial chemistry. (How many tonnes of sulphuric acid and phosphate rock are required to manufacture this batch of fertiliser? What proportion of monomers are required to prepare this plastic?) ...
... there in this sample of iron ore? How pure is this sample of anti-viral drug?) It is also critical for industrial chemistry. (How many tonnes of sulphuric acid and phosphate rock are required to manufacture this batch of fertiliser? What proportion of monomers are required to prepare this plastic?) ...
1 mol H 2
... factors in stoichiometric calculations With the chemical equation and the mole ratios, you can calculate the amount used of any reactant in the equation and the maximum amount of product you can obtain. ...
... factors in stoichiometric calculations With the chemical equation and the mole ratios, you can calculate the amount used of any reactant in the equation and the maximum amount of product you can obtain. ...
Theoretical Investigation of the Water
... for his encouragements to be strong. I thank God for helping me all the time. ...
... for his encouragements to be strong. I thank God for helping me all the time. ...
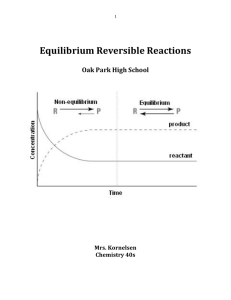
Equilibrium Reversible Reactions
... In graph two which components start with a concentration of 0mol/L? _____________ Are these the products or reactants? _____________________________________ This shows that these two graphs represent the same chemical reaction system, but with the reactions occurring in opposite directions. Most imp ...
... In graph two which components start with a concentration of 0mol/L? _____________ Are these the products or reactants? _____________________________________ This shows that these two graphs represent the same chemical reaction system, but with the reactions occurring in opposite directions. Most imp ...
Question Bank for Pre Board Exam(XII Chemistry)
... 37.In Corundum, O2– ions from hcp and Al3+ occupy two third of octahedral voids. Determine the formula of corundum. 38.Why is Frenkel defect not found in pure alkali metal halides? 39.Which point defect is observed in a crystal when a vacancy is created by an atom missing from a lattice site. 40. Wh ...
... 37.In Corundum, O2– ions from hcp and Al3+ occupy two third of octahedral voids. Determine the formula of corundum. 38.Why is Frenkel defect not found in pure alkali metal halides? 39.Which point defect is observed in a crystal when a vacancy is created by an atom missing from a lattice site. 40. Wh ...
Problem Authors - PianetaChimica
... contaminations. The reaction of sulfuric acid with colemanite takes place in two steps: In the first step colemanite is dissolved in sulfuric acid forming the calcium(II) ion and boric acid. In the second step, calcium sulfate, formed from Ca2+ and SO42− ions, precipitates as gypsum crystals. In an ...
... contaminations. The reaction of sulfuric acid with colemanite takes place in two steps: In the first step colemanite is dissolved in sulfuric acid forming the calcium(II) ion and boric acid. In the second step, calcium sulfate, formed from Ca2+ and SO42− ions, precipitates as gypsum crystals. In an ...
Stoichiometry
... Use reaction coefficients as a molar ratio Convert moles of your unknown into the desired units ...
... Use reaction coefficients as a molar ratio Convert moles of your unknown into the desired units ...
x - mrs. leinweber`s wiki
... two different acids (or bases) can have the same [ ] but have different strengths eg) 1 M CH3COOH(aq) and 1 M HCl(aq) will react in the same way but not to the same degree the stronger the acid, the more electricity it conducts, ...
... two different acids (or bases) can have the same [ ] but have different strengths eg) 1 M CH3COOH(aq) and 1 M HCl(aq) will react in the same way but not to the same degree the stronger the acid, the more electricity it conducts, ...
Communicating Chemistry Landscape Study
... (BCST) and Board on Science Education (BOSE) received funding from the National Science Foundation (NSF) to develop a framework for effective chemistry communication, outreach, and education in informal settings, with the ultimate goal of increasing the effectiveness of such efforts in engaging the ...
... (BCST) and Board on Science Education (BOSE) received funding from the National Science Foundation (NSF) to develop a framework for effective chemistry communication, outreach, and education in informal settings, with the ultimate goal of increasing the effectiveness of such efforts in engaging the ...
Stoichiometric Calculations
... The limiting reactant, or limiting reagent, is the reactant present in the smallest stoichiometric amount. This is not necessarily the one with the smallest mass. The limiting reactant is the reactant you’ll run out of first, and it is the one that determines the maximum amount of product that can b ...
... The limiting reactant, or limiting reagent, is the reactant present in the smallest stoichiometric amount. This is not necessarily the one with the smallest mass. The limiting reactant is the reactant you’ll run out of first, and it is the one that determines the maximum amount of product that can b ...
Stoichiometric Calculations
... The limiting reactant, or limiting reagent, is the reactant present in the smallest stoichiometric amount. This is not necessarily the one with the smallest mass. The limiting reactant is the reactant you’ll run out of first, and it is the one that determines the maximum amount of product that can b ...
... The limiting reactant, or limiting reagent, is the reactant present in the smallest stoichiometric amount. This is not necessarily the one with the smallest mass. The limiting reactant is the reactant you’ll run out of first, and it is the one that determines the maximum amount of product that can b ...