Name___________________________________ Physical
... CHEMICAL REACTIONS - P. 582 12) The process in which the atoms of one or more substance are rearranged to form different substances is called a(n) ____________________. A) nuclear reaction B) substantiation C) chemical reaction D) physical reaction ...
... CHEMICAL REACTIONS - P. 582 12) The process in which the atoms of one or more substance are rearranged to form different substances is called a(n) ____________________. A) nuclear reaction B) substantiation C) chemical reaction D) physical reaction ...
Reactions
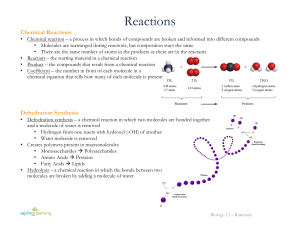
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
... • Chemical reaction – a process in which bonds of compounds are broken and reformed into different compounds • Molecules are rearranged during reactions, but composition stays the same • There are the same number of atoms in the products as there are in the reactants • Reactant – the starting materi ...
Chemical Reactions are…
... The combination of chemicals before the reaction are on the left side of the arrow, called the reactants The right side indicates the combination of chemicals after the reaction, called the product. ...
... The combination of chemicals before the reaction are on the left side of the arrow, called the reactants The right side indicates the combination of chemicals after the reaction, called the product. ...
Matter and Energy
... -determines the number of molecules (groups) of the formula -This number will be DISTRIBUTED just like in math. It applies to each element and is multiplied by each subscript to find the total number of atoms of each element and a total number of atoms in the molecule. ...
... -determines the number of molecules (groups) of the formula -This number will be DISTRIBUTED just like in math. It applies to each element and is multiplied by each subscript to find the total number of atoms of each element and a total number of atoms in the molecule. ...
Practical, Asymmetric Redox-Neutral Chemical Synthesis via Borrowing Hydrogen
... applied areas such as pharmaceuticals, chemical biology and material science. Due to resource constraints, the current trend in synthetic chemistry is not simply about preparing molecules of specific interest, but rather on how to prepare them in a highly efficient and economical manner. The concept ...
... applied areas such as pharmaceuticals, chemical biology and material science. Due to resource constraints, the current trend in synthetic chemistry is not simply about preparing molecules of specific interest, but rather on how to prepare them in a highly efficient and economical manner. The concept ...
California Enacts Nation`s First Green Chemistry Program
... This Mayer Brown LLP publication provides information and comments on legal issues and developments of interest to our clients and friends. The foregoing is not a comprehensive treatment of the subject matter covered and is not intended to provide legal advice. Readers should seek specific legal adv ...
... This Mayer Brown LLP publication provides information and comments on legal issues and developments of interest to our clients and friends. The foregoing is not a comprehensive treatment of the subject matter covered and is not intended to provide legal advice. Readers should seek specific legal adv ...
Document
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
Bacteria and Virus Research Jigsaw
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
Chemical Reactions
... 3. THINK VISUALLY The graphs below show the amount of energy present during two chemical reactions. One of the reactions is an energy-absorbing reaction, the other is an energy-releasing reaction. Label the type of reaction for each, label the energy level for the reactants and products, then draw a ...
... 3. THINK VISUALLY The graphs below show the amount of energy present during two chemical reactions. One of the reactions is an energy-absorbing reaction, the other is an energy-releasing reaction. Label the type of reaction for each, label the energy level for the reactants and products, then draw a ...
Study Guide – Unit Test (9-27-13)
... Boiling Point – temperature at which a liquid turns to a gas (water boils at 100 degrees Fahrenheit) Flammability – ability to burn (paper or wood burning) Combustibility – ability to explode (gun powder exploding) Reactivity (with water, acid, and oxygen) – ability to cause a chemical change when i ...
... Boiling Point – temperature at which a liquid turns to a gas (water boils at 100 degrees Fahrenheit) Flammability – ability to burn (paper or wood burning) Combustibility – ability to explode (gun powder exploding) Reactivity (with water, acid, and oxygen) – ability to cause a chemical change when i ...
Section 2-4 “Chemical Reactions and Enzymes”
... Reactants – Elements or compounds that are the “starter materials” for a reaction Products – Elements or compounds produced by a chemical reaction ...
... Reactants – Elements or compounds that are the “starter materials” for a reaction Products – Elements or compounds produced by a chemical reaction ...
Classifying Chemical Reactions 9-3
... Balance the atoms of an element one at a time by adding coefficients (the numbers in front) - save H and O until LAST! Check to make sure it is balanced. ...
... Balance the atoms of an element one at a time by adding coefficients (the numbers in front) - save H and O until LAST! Check to make sure it is balanced. ...
Estimating Mineral Weathering Rates in Catskills
... ◘ Basic Cations: Ca, Mg, K, Na ◘ Silica: H4SiO4 ◘ Aluminum: potentially toxic to aquatic biota ...
... ◘ Basic Cations: Ca, Mg, K, Na ◘ Silica: H4SiO4 ◘ Aluminum: potentially toxic to aquatic biota ...
Chemistry Standards Checklist
... and their placement on the Periodic Table. SC5. Students will understand that the rate at which a chemical reaction occurs can be affected by changing concentration, temperature, or pressure and the addition of a catalyst. a. Demonstrate the effects of changing concentration, temperature, and pres ...
... and their placement on the Periodic Table. SC5. Students will understand that the rate at which a chemical reaction occurs can be affected by changing concentration, temperature, or pressure and the addition of a catalyst. a. Demonstrate the effects of changing concentration, temperature, and pres ...
Ch. 3 - Chemical Reactions
... three units of aqueous copper(II) chloride to produce three atoms of copper and two units of aqueous aluminum chloride. • How many? • Of what? • In what state? ...
... three units of aqueous copper(II) chloride to produce three atoms of copper and two units of aqueous aluminum chloride. • How many? • Of what? • In what state? ...
Process Safety Management
... chemical to break down? • Does the process cause the chemical to boil or freeze? ...
... chemical to break down? • Does the process cause the chemical to boil or freeze? ...
Element - the simplest form of matter that can exist under normal
... Elements are the building blocks for all other substances There are now 117 known elements (as of 2006). All elements after uranium on the periodic table are man-made. A compound is a chemical combination of two or more different elements joined together in a fixed proportion. Every compound has its ...
... Elements are the building blocks for all other substances There are now 117 known elements (as of 2006). All elements after uranium on the periodic table are man-made. A compound is a chemical combination of two or more different elements joined together in a fixed proportion. Every compound has its ...
Chemical Equations and Tests for anions
... these anions are present and not to see how much of them is present this is known as Qualitative analysis • Testing to see how much of a substance is present is known as Quantitative analysis ...
... these anions are present and not to see how much of them is present this is known as Qualitative analysis • Testing to see how much of a substance is present is known as Quantitative analysis ...
The only sure evidence that a chemical reaction has occured is
... What is shown by A in Graph 1? What is shown by B in Graph 1? What type of reaction is shown in Graph 1? Which graph illustrates the type of reaction that occurs when wood burns? ...
... What is shown by A in Graph 1? What is shown by B in Graph 1? What type of reaction is shown in Graph 1? Which graph illustrates the type of reaction that occurs when wood burns? ...
Chemical industry

The chemical industry comprises the companies that produce industrial chemicals. Central to the modern world economy, it converts raw materials (oil, natural gas, air, water, metals, and minerals) into more than 70,000 different products.