Unit II - Chemical Thermodynamics
... spontaneously without the intervention of an external energy. Kelvin statement: It is impossible to take heat from a hot body and convert it completely into work by a cyclic process without transferring a part of heat to cold body. II law in terms of entropy: A spontaneous process is always accompan ...
... spontaneously without the intervention of an external energy. Kelvin statement: It is impossible to take heat from a hot body and convert it completely into work by a cyclic process without transferring a part of heat to cold body. II law in terms of entropy: A spontaneous process is always accompan ...
The state of a simple compressible system is completely specified by
... 4. Be able to produce and interpret processes using graphical representation to link states, properties, and paths. 5. Measureable thermodynamic properties – pressure and temperature. The state of a system is like its CONDITION, and is defined by a set of properties. Why do we need to know the state ...
... 4. Be able to produce and interpret processes using graphical representation to link states, properties, and paths. 5. Measureable thermodynamic properties – pressure and temperature. The state of a system is like its CONDITION, and is defined by a set of properties. Why do we need to know the state ...
Fundamentals of Equilibrium Thermodynamics
... classical context). Equilibrium states do not change with time (in contrast to microstates, which are perennially hustling about), and in practice such states are often metastable, meaning they change on a very long timescale, and can ‘therefore’ be viewed to be equilibrium states. It does not seem ...
... classical context). Equilibrium states do not change with time (in contrast to microstates, which are perennially hustling about), and in practice such states are often metastable, meaning they change on a very long timescale, and can ‘therefore’ be viewed to be equilibrium states. It does not seem ...
WRL0638.tmp - Symposium on Chemical Physics
... classical context). Equilibrium states do not change with time (in contrast to microstates, which are perennially hustling about), and in practice such states are often metastable, meaning they change on a very long timescale, and can ‘therefore’ be viewed to be equilibrium states. It does not seem ...
... classical context). Equilibrium states do not change with time (in contrast to microstates, which are perennially hustling about), and in practice such states are often metastable, meaning they change on a very long timescale, and can ‘therefore’ be viewed to be equilibrium states. It does not seem ...
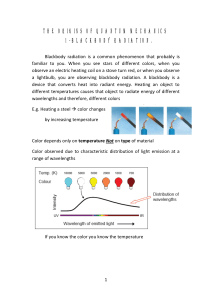
the origins of quantum mechanics 1
... is the energy density in units of energy per volume (e.g., J m-3), is the frequency of emitted radiation, T is the temperature of the blackbody, k is Boltzmann's constant, and c is the speed of light. This is known as the Rayleigh-Jeans law. A clear implication of this law is that as the frequency ...
... is the energy density in units of energy per volume (e.g., J m-3), is the frequency of emitted radiation, T is the temperature of the blackbody, k is Boltzmann's constant, and c is the speed of light. This is known as the Rayleigh-Jeans law. A clear implication of this law is that as the frequency ...
introduction
... substances at absolute zero (0 Kelvin), the temperature at which all atomic motion ceases, have entropy of zero. When these laws are applied to chemical reactions, they relate the equilibrium condition (whether the reaction is proceeding in the forward or reverse direction) and the temperature of th ...
... substances at absolute zero (0 Kelvin), the temperature at which all atomic motion ceases, have entropy of zero. When these laws are applied to chemical reactions, they relate the equilibrium condition (whether the reaction is proceeding in the forward or reverse direction) and the temperature of th ...
Chapter 3
... A is the energy that must be provided as work if we create the system out of nothing. The heat extracted from the surroundings is T∆S = T(Sf − Si) = TSf where Sf is the system final entropy and Si the system zero initial entropy. If we annihilate a system with initial entropy Si, A is the amount of ...
... A is the energy that must be provided as work if we create the system out of nothing. The heat extracted from the surroundings is T∆S = T(Sf − Si) = TSf where Sf is the system final entropy and Si the system zero initial entropy. If we annihilate a system with initial entropy Si, A is the amount of ...