Exergy - SABİS
... argon gas at 400 K and 350 kPa. Heat is now transferred to the argon from a furnace at 1200 K, and the argon expands isothermally until its volume is doubled. No heat transfer takes place between the argon and the surrounding atmospheric air, which is at T0 300 K and P0 100 kPa. Determine (a) the us ...
... argon gas at 400 K and 350 kPa. Heat is now transferred to the argon from a furnace at 1200 K, and the argon expands isothermally until its volume is doubled. No heat transfer takes place between the argon and the surrounding atmospheric air, which is at T0 300 K and P0 100 kPa. Determine (a) the us ...
Lecture 4 (Dec.9)
... into huge caverns as a form of energy storage. The volume of a cavern is 5.6 x 105 m3 and the pressure of the air in it is 7.7 x 106 Pa. Assume that air is a diatomic ideal gas whose internal energy U is given by If one home uses 30.0 kWh of energy per day, how many homes could this internal energy ...
... into huge caverns as a form of energy storage. The volume of a cavern is 5.6 x 105 m3 and the pressure of the air in it is 7.7 x 106 Pa. Assume that air is a diatomic ideal gas whose internal energy U is given by If one home uses 30.0 kWh of energy per day, how many homes could this internal energy ...
Chapter 4 - The First Law of Thermodynamics and Energy Transport
... a while, so he asked him to add up all the numbers from 1 to 100. That is, find X = 1 + 2 + 3 + … + 100. To the teacher’s surprise, Gauss returned a few minutes later and said that the sum was 5050. Apparently Gauss noticed that the sum is the same regardless of whether the terms are added forward ( ...
... a while, so he asked him to add up all the numbers from 1 to 100. That is, find X = 1 + 2 + 3 + … + 100. To the teacher’s surprise, Gauss returned a few minutes later and said that the sum was 5050. Apparently Gauss noticed that the sum is the same regardless of whether the terms are added forward ( ...
Heat energy
... A wheel and axle is a simple machine that is made up of a smaller cylinder (the axle) joined to a larger cylinder (the wheel). To work together, the axle must be connected to the wheel in such a way that it allows the wheel to rotate evenly about its center. ...
... A wheel and axle is a simple machine that is made up of a smaller cylinder (the axle) joined to a larger cylinder (the wheel). To work together, the axle must be connected to the wheel in such a way that it allows the wheel to rotate evenly about its center. ...
Energy
... on the amount of material present. • Examples are volume, entropy, enthalpy, mass, internal energy etc. • Intensive Property: This does not depend on the amount of material present. • It is an intrinsic property: e.g. temperature, pressure, ...
... on the amount of material present. • Examples are volume, entropy, enthalpy, mass, internal energy etc. • Intensive Property: This does not depend on the amount of material present. • It is an intrinsic property: e.g. temperature, pressure, ...
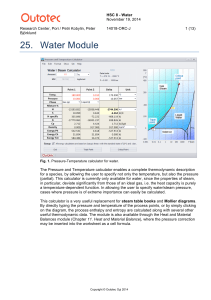
Click to open the TEOS-10 teaching aid slides(powerpoint)
... • Practical Salinity is calculated from the conductivity of seawater, and is not the mass fraction of salt in seawater. • The thermodynamic properties of seawater are more closely dependent on the mass fraction (Absolute Salinity SA) of dissolved material, not the conductivity or Practical Salinity ...
... • Practical Salinity is calculated from the conductivity of seawater, and is not the mass fraction of salt in seawater. • The thermodynamic properties of seawater are more closely dependent on the mass fraction (Absolute Salinity SA) of dissolved material, not the conductivity or Practical Salinity ...
Describe two energy transfers that happened in the
... A 2-point response demonstrates the student understands the Standard 6-8 PS3A: Energy exists in many forms: heat, light, chemical, electrical, motion of objects, and sound. Energy can be transformed from one form to another and transferred from one place to another. The response describes two energy ...
... A 2-point response demonstrates the student understands the Standard 6-8 PS3A: Energy exists in many forms: heat, light, chemical, electrical, motion of objects, and sound. Energy can be transformed from one form to another and transferred from one place to another. The response describes two energy ...
calcijlation of elastic properties from thermodynamic equation of
... in the spirit of an earlier review by Anderson et a1 (1968). Three broad areas bear on this topic: thermodynamic analysis, continuum mechanics, and solid state physics. Experiments provide the raw data and some important rules of thumb that make it possible to compute elastic velocities inside the E ...
... in the spirit of an earlier review by Anderson et a1 (1968). Three broad areas bear on this topic: thermodynamic analysis, continuum mechanics, and solid state physics. Experiments provide the raw data and some important rules of thumb that make it possible to compute elastic velocities inside the E ...
The Second Law of Thermodynamics
... an analysis of the e‰ciency of heat engines, which laid the foundation of the second law of thermodynamics. The Carnot heat engine is an idealized model for the operation of any heat engine. For our purpose, it can be represented by an ideal gas in a cylinder fitted with a movable, frictionless pisto ...
... an analysis of the e‰ciency of heat engines, which laid the foundation of the second law of thermodynamics. The Carnot heat engine is an idealized model for the operation of any heat engine. For our purpose, it can be represented by an ideal gas in a cylinder fitted with a movable, frictionless pisto ...
2 - MechFamily
... 2–3 ENERGY TRANSFER BY HEAT • Energy can cross the boundary of a closed system in two distinct forms: heat and work • Heat H t is i defined d fi d as the th form f off energy that is transferred between two systems ( a system (or t andd its it surroundings) di ) by b virtue of a temperature differe ...
... 2–3 ENERGY TRANSFER BY HEAT • Energy can cross the boundary of a closed system in two distinct forms: heat and work • Heat H t is i defined d fi d as the th form f off energy that is transferred between two systems ( a system (or t andd its it surroundings) di ) by b virtue of a temperature differe ...
The Kinetic Theory of Gases
... such large-scale properties can be described on a microscopic scale, where matter is treated as a collection of molecules. Newton’s laws of motion applied in a statistical manner to a collection of particles provide a reasonable description of thermodynamic processes. To keep the mathematics relativ ...
... such large-scale properties can be described on a microscopic scale, where matter is treated as a collection of molecules. Newton’s laws of motion applied in a statistical manner to a collection of particles provide a reasonable description of thermodynamic processes. To keep the mathematics relativ ...
Pdf - Text of NPTEL IIT Video Lectures
... 1 minus T2 that is the temperature of the thermal reservoirs where heat is rejected divided by T1. It is a thermal temperature in thermodynamic absolute scale of the reservoir from where heat is being taken that means etaengine is 0.5 into 1 minus 333 divided by 944 and if we equate this value, this ...
... 1 minus T2 that is the temperature of the thermal reservoirs where heat is rejected divided by T1. It is a thermal temperature in thermodynamic absolute scale of the reservoir from where heat is being taken that means etaengine is 0.5 into 1 minus 333 divided by 944 and if we equate this value, this ...
About the Guide - American Chemical Society
... never occur in nature. For example, if we bring a hot object into contact with a cold object, we observe that the hot object cools down and the cold object heats up until an equilibrium is reached. The transfer of heat goes from the hot object to the cold object. We can imagine a system, however, in ...
... never occur in nature. For example, if we bring a hot object into contact with a cold object, we observe that the hot object cools down and the cold object heats up until an equilibrium is reached. The transfer of heat goes from the hot object to the cold object. We can imagine a system, however, in ...
2.01 The Electromagnetic Spectrum
... Energy is defined as the ability to do work. Sometimes it’s easier to describe what energy does that what energy is. That is because, unlike matter, energy is not something you can see or touch. Energy is a property of matter, and all matter has it. Whenever a light bulb is lit, a turkey is roasted, ...
... Energy is defined as the ability to do work. Sometimes it’s easier to describe what energy does that what energy is. That is because, unlike matter, energy is not something you can see or touch. Energy is a property of matter, and all matter has it. Whenever a light bulb is lit, a turkey is roasted, ...