7. The Second Law and the Concept of Entropy
... No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature. Spontaneously, heat cannot flow from cold regions to hot regions without external work being performed on the system, which is evident from ordinary experience of refrig ...
... No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature. Spontaneously, heat cannot flow from cold regions to hot regions without external work being performed on the system, which is evident from ordinary experience of refrig ...
Chapter 15 Lesson 2
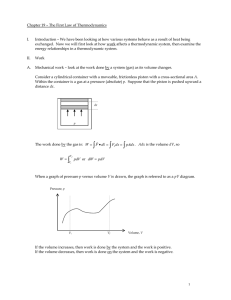
... A reversible process is one in which both the system and the environment can be returned to exactly the states they were in before the process occurred. ...
... A reversible process is one in which both the system and the environment can be returned to exactly the states they were in before the process occurred. ...
Combustion Chemistry
... • The Kelvin-Planck statement: It is impossible to construct a device that will operate in a cycle and produce no effect other than the raising of a weight and the exchange of heat with a single reservoir. ...
... • The Kelvin-Planck statement: It is impossible to construct a device that will operate in a cycle and produce no effect other than the raising of a weight and the exchange of heat with a single reservoir. ...
The Laws of Thermodynamics
... energy at the temperature of the environment, even when it isn't used to perform useful work, e.g., as in flashlight batteries. The second law can also be stated as: -"A process whose only effect is a local lowering of entropy cannot exist." This means that a local lowering of entropy can occur, but ...
... energy at the temperature of the environment, even when it isn't used to perform useful work, e.g., as in flashlight batteries. The second law can also be stated as: -"A process whose only effect is a local lowering of entropy cannot exist." This means that a local lowering of entropy can occur, but ...
The “Second Law” of Probability: Entropy Growth in the Central Limit
... It costs k questions to identify a state from among 2k equally likely states. It costs log2 n questions to identify a state from among n equally likely states: to identify a state with probability 1/n. ...
... It costs k questions to identify a state from among 2k equally likely states. It costs log2 n questions to identify a state from among n equally likely states: to identify a state with probability 1/n. ...
HEAT- Chapter 9
... In general, the volume of a liquid will decrease as temperature decreases; the exception is water Solids tend to have the smallest coefficient of volume Coefficient of Volume Expansion- a number assigned to different material to show the thermal expansion characteristic of the material Gases ...
... In general, the volume of a liquid will decrease as temperature decreases; the exception is water Solids tend to have the smallest coefficient of volume Coefficient of Volume Expansion- a number assigned to different material to show the thermal expansion characteristic of the material Gases ...
Thermodynamics of ideal gases
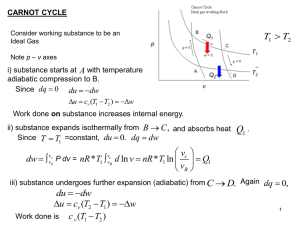
... The First Law states that the energy is unchanged under any process in an isolated system. This implies that the energy of an open system can only change by exchange of heat or work with the environment. We actually used this law implicitly in deriving the heat capacities and the entropy. The Second ...
... The First Law states that the energy is unchanged under any process in an isolated system. This implies that the energy of an open system can only change by exchange of heat or work with the environment. We actually used this law implicitly in deriving the heat capacities and the entropy. The Second ...
Thermodynamic functions - Phase Transformations Group
... The Helmholtz free energy F is the corresponding term at constant volume, when H is replaced by U in equation 9. A process can occur spontaneously if it leads to a reduction in the free energy. Quantities such as H, G and S are path independent and therefore are called functions of state. More About ...
... The Helmholtz free energy F is the corresponding term at constant volume, when H is replaced by U in equation 9. A process can occur spontaneously if it leads to a reduction in the free energy. Quantities such as H, G and S are path independent and therefore are called functions of state. More About ...
Lecture 12
... calorimeter measurements give )rUo = -2808 kJ mol-1 and )rSo = +182.4 J K mol-1 at 25oC. How much of the energy is extracted as (a) heat at constant pressure and (b) work? (a) )ng = 0, so )rUo = )rHo = -2808 kJ mol-1. So at constant pressure, energy available as heat is q = -2808 kJ mol-1 (b) At T = ...
... calorimeter measurements give )rUo = -2808 kJ mol-1 and )rSo = +182.4 J K mol-1 at 25oC. How much of the energy is extracted as (a) heat at constant pressure and (b) work? (a) )ng = 0, so )rUo = )rHo = -2808 kJ mol-1. So at constant pressure, energy available as heat is q = -2808 kJ mol-1 (b) At T = ...
The Second Law and the Concept of Entropy
... concepts of state and process. A process occurs when the thermodynamic system undergoes a change in state or an energy transfer at steady state. A process can be perfect or reversible or imperfect and irreversible. A reversible process occurs when the process can be completely reversed and leave no ...
... concepts of state and process. A process occurs when the thermodynamic system undergoes a change in state or an energy transfer at steady state. A process can be perfect or reversible or imperfect and irreversible. A reversible process occurs when the process can be completely reversed and leave no ...
МІНІСТЕРСТВО ОХОРОНИ ЗДОРОВ`Я УКРАЇНИ ХАРКІВСЬКИЙ
... In a salt-water system energetic minimum corresponds to the crystalline state of salt. However, a more probable state is achieved by random distribution of salt in liquid water. According to the same principle, in chemical reactions atoms tend to join in molecules whose formation results in the rele ...
... In a salt-water system energetic minimum corresponds to the crystalline state of salt. However, a more probable state is achieved by random distribution of salt in liquid water. According to the same principle, in chemical reactions atoms tend to join in molecules whose formation results in the rele ...