Amino Acid Alphabet
... Mutate multiple residues simultaneously to see if a large set of amino acids can be altered in combination to a much smaller set without perturbing the structure and function irreversibly Residues 39 to 50 T4 lysozyme form an alpha helix Mutate residues 40 to 49 with 10 consecutive Ala ...
... Mutate multiple residues simultaneously to see if a large set of amino acids can be altered in combination to a much smaller set without perturbing the structure and function irreversibly Residues 39 to 50 T4 lysozyme form an alpha helix Mutate residues 40 to 49 with 10 consecutive Ala ...
http://gslc. genetics. utah.edu/units/basics/transcribe/
... http:// gslc. genetics. utah.edu/units/basics/transcribe/ Defme the following terms: Transcription, Translation, Codon Complete the "Build a Protein" Activity You will need to record the sequence of bases in the mRNA as well as the sequence of amino acids on a separate piece of paper that I will col ...
... http:// gslc. genetics. utah.edu/units/basics/transcribe/ Defme the following terms: Transcription, Translation, Codon Complete the "Build a Protein" Activity You will need to record the sequence of bases in the mRNA as well as the sequence of amino acids on a separate piece of paper that I will col ...
CH 460 Dr. Muccio What are the 4 levels of protein structure and
... What are the four types of secondary structure? What are they stabilized by? Can you draw them? Alpha helix – h-bonds in the same chain, few steric interaction Beta sheets - h-bonds between neighboring beta-strands, few steric interaction Beta turn – ONE h-bond, few steric interactions Loop/random – ...
... What are the four types of secondary structure? What are they stabilized by? Can you draw them? Alpha helix – h-bonds in the same chain, few steric interaction Beta sheets - h-bonds between neighboring beta-strands, few steric interaction Beta turn – ONE h-bond, few steric interactions Loop/random – ...
HW #6 BP401/P475 Fall 2015 Assigned Fr 10/02/15: due: Thursday
... isolated unique secondary structure, according to the energy rule? e. Secondary structure is never isolated in proteins. Introducing three black circles as “hydrophobic amino acids,” draw a structure of a perfect amphipathic (having both a hydrophobic and hydrophilic) helix on the lattice. Let us sa ...
... isolated unique secondary structure, according to the energy rule? e. Secondary structure is never isolated in proteins. Introducing three black circles as “hydrophobic amino acids,” draw a structure of a perfect amphipathic (having both a hydrophobic and hydrophilic) helix on the lattice. Let us sa ...
Proteins - CasimiroSBI4U
... Peptide bond = covalent bond formed by condensation reaction that links carboxyl group of one amino acid to amino group of another. ...
... Peptide bond = covalent bond formed by condensation reaction that links carboxyl group of one amino acid to amino group of another. ...
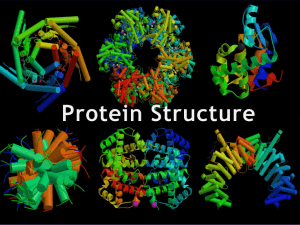
Protein structure
... • All peptide unit planes are roughly parallel to the helix axis • Each peptide bond is a small dipole • The dipoles within the helix are aligned, i.e. all C=O groups point in the same direction and all N-H groups point the other way • The helix becomes a net dipole with +0.5 charge units at the N-t ...
... • All peptide unit planes are roughly parallel to the helix axis • Each peptide bond is a small dipole • The dipoles within the helix are aligned, i.e. all C=O groups point in the same direction and all N-H groups point the other way • The helix becomes a net dipole with +0.5 charge units at the N-t ...
Biological Macromolecules Worksheet
... a. the number _____ of different nitrogenous bases in DNA b. the number _____ of different chemical classes of amino acids c. the number _____ of chains of nucleotides in a DNA molecule d. the number _____ of different nitrogenous bases in RNA e. the number _____ of different amino acids found in pr ...
... a. the number _____ of different nitrogenous bases in DNA b. the number _____ of different chemical classes of amino acids c. the number _____ of chains of nucleotides in a DNA molecule d. the number _____ of different nitrogenous bases in RNA e. the number _____ of different amino acids found in pr ...
Membrane Proteins Integral membrane proteins often contain
... Integral membrane proteins often contain helical segments of appropriate length to span the lipid bilayer. In a protein that has a single segment that spans the membrane, the helix usually only contains hydrophobic residues and is called a single-span membrane protein. In transmembrane proteins with ...
... Integral membrane proteins often contain helical segments of appropriate length to span the lipid bilayer. In a protein that has a single segment that spans the membrane, the helix usually only contains hydrophobic residues and is called a single-span membrane protein. In transmembrane proteins with ...
Complex Protein Structure
... spiraling or pleating of a primary chain caused by hydrogen bonding. Examples include: a) Alpha helix (alpha keratin) soft hair or hard nails b) Beta pleated sheets (flexible but resist ...
... spiraling or pleating of a primary chain caused by hydrogen bonding. Examples include: a) Alpha helix (alpha keratin) soft hair or hard nails b) Beta pleated sheets (flexible but resist ...
PROTEIN SECONDARY STRUCTURE
... if a polypeptide chain is twisted by the same amount about each of its Ca atoms, it assumes a helical conformation helix characterization n=number of peptide units per helical turn pitch=distance helix rises along axis/turn ...
... if a polypeptide chain is twisted by the same amount about each of its Ca atoms, it assumes a helical conformation helix characterization n=number of peptide units per helical turn pitch=distance helix rises along axis/turn ...
Protein Chemistry Ben and Cody
... They are the building blocks of the biological process Proteins are created by amino acids bonded together ...
... They are the building blocks of the biological process Proteins are created by amino acids bonded together ...
Proteins have a higher order of folding known as tertiary structure
... You should know that hydrogen bonds between the polypeptide backbone are responsible for maintaining a protein’s secondary structure. However, many amino acids have side chains which contain a hydrogen atom attached to an oxygen or nitrogen, and can therefore form hydrogen bonds. For example, serine ...
... You should know that hydrogen bonds between the polypeptide backbone are responsible for maintaining a protein’s secondary structure. However, many amino acids have side chains which contain a hydrogen atom attached to an oxygen or nitrogen, and can therefore form hydrogen bonds. For example, serine ...
hw1009-aminoacids-proteins
... In this video, we see molecules hooking together to form macromolecules. The molecule is an amino acid or peptide, joining together to form a poplypeptide or protein. Please answer these questions: ...
... In this video, we see molecules hooking together to form macromolecules. The molecule is an amino acid or peptide, joining together to form a poplypeptide or protein. Please answer these questions: ...
Biochemistry Homework
... Electrophoresis can be used to identify the amino acids present in a given protein. The protein must first be hydrolyzed. ...
... Electrophoresis can be used to identify the amino acids present in a given protein. The protein must first be hydrolyzed. ...
Discussion Problem Set 3 C483 Spring 2014
... information obtained by these two methods? 5. Describe three chromatographic techniques used to purify proteins. 6. How is the total amino acid composition of a protein determined? Which amino acids cannot be determined in this experiment? Which amino acid reacts under these conditions, but can stil ...
... information obtained by these two methods? 5. Describe three chromatographic techniques used to purify proteins. 6. How is the total amino acid composition of a protein determined? Which amino acids cannot be determined in this experiment? Which amino acid reacts under these conditions, but can stil ...
The linear sequence of amino acids (primary structure) is able to coil
... The linear sequence of amino acids (primary structure) is able to coil and fold upon itself, resulting in 3D formations such as α-helices and β-sheets. These are held together by hydrogen bonding between amino acids. The term for these 3D formations is the secondary structure of the protein. ...
... The linear sequence of amino acids (primary structure) is able to coil and fold upon itself, resulting in 3D formations such as α-helices and β-sheets. These are held together by hydrogen bonding between amino acids. The term for these 3D formations is the secondary structure of the protein. ...
Chemistry 160 Protein Structure Homework
... 2. What are prosthetic groups? 3. What are glycoproteins and lipoproteins? 4. Describe the 4 levels of protein structure. 5. Describe 3 types of interactions that stabilize protein structure. 6. What drives protein folding? 7. Give two ways amino acid sequences are determined. 8. A small protein was ...
... 2. What are prosthetic groups? 3. What are glycoproteins and lipoproteins? 4. Describe the 4 levels of protein structure. 5. Describe 3 types of interactions that stabilize protein structure. 6. What drives protein folding? 7. Give two ways amino acid sequences are determined. 8. A small protein was ...
Secondary structure of proteins - Home
... • A polypeptide chain in the β-sheets is almost fully extended rather than being tightly coiled as in the α-helix • The axial distance between adjacent a.a in β-sheets is 3.5A while it is 1.5A in α-helix . • β-sheet is stabilized by hydrogen bonds between NH and CO groups in different polypeptide ch ...
... • A polypeptide chain in the β-sheets is almost fully extended rather than being tightly coiled as in the α-helix • The axial distance between adjacent a.a in β-sheets is 3.5A while it is 1.5A in α-helix . • β-sheet is stabilized by hydrogen bonds between NH and CO groups in different polypeptide ch ...