Gizmos: Types of Reactions
... Introduction: Chemical equations show how compounds and elements react with one another. An element is a substance consisting of one kind of atom, such as aluminum (Al) or oxygen gas (O2). A compound is a substance made of more than one kind of atom, such as water (H2O) or table salt (NaCl). Questio ...
... Introduction: Chemical equations show how compounds and elements react with one another. An element is a substance consisting of one kind of atom, such as aluminum (Al) or oxygen gas (O2). A compound is a substance made of more than one kind of atom, such as water (H2O) or table salt (NaCl). Questio ...
Ch 5.1 The Nature of Chemical Reactions
... • Understand parts to a chemical equation (reactants, products, yeild sign, double arrow) • Conservation of matter is expressed through balancing chemical equations • Describe difference between endothermic and exothermic reactions ...
... • Understand parts to a chemical equation (reactants, products, yeild sign, double arrow) • Conservation of matter is expressed through balancing chemical equations • Describe difference between endothermic and exothermic reactions ...
weekly schedule and topics
... This course will discuss the fundamental issues and problems related to a range of topics which are currently at the forefront of heavy inorganic industrial chemistry. The general topics deal with such areas as the development of industrial chemical processes, the environmental protection and air po ...
... This course will discuss the fundamental issues and problems related to a range of topics which are currently at the forefront of heavy inorganic industrial chemistry. The general topics deal with such areas as the development of industrial chemical processes, the environmental protection and air po ...
Transport of Material through Air, Soil, and Water
... over a wide area, the toxicity of the chemical will be reduced. The problem lies with insoluble substances deposited near the pollution source. As they accumulate, the substances will create an area of contamination. ...
... over a wide area, the toxicity of the chemical will be reduced. The problem lies with insoluble substances deposited near the pollution source. As they accumulate, the substances will create an area of contamination. ...
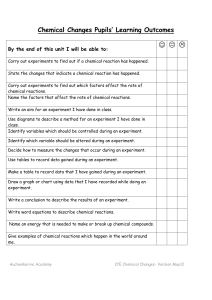
Chemical Reactions Unit Pupils` Learning Outcomes
... Identify variables which should be controlled during an experiment. Identify which variable should be altered during an experiment. Decide how to measure the changes that occur during an experiment. Use tables to record data gained during an experiment. Make a table to record data that I have gained ...
... Identify variables which should be controlled during an experiment. Identify which variable should be altered during an experiment. Decide how to measure the changes that occur during an experiment. Use tables to record data gained during an experiment. Make a table to record data that I have gained ...
Document
... In a chemical reaction a new substance is always formed. Most chemical changes are not easily reversed; they are irreversible. In a physical change no new substance is formed. Melting and evaporation are examples of physical changes. Physical changes are usually reversible. You can tell that a react ...
... In a chemical reaction a new substance is always formed. Most chemical changes are not easily reversed; they are irreversible. In a physical change no new substance is formed. Melting and evaporation are examples of physical changes. Physical changes are usually reversible. You can tell that a react ...
Balancing chemical equations notes
... chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, and this requires that all chemical reactions be balanced. Consider the following balanced equation: Cu (s) + 4 HNO3 (aq) Cu(NO ...
... chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, and this requires that all chemical reactions be balanced. Consider the following balanced equation: Cu (s) + 4 HNO3 (aq) Cu(NO ...
Physical Science Chapter 6
... Paragraph 4 Synthesis: combine 2+ elements/compounds to make new substance; ...
... Paragraph 4 Synthesis: combine 2+ elements/compounds to make new substance; ...
Chapter 4
... Compute the amount of product produced or reactant consumed by two or more simultaneous reactions. ...
... Compute the amount of product produced or reactant consumed by two or more simultaneous reactions. ...
Avoid Chemicals - Clean Eating Living Well
... and Read the ingredient labels of EVERYTHING you eat. The only place to look for what is really in the food is on the INGREDIENT LABEL, you cannot rely on the front packaging. Anything on the front of a package or box is considered the “marketing space” and it is NOT regulated by the FDA (Food and D ...
... and Read the ingredient labels of EVERYTHING you eat. The only place to look for what is really in the food is on the INGREDIENT LABEL, you cannot rely on the front packaging. Anything on the front of a package or box is considered the “marketing space” and it is NOT regulated by the FDA (Food and D ...
8492_Chemichal Weapons Production Indicators
... At room temperature (70°F), phosgene is a poisonous gas. ...
... At room temperature (70°F), phosgene is a poisonous gas. ...
TERM 2 Unit 3 YR 9 SCI It is elementary
... In this unit students explore the historical development of understandings of atomic structure. Students model an atom according to currently accepted understandings. They will identify patterns in atomic structure that allow prediction of the products of chemical reactions and are reflected by the ...
... In this unit students explore the historical development of understandings of atomic structure. Students model an atom according to currently accepted understandings. They will identify patterns in atomic structure that allow prediction of the products of chemical reactions and are reflected by the ...
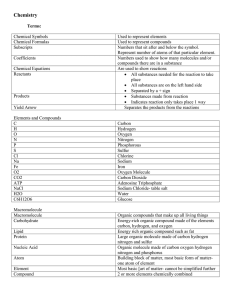
Unit 2: Chemical Reactions
... • A chemical formula is an abbreviation for a chemical compound using chemical symbols and numbers. • The subscript number tells how many atoms of the element are present in the compound • Example: CO2 = Carbon Dioxide – Di = 2 – 1 Carbon atom and 2 oxygen atoms ...
... • A chemical formula is an abbreviation for a chemical compound using chemical symbols and numbers. • The subscript number tells how many atoms of the element are present in the compound • Example: CO2 = Carbon Dioxide – Di = 2 – 1 Carbon atom and 2 oxygen atoms ...
Chemistry and the Environment - Teach-n-Learn-Chem
... 6. What class of compounds used as coolants and insulators was banned because of environmental hazards? PCB’s 7. What is the name of the 1980 law to clean up waste sites? Super Fund 8. When chemical wastes are burned what materials are released into the environment? Carbon dioxide and water vapor if ...
... 6. What class of compounds used as coolants and insulators was banned because of environmental hazards? PCB’s 7. What is the name of the 1980 law to clean up waste sites? Super Fund 8. When chemical wastes are burned what materials are released into the environment? Carbon dioxide and water vapor if ...
Episode 25 0 Chemistry and the Environment
... 6. What class of compounds used as coolants and insulators was banned because of environmental hazards? PCB’s 7. What is the name of the 1980 law to clean up waste sites? Super Fund 8. When chemical wastes are burned what materials are released into the environment? Carbon dioxide and water vapor if ...
... 6. What class of compounds used as coolants and insulators was banned because of environmental hazards? PCB’s 7. What is the name of the 1980 law to clean up waste sites? Super Fund 8. When chemical wastes are burned what materials are released into the environment? Carbon dioxide and water vapor if ...
Answer Key - La Quinta High School
... takes place. However, the only evidence for this reaction is the release of heat energy, which should be evident as a temperature change for the mixture. Since water has a relatively high specific heat capacity, however, if the acid and base solutions are very dilute, the temperature may change only ...
... takes place. However, the only evidence for this reaction is the release of heat energy, which should be evident as a temperature change for the mixture. Since water has a relatively high specific heat capacity, however, if the acid and base solutions are very dilute, the temperature may change only ...
green chemistry
... control during syntheses to minimize or eliminate the formation of byproducts. 6. Minimize the potential for accidents: Design chemicals and their forms (solid, liquid, or gas) to minimize the potential for chemical accidents including explosions, fires, and releases to the environment. 7. Prevent w ...
... control during syntheses to minimize or eliminate the formation of byproducts. 6. Minimize the potential for accidents: Design chemicals and their forms (solid, liquid, or gas) to minimize the potential for chemical accidents including explosions, fires, and releases to the environment. 7. Prevent w ...
Unit 5 Chemical Properties and Changes Video Notes A ______ is a
... ________________________ A change that alters the identity of a substance resulting in a new substance or substances with different properties ________________________ Those characteristics that can be observed when a chemical reaction changes the identity of the substance, such as potential to rus ...
... ________________________ A change that alters the identity of a substance resulting in a new substance or substances with different properties ________________________ Those characteristics that can be observed when a chemical reaction changes the identity of the substance, such as potential to rus ...
Chemical Reaction Basics
... Advanced Chemistry – Chapter 8 A ____________ ____________ is a process by which one or more substances are changed into one or more ____________ substances. Chemical reactions are represented by some type of ____________. The general form is as follows: ...
... Advanced Chemistry – Chapter 8 A ____________ ____________ is a process by which one or more substances are changed into one or more ____________ substances. Chemical reactions are represented by some type of ____________. The general form is as follows: ...
Unlike other amorphous thermoplastics, ULTEM resin offers
... Unlike other amorphous thermoplastics, ULTEM resin offers unusually good resistance to many types of chemicals. Typically, this performance can be demonstrated by ULTEM resin’s ability to perform better in aggressive environments at higher temperatures, for longer periods of time, and under higher l ...
... Unlike other amorphous thermoplastics, ULTEM resin offers unusually good resistance to many types of chemicals. Typically, this performance can be demonstrated by ULTEM resin’s ability to perform better in aggressive environments at higher temperatures, for longer periods of time, and under higher l ...
Chemical industry

The chemical industry comprises the companies that produce industrial chemicals. Central to the modern world economy, it converts raw materials (oil, natural gas, air, water, metals, and minerals) into more than 70,000 different products.