Chp-1. Pure substances
... Superheating is done by utilizing the heat of waste furnace gases which otherwise passes uselessly to the atmosphere. Steam generation process, as derived above, has been under standard atmospheric pressure. At different pressures below critical pressure the nature of the various processes will be i ...
... Superheating is done by utilizing the heat of waste furnace gases which otherwise passes uselessly to the atmosphere. Steam generation process, as derived above, has been under standard atmospheric pressure. At different pressures below critical pressure the nature of the various processes will be i ...
Energy - Clover Park School District
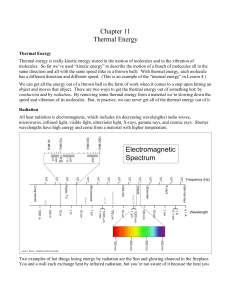
... Motion energy is also called kinetic energy; defined in a given reference frame, it is proportional to the mass of the moving object and grows with the square of its speed. Matter at any temperature above absolute zero contains thermal energy. Thermal energy is the random motion of particles (whethe ...
... Motion energy is also called kinetic energy; defined in a given reference frame, it is proportional to the mass of the moving object and grows with the square of its speed. Matter at any temperature above absolute zero contains thermal energy. Thermal energy is the random motion of particles (whethe ...
Topic # 3
... Warm-core lows… Warm at its center Example: a hurricane (Fig. (3.3)) Intensity decreases with height ...
... Warm-core lows… Warm at its center Example: a hurricane (Fig. (3.3)) Intensity decreases with height ...
ppt - Charles W. Davidson College of Engineering
... • We build machines to ‘manage’ energy conversion. • While energy cannot be destroyed, once it is transformed into a certain form (heat, often the case), it is basically difficult to use (some say “lost” but we cannot actually lose energy). based on notes of P. Hsu 2007 ...
... • We build machines to ‘manage’ energy conversion. • While energy cannot be destroyed, once it is transformed into a certain form (heat, often the case), it is basically difficult to use (some say “lost” but we cannot actually lose energy). based on notes of P. Hsu 2007 ...
What is the conservation of energy?
... Fill a car up with gasoline and you have a closed system. All the energy you have at your disposal is locked inside the gas in your tank in chemical form. When the gas flows into your engine, it burns with oxygen in the air. The chemical energy in the gas is converted first into heat energy: the bur ...
... Fill a car up with gasoline and you have a closed system. All the energy you have at your disposal is locked inside the gas in your tank in chemical form. When the gas flows into your engine, it burns with oxygen in the air. The chemical energy in the gas is converted first into heat energy: the bur ...
Chapter 11 - Wolaver.org
... same direction and all with the same speed (like in a thrown ball). With thermal energy, each molecule has a different direction and different speed. (This is an example of the “internal energy” in Lesson 4.) We can get all the energy out of a thrown ball in the form of work when it comes to a stop ...
... same direction and all with the same speed (like in a thrown ball). With thermal energy, each molecule has a different direction and different speed. (This is an example of the “internal energy” in Lesson 4.) We can get all the energy out of a thrown ball in the form of work when it comes to a stop ...
Kinetic Energy - SJSU Engineering
... • We build machines to ‘manage’ energy conversion. • While energy cannot be destroyed, once it is transformed into a certain form (heat, often the case), it is basically difficult to use (some say “lost” but we cannot actually lose energy). based on notes of P. Hsu 2007 ...
... • We build machines to ‘manage’ energy conversion. • While energy cannot be destroyed, once it is transformed into a certain form (heat, often the case), it is basically difficult to use (some say “lost” but we cannot actually lose energy). based on notes of P. Hsu 2007 ...
temperature 2015 10 13
... There exists a coldest place of hotness, but not a hottest place of hotness. Name the coldest place of hotness zero. Name all other places of hotness by a single, positive, continuous variable. Such a scale of hotness is called an absolute scale. ...
... There exists a coldest place of hotness, but not a hottest place of hotness. Name the coldest place of hotness zero. Name all other places of hotness by a single, positive, continuous variable. Such a scale of hotness is called an absolute scale. ...
Etymology Physical characteristics
... determining physical properties of a gas such as viscosity and flow rate (see physical characteristics section). Ignoring these forces in certain conditions (see Kinetic-molecular theory) allows a real gas to be treated like an ideal gas. This assumption allows the use of ideal gas laws which greatl ...
... determining physical properties of a gas such as viscosity and flow rate (see physical characteristics section). Ignoring these forces in certain conditions (see Kinetic-molecular theory) allows a real gas to be treated like an ideal gas. This assumption allows the use of ideal gas laws which greatl ...
How much kinetic energy does the mass have
... Engaging with the concepts A battery contains 500 J of energy. Assume a motor converts battery energy to kinetic energy with perfect ...
... Engaging with the concepts A battery contains 500 J of energy. Assume a motor converts battery energy to kinetic energy with perfect ...
Calorimetry Measurement
... One of the forms in which energy can be present in a system is the random, internal kinetic energy of the particles (molecules or atoms) of a system, which can intuitively be called “thermal energy.” This is to be distinguished from the average, external movement of a system of particles as a whole, ...
... One of the forms in which energy can be present in a system is the random, internal kinetic energy of the particles (molecules or atoms) of a system, which can intuitively be called “thermal energy.” This is to be distinguished from the average, external movement of a system of particles as a whole, ...
Thermo fundamentals
... Therefore, it is required to have more accurate predictions for a substance, over a larger region and without limitations. Several equations are proposed by various scientists and researchers. 1. Van der Waal’s Equation of State : ...
... Therefore, it is required to have more accurate predictions for a substance, over a larger region and without limitations. Several equations are proposed by various scientists and researchers. 1. Van der Waal’s Equation of State : ...
Meandering Road From Dynamics To Thermodynamics And Vice
... displacement) on the system. Work, like heat, is a function of the path of a process and is an inexact differential. Systems never possess heat or work, but either or both cross the system boundary when a system undergoes a process. Both heat and work are transient phenomena as well as boundary phen ...
... displacement) on the system. Work, like heat, is a function of the path of a process and is an inexact differential. Systems never possess heat or work, but either or both cross the system boundary when a system undergoes a process. Both heat and work are transient phenomena as well as boundary phen ...
Energy - World of Teaching
... water molecules slowly leak through the walls of the glass. water molecules move into the air as gas molecules. water left in a glass starts to boil and becomes a gas. water slowly combines with oxygen to become carbon dioxide. ...
... water molecules slowly leak through the walls of the glass. water molecules move into the air as gas molecules. water left in a glass starts to boil and becomes a gas. water slowly combines with oxygen to become carbon dioxide. ...
Basic Concepts
... Quasistatic Process We assume that the outside pressure is always nearly equal to the inside pressure, a situation called quasistatic. In such a case, the motion of the movable wall separating the system from the surroundings is infinitely slow. Compression can easily be reversed by expansion; i.e. ...
... Quasistatic Process We assume that the outside pressure is always nearly equal to the inside pressure, a situation called quasistatic. In such a case, the motion of the movable wall separating the system from the surroundings is infinitely slow. Compression can easily be reversed by expansion; i.e. ...