heat and temperature
... thermometric liquid. To find out the relation, fix any two values of To and write down these values together with the corresponding heights. As T-H is a linear relation of the form T=a*H+B, replace T and H with the values that you have just written down and solve the system of two equations to find ...
... thermometric liquid. To find out the relation, fix any two values of To and write down these values together with the corresponding heights. As T-H is a linear relation of the form T=a*H+B, replace T and H with the values that you have just written down and solve the system of two equations to find ...
Review of Thermodynamics
... boundary. To be specific, suppose that the system is contained within rigid, impermeable, adiathermal walls. These boundary conditions specify the volume V , particle number N , and internal energy U . Now suppose that the volume of the system is changed by moving a piston that might comprise one of ...
... boundary. To be specific, suppose that the system is contained within rigid, impermeable, adiathermal walls. These boundary conditions specify the volume V , particle number N , and internal energy U . Now suppose that the volume of the system is changed by moving a piston that might comprise one of ...
Energy Transfer - RGS Physics
... contain free electrons which enable current to flow through them. These same free electrons can help transfer thermal energy quickly. Metals are very good conductors because they have many free electrons to carry the energy. Insulators do not have these free electrons and therefore they do not condu ...
... contain free electrons which enable current to flow through them. These same free electrons can help transfer thermal energy quickly. Metals are very good conductors because they have many free electrons to carry the energy. Insulators do not have these free electrons and therefore they do not condu ...
The Kinetic Theory of Gases
... This result indicates that the pressure of a gas is proportional to (1) the number of molecules per unit volume and (2) the average translational kinetic energy of the molecules, 12m 0v 2 . In analyzing this simplified model of an ideal gas, we obtain an important result that relates the macroscopic ...
... This result indicates that the pressure of a gas is proportional to (1) the number of molecules per unit volume and (2) the average translational kinetic energy of the molecules, 12m 0v 2 . In analyzing this simplified model of an ideal gas, we obtain an important result that relates the macroscopic ...
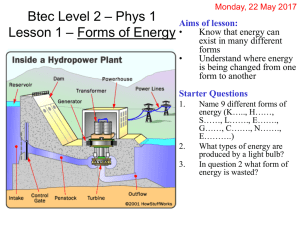
ENERGY MANAGEMENT
... black coal - when mining at 13 to 15106 t/year and with deposits of 2350106 t, it should last until 2150. However, in recent years mining is in decline and the figure may change under economic pressure and based on political decisions, brown coal - if mining at 60106 t/year and with deposits of 3 ...
... black coal - when mining at 13 to 15106 t/year and with deposits of 2350106 t, it should last until 2150. However, in recent years mining is in decline and the figure may change under economic pressure and based on political decisions, brown coal - if mining at 60106 t/year and with deposits of 3 ...
!!!Energy!!!
... c. Determine the heat capacity of a substance using mass, specific heat, and temperature. ...
... c. Determine the heat capacity of a substance using mass, specific heat, and temperature. ...
The Kinetic Theory of Gases - Department of Applied Physics
... only does the internal energy of the gas increase, but the gas also does work because of the change in volume. Therefore, the heat Q constant P must account for both the increase in internal energy and the transfer of energy out of the system by work, and so Q constant P is greater than Q constant V ...
... only does the internal energy of the gas increase, but the gas also does work because of the change in volume. Therefore, the heat Q constant P must account for both the increase in internal energy and the transfer of energy out of the system by work, and so Q constant P is greater than Q constant V ...
Thermodynamics Theory + Questions.0001
... (a) Both A and R are individually true and R is the correct explanation of A (b) Both A and R are individually true but R is NOT the correct explanation of A (c) A is true but R is false ...
... (a) Both A and R are individually true and R is the correct explanation of A (b) Both A and R are individually true but R is NOT the correct explanation of A (c) A is true but R is false ...
Atomic Structure
... The energy gained by the water is the Ematch = 4180 J.kg-1.K-1 m T. This is the energy supplied by the match. A bath tub contains around 300 l or 0.3 m3 which is around 300 kg of water. A nice hot bath is around 50oC, and tap water is typically at around 20oC, so you need a temperature change o ...
... The energy gained by the water is the Ematch = 4180 J.kg-1.K-1 m T. This is the energy supplied by the match. A bath tub contains around 300 l or 0.3 m3 which is around 300 kg of water. A nice hot bath is around 50oC, and tap water is typically at around 20oC, so you need a temperature change o ...
Chapter 14: Gas-Vapor Mixtures and Air
... If the mixture leaving the duct is saturated and if the process is adiabatic, the temperature of the mixture on leaving the device is known as the adiabatic saturation temperature. For this to be a steady-flow process, makeup water at the adiabatic saturation temperature is added at the same rate a ...
... If the mixture leaving the duct is saturated and if the process is adiabatic, the temperature of the mixture on leaving the device is known as the adiabatic saturation temperature. For this to be a steady-flow process, makeup water at the adiabatic saturation temperature is added at the same rate a ...
THE FIRST LAW OF THERMODYNAMICS 3·5 So far we have
... Now we are in a position to consider simultaneous heat and work interactions. As you may have already guessed, when a system involves both heat and work interactions during a process, their contributions are simply added. That is, if a system receives 12 kJ of heat while a paddle wheel does 6 kJ of ...
... Now we are in a position to consider simultaneous heat and work interactions. As you may have already guessed, when a system involves both heat and work interactions during a process, their contributions are simply added. That is, if a system receives 12 kJ of heat while a paddle wheel does 6 kJ of ...
Energy Unit Packet energy_unit_packet
... When Herman lifts this barbell, he is applying an upward force. The distance the barbell moved is measured from its resting place on the floor to the position over Herman’s head. In order for Herman to apply the lifting force, he must use energy. Kinds of Energy We will study two kinds of energy: po ...
... When Herman lifts this barbell, he is applying an upward force. The distance the barbell moved is measured from its resting place on the floor to the position over Herman’s head. In order for Herman to apply the lifting force, he must use energy. Kinds of Energy We will study two kinds of energy: po ...
20 Entropy and the Second Law of Thermodynamics
... it for granted. If these processes were to occur spontaneously (on their own) in the “wrong” direction, we would be astonished beyond belief. Yet none of these “wrongway” events would violate the law of conservation of energy. In the “cold hands-warm mug” example, energy would be conserved even for ...
... it for granted. If these processes were to occur spontaneously (on their own) in the “wrong” direction, we would be astonished beyond belief. Yet none of these “wrongway” events would violate the law of conservation of energy. In the “cold hands-warm mug” example, energy would be conserved even for ...
Outline Introduction State Functions Energy, Heat, and Work
... function depends only on thermodynamic state of the system (e.g. P, V, T for a simple system). q – energy added to the system as heat. Positive (+) when the system gains heat from outside (……………. process), negative (-) when heat flows out of the system (……………. process). w - work done by the system o ...
... function depends only on thermodynamic state of the system (e.g. P, V, T for a simple system). q – energy added to the system as heat. Positive (+) when the system gains heat from outside (……………. process), negative (-) when heat flows out of the system (……………. process). w - work done by the system o ...