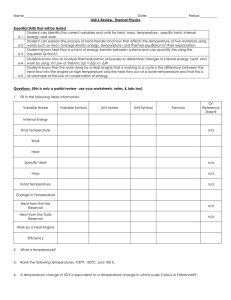
1st law of Thermodynamics Worksheet
... 8. A system releases 125 kJ of heat while 104 kJ of work is done on the system. Calculate the change in internal energy (in kJ). ∆U = 21 KJ 9. A system undergoes a process consisting of the following two steps: Step 1: The system absorbs 73 J of heat while 35 J of work is done on it. Step 2: The sys ...
... 8. A system releases 125 kJ of heat while 104 kJ of work is done on the system. Calculate the change in internal energy (in kJ). ∆U = 21 KJ 9. A system undergoes a process consisting of the following two steps: Step 1: The system absorbs 73 J of heat while 35 J of work is done on it. Step 2: The sys ...
Thermodynamics - Faculty
... 5. There are two things to always keep in mind when doing calculations with heat engines: a) W is positive (and Wenv is negative) when the system contracts (i.e., ∆V < 0), and negative (and Wenv positive) when it expands (i.e., ∆V > 0). b) ...
... 5. There are two things to always keep in mind when doing calculations with heat engines: a) W is positive (and Wenv is negative) when the system contracts (i.e., ∆V < 0), and negative (and Wenv positive) when it expands (i.e., ∆V > 0). b) ...
INTRODUCTION - WordPress.com
... An actual process occurs only when the equilibrium state does not exist. ...
... An actual process occurs only when the equilibrium state does not exist. ...
Survival of the Likeliest?
... branches of physics have been able to formulate the second law of thermodynamics independently. This has meant that other fields, such as computing and ecology, can use the concept of entropy, and so entropy takes rather different forms in different systems. In thermodynamics, entropy is uselessness. ...
... branches of physics have been able to formulate the second law of thermodynamics independently. This has meant that other fields, such as computing and ecology, can use the concept of entropy, and so entropy takes rather different forms in different systems. In thermodynamics, entropy is uselessness. ...
Quiz_MATH.rtf
... C) Q = (5/2)nRT, Eint = (5/2)nRT, W = 0 D) Q = (3/2)nRT, Eint = 0, W = –nRT E) Q = (5/2)nRT, Eint = (3/2)nRT, W = –nRT 17. (B) The temperature of n moles of an ideal monatomic gas is increased by T at constant volume. The energy Q absorbed as heat, change Eint in internal energy, and wo ...
... C) Q = (5/2)nRT, Eint = (5/2)nRT, W = 0 D) Q = (3/2)nRT, Eint = 0, W = –nRT E) Q = (5/2)nRT, Eint = (3/2)nRT, W = –nRT 17. (B) The temperature of n moles of an ideal monatomic gas is increased by T at constant volume. The energy Q absorbed as heat, change Eint in internal energy, and wo ...
The First, Second, and Third Law of Thermodynamics (ThLaws05.tex)
... The laws of thermodynamics apply to well-de…ned systems. First we will discuss a quite general form of the …rst and second law. I.e. we consider a system which is inhomogeneous, we allow mass transfer across the boundaries (open system), and we allow the boundaries to move. Fig.1 is a general repres ...
... The laws of thermodynamics apply to well-de…ned systems. First we will discuss a quite general form of the …rst and second law. I.e. we consider a system which is inhomogeneous, we allow mass transfer across the boundaries (open system), and we allow the boundaries to move. Fig.1 is a general repres ...
2nd law of thermodynamics
... All real spontaneous processes - irreversible. Invertible only ideal process. In real systems, only the irreversible part of the energy is converted into useful work. To characterize this energy related Clausius introduced a new state function, called entropy «S». Quantitative measure of entropy cal ...
... All real spontaneous processes - irreversible. Invertible only ideal process. In real systems, only the irreversible part of the energy is converted into useful work. To characterize this energy related Clausius introduced a new state function, called entropy «S». Quantitative measure of entropy cal ...
GRE-thermo
... 21. In the cycle shown on the right, KL and NM represent isotherms, while KN and LM represent reversible adiabats. A system is carried through the Carnot cycle KLMN, taking in heat Q2 from the hot reservoir T2 and releasing heat Q1 to the cold reservoir T2. All of the following statements are true E ...
... 21. In the cycle shown on the right, KL and NM represent isotherms, while KN and LM represent reversible adiabats. A system is carried through the Carnot cycle KLMN, taking in heat Q2 from the hot reservoir T2 and releasing heat Q1 to the cold reservoir T2. All of the following statements are true E ...
Thermodynamic Systems and State Functions
... transformations of heat into mechanical work and the opposite ones that transform mechanical work into heat.” Obviously, when chemical systems are of interest, other forms of work can be taken into account besides the purely mechanical one, such as electrical or chemical ones. On a more general basi ...
... transformations of heat into mechanical work and the opposite ones that transform mechanical work into heat.” Obviously, when chemical systems are of interest, other forms of work can be taken into account besides the purely mechanical one, such as electrical or chemical ones. On a more general basi ...
Thermodynamic system
... • Temperature quantifies the kinetic energy of microscopic thermal motion (recall the equipartition theorem) • Heat is energy transfer between two systems (apart from work) • Temperature is a state variable, heat is not (depends on process path) • Isothermal vs. adiabatic process: – if system is the ...
... • Temperature quantifies the kinetic energy of microscopic thermal motion (recall the equipartition theorem) • Heat is energy transfer between two systems (apart from work) • Temperature is a state variable, heat is not (depends on process path) • Isothermal vs. adiabatic process: – if system is the ...