ProteinChipâ technology is one of the most exciting advancements
... ProteinChip technology is one of the most exciting advancements in protein analysis in the last 5 years. The Protein Biology SystemTM (PBS) combines the power of mass analysis with chromatography surfaces on an integrated platform. The PBS can easily be used by biologists, biochemists, and clinicia ...
... ProteinChip technology is one of the most exciting advancements in protein analysis in the last 5 years. The Protein Biology SystemTM (PBS) combines the power of mass analysis with chromatography surfaces on an integrated platform. The PBS can easily be used by biologists, biochemists, and clinicia ...
Protein misfolding associated to mild modifications of local cellular pH
... and the hydrophobic cavities present in the native state of the protein. This means that misfolding could be associated with intermediate folding states, and protonation of residues. Even though natural pathological mutants show higher tendency to aggregate as amyloid-like structures, wt apoA-I also ...
... and the hydrophobic cavities present in the native state of the protein. This means that misfolding could be associated with intermediate folding states, and protonation of residues. Even though natural pathological mutants show higher tendency to aggregate as amyloid-like structures, wt apoA-I also ...
Estimation of the protein secondary structure in aqueous solutions
... Estimation of the protein secondary structure in aqueous solutions based on IR absorption spectra. N.M.Romanov The secondary structure of proteins is very important for their proper functioning. The investigation of the secondary structure gives us an insight into the mechanisms of protein functioni ...
... Estimation of the protein secondary structure in aqueous solutions based on IR absorption spectra. N.M.Romanov The secondary structure of proteins is very important for their proper functioning. The investigation of the secondary structure gives us an insight into the mechanisms of protein functioni ...
Protein: How Cows and Carrots Become People 1. Your body can
... 9. Believe it or not, you have hydrochloric acid in your stomach. It’s nasty stuff that can burn skin and eat away at almost anything. What is it doing in your stomach and why ...
... 9. Believe it or not, you have hydrochloric acid in your stomach. It’s nasty stuff that can burn skin and eat away at almost anything. What is it doing in your stomach and why ...
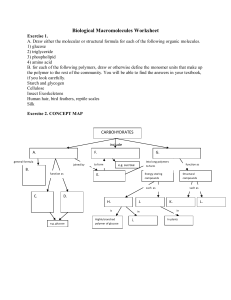
Biological Macromolecules Worksheet
... Exercise 3. 1. A triglyceride contains ______ and _______. 2. A fatty acid is unsaturated if it contains ____________. 3. Saturated fatty acids and unsaturated fatty acids differ in ___________. 4. Explain why phospholipids form a bilayer membrane. Exercise 4. Define what a protein is and/or of wha ...
... Exercise 3. 1. A triglyceride contains ______ and _______. 2. A fatty acid is unsaturated if it contains ____________. 3. Saturated fatty acids and unsaturated fatty acids differ in ___________. 4. Explain why phospholipids form a bilayer membrane. Exercise 4. Define what a protein is and/or of wha ...
6. 3-D structure of proteins
... • It is left-handed and had three amino acid residues per term. ...
... • It is left-handed and had three amino acid residues per term. ...
Proteins
... Primary: The amino acid sequence. – This is simply a list of the actual sequence of amino acids in the "backbone" of the protein. – You just start at one end and read off the names of the amino acids in sequence and you have described the primary sequence. ...
... Primary: The amino acid sequence. – This is simply a list of the actual sequence of amino acids in the "backbone" of the protein. – You just start at one end and read off the names of the amino acids in sequence and you have described the primary sequence. ...
Capturing denaturing proteins * Small Heat Shock Protein substrate
... Capturing denaturing proteins – Small Heat Shock Protein substrate recognition Elizabeth Vierling and Indu Santhanagopalan Protein aggregation resulting from stress, disease or mutation poses a major threat to all cells. The ubiquitous small heat shock proteins (sHSPs) act as molecular chaperones to ...
... Capturing denaturing proteins – Small Heat Shock Protein substrate recognition Elizabeth Vierling and Indu Santhanagopalan Protein aggregation resulting from stress, disease or mutation poses a major threat to all cells. The ubiquitous small heat shock proteins (sHSPs) act as molecular chaperones to ...
The_Structure_of_Protein_Activity
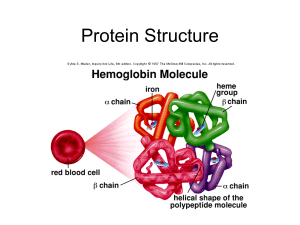
... Primary structure sequence of amino acids in polypeptide chain or chains; Secondary structure describes the repeating patterns in the of sections of the polypeptide chains (example: fibrous proteins such as α-keratin in hair have helical chains of amino acids held together by hydrogen bonds); Tertia ...
... Primary structure sequence of amino acids in polypeptide chain or chains; Secondary structure describes the repeating patterns in the of sections of the polypeptide chains (example: fibrous proteins such as α-keratin in hair have helical chains of amino acids held together by hydrogen bonds); Tertia ...
Proteins are biopolymers construced from similar building blocks
... Proteins are biopolymers construced from similar building blocks called amino acids. The unique feature is that these polypeptide chains are folded in a certain three-dimensional structure (called native structure), which enables them to perform their biological funtion. Studies on protein structure ...
... Proteins are biopolymers construced from similar building blocks called amino acids. The unique feature is that these polypeptide chains are folded in a certain three-dimensional structure (called native structure), which enables them to perform their biological funtion. Studies on protein structure ...
Lecture 1
... hydrogen bonds with Ala residues located in an ahelix? A. Residues in a neighbouring a-helix. ...
... hydrogen bonds with Ala residues located in an ahelix? A. Residues in a neighbouring a-helix. ...
CH 107 SI Summer 2015 Worksheet 13 Answers What are the two
... α-helices and β-sheets Hydrogen bonds 2. What types of interactions can be present in tertiary protein structure? Rank the interactions from strongest to weakest. disulfide bonds >> salt bridges > hydrogen bonds > hydrophilic interactions > hydrophobic interactions 3. Compare and contrast globular a ...
... α-helices and β-sheets Hydrogen bonds 2. What types of interactions can be present in tertiary protein structure? Rank the interactions from strongest to weakest. disulfide bonds >> salt bridges > hydrogen bonds > hydrophilic interactions > hydrophobic interactions 3. Compare and contrast globular a ...
A dead-end street of protein folding
... expected to lie in the protein backbone, common to all proteins.(Nelson, R. et al. Nature 2005, 435, 773-778., and Wright, C. F. etal. Nature 2005, 438, 878-881.) These structures show low sequence specificity and have the common motif of multiple stranded -pleated sheets. For dozens of ordinary pr ...
... expected to lie in the protein backbone, common to all proteins.(Nelson, R. et al. Nature 2005, 435, 773-778., and Wright, C. F. etal. Nature 2005, 438, 878-881.) These structures show low sequence specificity and have the common motif of multiple stranded -pleated sheets. For dozens of ordinary pr ...
Protein domain

A protein domain is a conserved part of a given protein sequence and (tertiary) structure that can evolve, function, and exist independently of the rest of the protein chain. Each domain forms a compact three-dimensional structure and often can be independently stable and folded. Many proteins consist of several structural domains. One domain may appear in a variety of different proteins. Molecular evolution uses domains as building blocks and these may be recombined in different arrangements to create proteins with different functions. Domains vary in length from between about 25 amino acids up to 500 amino acids in length. The shortest domains such as zinc fingers are stabilized by metal ions or disulfide bridges. Domains often form functional units, such as the calcium-binding EF hand domain of calmodulin. Because they are independently stable, domains can be ""swapped"" by genetic engineering between one protein and another to make chimeric proteins.