Thermodynamics - Faculty
... 1. In the early 1800s, Carnot pointed out the basic working of an ideal (one without internal friction) heat engine. 2. The Carnot cycle (see Figure 12.17 in your textbook) can be described in 4 steps: a) Step 1: The cycle starts with the piston positioned such that V is at a minimum. At this point, ...
... 1. In the early 1800s, Carnot pointed out the basic working of an ideal (one without internal friction) heat engine. 2. The Carnot cycle (see Figure 12.17 in your textbook) can be described in 4 steps: a) Step 1: The cycle starts with the piston positioned such that V is at a minimum. At this point, ...
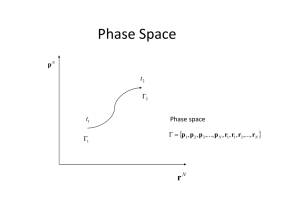
Phase Space Phase Space
... Within the statistical description, the motion of one single system with given initial conditions is not considered; thus, p(t), q(t) are not sought. Instead, the motion of a whole set of phase points, representing the collection of possible states of the given system. Such a set of phase points is ...
... Within the statistical description, the motion of one single system with given initial conditions is not considered; thus, p(t), q(t) are not sought. Instead, the motion of a whole set of phase points, representing the collection of possible states of the given system. Such a set of phase points is ...
Examples Paper 2 (1-2)
... The second law in differential form for a closed system undergoing a reversible process is dQ = TdS and hence dW = TdS – dU for such a process. For our system undergoing the process with dT = 0, we have d(TS) = TdS, so that dW = d(TS) – dU = -d(U – TS) = -dF. This expression says that the negative ...
... The second law in differential form for a closed system undergoing a reversible process is dQ = TdS and hence dW = TdS – dU for such a process. For our system undergoing the process with dT = 0, we have d(TS) = TdS, so that dW = d(TS) – dU = -d(U – TS) = -dF. This expression says that the negative ...
solutions
... • Zeroth: If two systems are both in thermal equilibrium with a third then they are in thermal equilibrium with each other. • First: The increase in internal energy of a closed system is equal to the heat supplied to the system minus work done by it. • Second: The entropy of any isolated system neve ...
... • Zeroth: If two systems are both in thermal equilibrium with a third then they are in thermal equilibrium with each other. • First: The increase in internal energy of a closed system is equal to the heat supplied to the system minus work done by it. • Second: The entropy of any isolated system neve ...
Chemical Bonds in Biochemistry - Biochemistry
... 1.3.3. Entropy and the Laws of Thermodynamics The highly structured, organized nature of living organisms is apparent and astonishing. This organization extends from the organismal through the cellular to the molecular level. Indeed, biological processes can seem magical in that the well-ordered str ...
... 1.3.3. Entropy and the Laws of Thermodynamics The highly structured, organized nature of living organisms is apparent and astonishing. This organization extends from the organismal through the cellular to the molecular level. Indeed, biological processes can seem magical in that the well-ordered str ...
Max Planck: the reluctant revolutionary Helge Kaigh, Physics World
... the life of one of them," he predicted. As to the outcome of the …ght, he wrote that "in spite of the great successes of the atomistic theory in the past, we will …nally have to give it up and to decide in favour of the assumption of continuous matter". However, Planck’s opposition to atomism waned ...
... the life of one of them," he predicted. As to the outcome of the …ght, he wrote that "in spite of the great successes of the atomistic theory in the past, we will …nally have to give it up and to decide in favour of the assumption of continuous matter". However, Planck’s opposition to atomism waned ...
Friction force: from mechanics to thermodynamics
... It is well known that thermodynamics was first developed by Carnot in 1824 to study the production and the transport of heat1 . However since the axiomatic work of Carathéodory in 1909 on equilibrium thermodynamics2 , also called thermostatics, one has very often considered thermodynamics as a theo ...
... It is well known that thermodynamics was first developed by Carnot in 1824 to study the production and the transport of heat1 . However since the axiomatic work of Carathéodory in 1909 on equilibrium thermodynamics2 , also called thermostatics, one has very often considered thermodynamics as a theo ...
2 nd Law of Thermodynamics
... • Strictly speaking, this statement is true ONLY if the quantum mechanical ground state is nondegenerate. If it is degenerate, the entropy at T = 0 K is a small constant & not 0! ...
... • Strictly speaking, this statement is true ONLY if the quantum mechanical ground state is nondegenerate. If it is degenerate, the entropy at T = 0 K is a small constant & not 0! ...
Classical thermodynamics of particles in harmonic traps
... intuition that we develop in studying ideal gases in rigid containers. It might seem more natural to express the term describing mechanical work as −b⌬A instead of A⌬b. The former would make the coefficient of the differential element an intensive variable 共analogous to pressure in the familiar firs ...
... intuition that we develop in studying ideal gases in rigid containers. It might seem more natural to express the term describing mechanical work as −b⌬A instead of A⌬b. The former would make the coefficient of the differential element an intensive variable 共analogous to pressure in the familiar firs ...
Document
... work and reaffirms the principle of conservation of energy. The second law states that heat does not of itself pass from a cooler to a hotter body. Another, equivalent, formulation of the second law is that the entropy of a closed system can only increase. The third law (also called Nernst's heat th ...
... work and reaffirms the principle of conservation of energy. The second law states that heat does not of itself pass from a cooler to a hotter body. Another, equivalent, formulation of the second law is that the entropy of a closed system can only increase. The third law (also called Nernst's heat th ...
The Helmholtz Function
... The quantity G is tabulated for a variety of chemical reactions and other processes. G = H - TS In most work we do not need to choose a reference point as, usually, only changes in the quantities are of interest. {One source of tabulations of thermodynamic properties is the CRC Handbook of Chem ...
... The quantity G is tabulated for a variety of chemical reactions and other processes. G = H - TS In most work we do not need to choose a reference point as, usually, only changes in the quantities are of interest. {One source of tabulations of thermodynamic properties is the CRC Handbook of Chem ...
Lecture VIII_IX
... with Cv,m = 1.5 R, initially at 298 K and 10 L, is expanded, with the surroundings maintained at 298 K, to a final volume of 20 L, in three ways (a) isothermally and reversibly (b) isothermally against a constant external pressure of 0.5 atm (c) adiabatically against a constant external pressure of ...
... with Cv,m = 1.5 R, initially at 298 K and 10 L, is expanded, with the surroundings maintained at 298 K, to a final volume of 20 L, in three ways (a) isothermally and reversibly (b) isothermally against a constant external pressure of 0.5 atm (c) adiabatically against a constant external pressure of ...
1 - WordPress.com
... c) (6 points) On the same PV diagram, draw the two-step process corresponding to an isothermal expansion from state 1 to volume V2, followed by isovolumetric increase in temperature to state 2, and label this process B. Show the direction in which the process goes with arrows. d) (8 points) Find the ...
... c) (6 points) On the same PV diagram, draw the two-step process corresponding to an isothermal expansion from state 1 to volume V2, followed by isovolumetric increase in temperature to state 2, and label this process B. Show the direction in which the process goes with arrows. d) (8 points) Find the ...
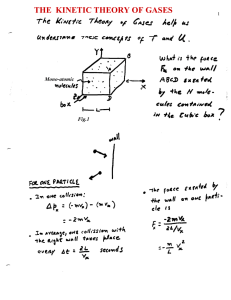
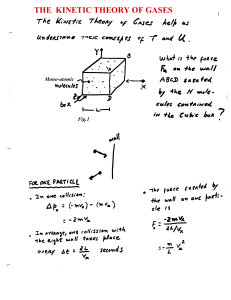
the kinetic theory of gases
... Question: Is expression (9) still valid when considering diatomic molecules? First, let’s put expression (9) again in terms of the average kinetic energy per molecule, PV = (2/3) U = (2/3) N < ½ m v2 > (mono-atomic gas) Notice that, during its derivation we were invoking the collision of mono-atomic ...
... Question: Is expression (9) still valid when considering diatomic molecules? First, let’s put expression (9) again in terms of the average kinetic energy per molecule, PV = (2/3) U = (2/3) N < ½ m v2 > (mono-atomic gas) Notice that, during its derivation we were invoking the collision of mono-atomic ...
H-theorem

In classical statistical mechanics, the H-theorem, introduced by Ludwig Boltzmann in 1872, describes the tendency to increase in the quantity H (defined below) in a nearly-ideal gas of molecules. As this quantity H was meant to represent the entropy of thermodynamics, the H-theorem was an early demonstration of the power of statistical mechanics as it claimed to derive the second law of thermodynamics—a statement about fundamentally irreversible processes—from reversible microscopic mechanics.The H-theorem is a natural consequence of the kinetic equation derived by Boltzmann that has come to be known as Boltzmann's equation. The H-theorem has led to considerable discussion about its actual implications, with major themes being: What is entropy? In what sense does Boltzmann's quantity H correspond to the thermodynamic entropy? Are the assumptions (such as the Stosszahlansatz described below) behind Boltzmann's equation too strong? When are these assumptions violated?↑