heat processes
... In the present work, a new shell-and-tube heat exchanger optimization design approach is developed, wherein the dimensionless entropy generation rate obtained by scaling the entropy generation on the ratio of the heat transfer rate to the inlet temperature of cold fluid is employed as the objective ...
... In the present work, a new shell-and-tube heat exchanger optimization design approach is developed, wherein the dimensionless entropy generation rate obtained by scaling the entropy generation on the ratio of the heat transfer rate to the inlet temperature of cold fluid is employed as the objective ...
Basic Thermodynamics - CERN Accelerator School
... a gas in a box, and assume that the gas is on one side of the box at some initial time corresponding to 1 . If the gas is free to fill up the whole box, corresponding to 2 , it will ‘spontaneously’ go to that state. Indeed, 2 1 , and there is a higher probability of finding the gas t ...
... a gas in a box, and assume that the gas is on one side of the box at some initial time corresponding to 1 . If the gas is free to fill up the whole box, corresponding to 2 , it will ‘spontaneously’ go to that state. Indeed, 2 1 , and there is a higher probability of finding the gas t ...
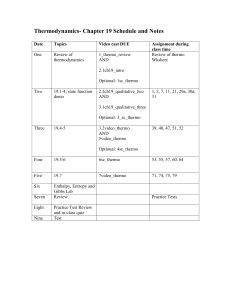
Schedule and sample problems
... (a) The sign of ∆S˚ is (+). There is an increase in the number of gas molecules as well as a change from a pure gas to a mixture of gases. (b) ∆G˚ = ∆H˚ – T∆S˚. Both ∆S˚ and ∆H˚ are (+). As temperature increases, at some point the sign of ∆G˚ will change from (+) to (–), when the system will become ...
... (a) The sign of ∆S˚ is (+). There is an increase in the number of gas molecules as well as a change from a pure gas to a mixture of gases. (b) ∆G˚ = ∆H˚ – T∆S˚. Both ∆S˚ and ∆H˚ are (+). As temperature increases, at some point the sign of ∆G˚ will change from (+) to (–), when the system will become ...
Physics 201 - University of Virginia
... Order, Disorder, and Entropy As we have stated, entropy is related to disorder. As the entropy of a system increases, its disorder increases as well. GOOD NEWS: When you go home, and your mother fusses about how messy your bedroom is, tell her it is because entropy is increasing, and it is the natu ...
... Order, Disorder, and Entropy As we have stated, entropy is related to disorder. As the entropy of a system increases, its disorder increases as well. GOOD NEWS: When you go home, and your mother fusses about how messy your bedroom is, tell her it is because entropy is increasing, and it is the natu ...
heat engine
... temperature, and all the rejected heat goes into a cold reservoir at a single temperature. Since the efficiency can only depend on the reservoir temperatures, the ratio of heats can only depend on those temperatures. QC QH ...
... temperature, and all the rejected heat goes into a cold reservoir at a single temperature. Since the efficiency can only depend on the reservoir temperatures, the ratio of heats can only depend on those temperatures. QC QH ...
4.1 The Concepts of Force and Mass
... temperature, and all the rejected heat goes into a cold reservoir at a single temperature. Since the efficiency can only depend on the reservoir temperatures, the ratio of heats can only depend on those temperatures. QC QH ...
... temperature, and all the rejected heat goes into a cold reservoir at a single temperature. Since the efficiency can only depend on the reservoir temperatures, the ratio of heats can only depend on those temperatures. QC QH ...
Chapter 17. Statistical thermodynamics 2: applications
... • Method: Using eqn 17.9. For the standard value, we evaluate the translational partition function at p (105 Pa). The vibrational partition function was calculated in Ex.17.3. Use the expressions in Table 17-3 for the other contributions. • Answer: As m = 18.015 u, q mT /NA = 1.706 × 108. For • Vibr ...
... • Method: Using eqn 17.9. For the standard value, we evaluate the translational partition function at p (105 Pa). The vibrational partition function was calculated in Ex.17.3. Use the expressions in Table 17-3 for the other contributions. • Answer: As m = 18.015 u, q mT /NA = 1.706 × 108. For • Vibr ...
Thermal Properties of Matter
... The ideal gas law gives the relationship between pressure, temperature, volume and mass, all state variables. The quantity, n, is the mole count, I.e. the number of moles present in the gas, and therefore the mass of the gas is given by: ...
... The ideal gas law gives the relationship between pressure, temperature, volume and mass, all state variables. The quantity, n, is the mole count, I.e. the number of moles present in the gas, and therefore the mass of the gas is given by: ...
Chemistry 205 - Introductory General Chemistry
... See attached CNSM withdrawal policy. Note, if you wish to withdraw because of poor grades you must do so before the end of the eighth week of class. By this time you will have ample graded material by which to judge your performance. If you are unsure whether you will pass, or need help to do better ...
... See attached CNSM withdrawal policy. Note, if you wish to withdraw because of poor grades you must do so before the end of the eighth week of class. By this time you will have ample graded material by which to judge your performance. If you are unsure whether you will pass, or need help to do better ...
H-theorem

In classical statistical mechanics, the H-theorem, introduced by Ludwig Boltzmann in 1872, describes the tendency to increase in the quantity H (defined below) in a nearly-ideal gas of molecules. As this quantity H was meant to represent the entropy of thermodynamics, the H-theorem was an early demonstration of the power of statistical mechanics as it claimed to derive the second law of thermodynamics—a statement about fundamentally irreversible processes—from reversible microscopic mechanics.The H-theorem is a natural consequence of the kinetic equation derived by Boltzmann that has come to be known as Boltzmann's equation. The H-theorem has led to considerable discussion about its actual implications, with major themes being: What is entropy? In what sense does Boltzmann's quantity H correspond to the thermodynamic entropy? Are the assumptions (such as the Stosszahlansatz described below) behind Boltzmann's equation too strong? When are these assumptions violated?↑