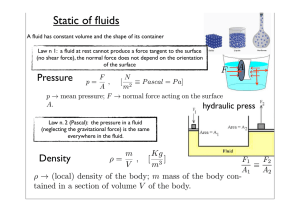
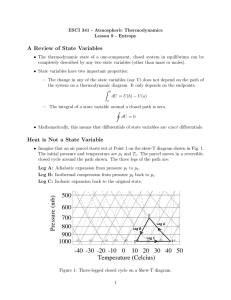
1 Thermodynamics All biochemical and cellular processes obey the
... Equilibrium is the state that all processes tend to approach. Life is (among other things), an ultimately futile attempt to avoid equilibrium. For biological systems (where pressure and volume changes are negligible), ∆G represents the maximum possible work resulting from a reaction. In other words, ...
... Equilibrium is the state that all processes tend to approach. Life is (among other things), an ultimately futile attempt to avoid equilibrium. For biological systems (where pressure and volume changes are negligible), ∆G represents the maximum possible work resulting from a reaction. In other words, ...
NOTES on THERMODYNAMICS - University of Utah Physics
... matical structure is then constructed on the basis of these observations, which leads to a variety of useful concepts, and to testable relationships among various quantities. The laws of thermodynamics can only be justified by a more fundamental (microscopic) theory of nature. For example, statistica ...
... matical structure is then constructed on the basis of these observations, which leads to a variety of useful concepts, and to testable relationships among various quantities. The laws of thermodynamics can only be justified by a more fundamental (microscopic) theory of nature. For example, statistica ...
I Thermodynamics - Stanford University
... If two systems are placed in contact there will be heat flow between them until thermal equilibrium is reached (they are at the same temperature). It also expresses how much work can be done by or on a system given an amount of heat flow. -The change in internal energy of a closed system is equal to ...
... If two systems are placed in contact there will be heat flow between them until thermal equilibrium is reached (they are at the same temperature). It also expresses how much work can be done by or on a system given an amount of heat flow. -The change in internal energy of a closed system is equal to ...
Chapter 2. Entropy and Temperature
... Consider two 10 g pieces of copper, one initally at 350 K and the other at 290 K. Assuming that the heat capacity of copper is 0.389J g−1 K−1 , what is the entropy increase if the 2 blocks are brought into thermal contact and allowed to come into equilibrium with one another? First, the total heat c ...
... Consider two 10 g pieces of copper, one initally at 350 K and the other at 290 K. Assuming that the heat capacity of copper is 0.389J g−1 K−1 , what is the entropy increase if the 2 blocks are brought into thermal contact and allowed to come into equilibrium with one another? First, the total heat c ...
Chemistry 331 In Class Exercise Review for Final #1) (a) What are
... #27) Using pictures and words describe the isothermal compression of an ideal gas in which a minimum amount of work has been done on the system. ...
... #27) Using pictures and words describe the isothermal compression of an ideal gas in which a minimum amount of work has been done on the system. ...
Entropy in chemical thermodynamics
... The statistical definition of entropy matches up with the thermodynamic formula for calculating entropy, because adding heat to a system, which increases its classical thermodynamic entropy, also increases the system's thermal fluctuations, so giving an increased lack of information about the exact ...
... The statistical definition of entropy matches up with the thermodynamic formula for calculating entropy, because adding heat to a system, which increases its classical thermodynamic entropy, also increases the system's thermal fluctuations, so giving an increased lack of information about the exact ...
Lecture 5 Entropy
... A microstate is the detailed state of a physical system. Example: In an ideal gas, a microstate consists of the position and velocity of every molecule in the system. So the number of microstates is just what Feynman said: the number of different ways the inside of the system can be changed without ...
... A microstate is the detailed state of a physical system. Example: In an ideal gas, a microstate consists of the position and velocity of every molecule in the system. So the number of microstates is just what Feynman said: the number of different ways the inside of the system can be changed without ...
Introduction to Physical Chemistry – Lecture 7
... Regarding the Third Law, this simply allows us to pick a reference point where an absolute value of entropy can be assigned. The law dS = (δQ/T )rev only allows us to compute changes in entropy. The Third Law says that at 0 K a perfect crystal has no entropy. This makes sense, since an object at 0 K ...
... Regarding the Third Law, this simply allows us to pick a reference point where an absolute value of entropy can be assigned. The law dS = (δQ/T )rev only allows us to compute changes in entropy. The Third Law says that at 0 K a perfect crystal has no entropy. This makes sense, since an object at 0 K ...
LAW: The first law of thermodynamics states that the total energy in
... ◦ Describe the physical meaning of each of the terms within both the integral and differential form of the first law ◦ Identify when the open and closed forms of the first law are applicable ◦ Determine when each term in the first law is zero or negligible • Establish whether the ideal gas law if ap ...
... ◦ Describe the physical meaning of each of the terms within both the integral and differential form of the first law ◦ Identify when the open and closed forms of the first law are applicable ◦ Determine when each term in the first law is zero or negligible • Establish whether the ideal gas law if ap ...
LAW: The first law of thermodynamics states that the total energy in
... ◦ Describe the physical meaning of each of the terms within both the integral and differential form of the first law ◦ Identify when the open and closed forms of the first law are applicable ◦ Determine when each term in the first law is zero or negligible • Establish whether the ideal gas law if ap ...
... ◦ Describe the physical meaning of each of the terms within both the integral and differential form of the first law ◦ Identify when the open and closed forms of the first law are applicable ◦ Determine when each term in the first law is zero or negligible • Establish whether the ideal gas law if ap ...
Chemistry Entropy Notes 1. What is entropy? How many ways can
... Free Energy of a system. It incorporates the system’s entropy change and the system’s heat change (heat from the system increases the entropy of the surroundings). So, ∆G° is a useful predictor of whether or not reactions will occur because it incorporates changes in both the system and the surround ...
... Free Energy of a system. It incorporates the system’s entropy change and the system’s heat change (heat from the system increases the entropy of the surroundings). So, ∆G° is a useful predictor of whether or not reactions will occur because it incorporates changes in both the system and the surround ...
Tutorial II (thermodynamics)
... or false giving your thermodynamic reasoning. If the statement is false you may either state which law or laws of thermodynamics it violates or provide a physical counter example or any other plausible physical reason. Finally, correct the false statement with a clarifying phrase that makes the stat ...
... or false giving your thermodynamic reasoning. If the statement is false you may either state which law or laws of thermodynamics it violates or provide a physical counter example or any other plausible physical reason. Finally, correct the false statement with a clarifying phrase that makes the stat ...
Atomic Structure
... broken down and oxidized (has oxygen added), and there is energy released. c. We gain energy from the food, air and water that we take in. This energy is converted to heat and into work, and stored as potential energy, for example in fats. The total energy is always conserved, and the change in inte ...
... broken down and oxidized (has oxygen added), and there is energy released. c. We gain energy from the food, air and water that we take in. This energy is converted to heat and into work, and stored as potential energy, for example in fats. The total energy is always conserved, and the change in inte ...