Atoms, Elements, Compounds File
... Compounds can be represented by chemical formulas. Each different element in the compound is represented by its unique symbol. The number of each type of element in the compound (other than 1) is represented by a small number (the subscript) to the right of the element symbol. ...
... Compounds can be represented by chemical formulas. Each different element in the compound is represented by its unique symbol. The number of each type of element in the compound (other than 1) is represented by a small number (the subscript) to the right of the element symbol. ...
Chem vocab quiz definitons
... Proton is a positively charged particles that are found in the nucleus, and has the mass of 1 atomic unit (amu). Neutron is an electrically neutral particles that are found in the nucleus, and has a mass of 1 amu. Electron is a negatively charged sub atomic particle found in an area outside the nucl ...
... Proton is a positively charged particles that are found in the nucleus, and has the mass of 1 atomic unit (amu). Neutron is an electrically neutral particles that are found in the nucleus, and has a mass of 1 amu. Electron is a negatively charged sub atomic particle found in an area outside the nucl ...
Stars: Element factories.
... • Mass number (A): number of protons + number of neutrons in an atom •A is often indicated in the top left corner of the element symbol ...
... • Mass number (A): number of protons + number of neutrons in an atom •A is often indicated in the top left corner of the element symbol ...
Chemical reactions revision
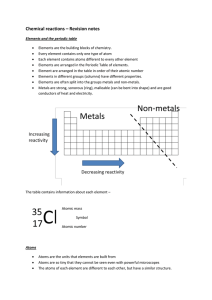
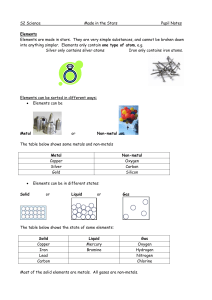
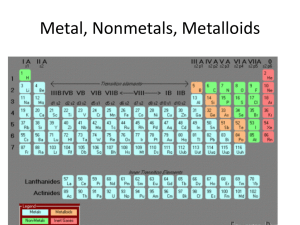
... Elements in different groups (columns) have different properties. Elements are often split into the groups metals and non-metals. Metals are strong, sonorous (ring), malleable (can be bent into shape) and are good conductors of heat and electricity. ...
... Elements in different groups (columns) have different properties. Elements are often split into the groups metals and non-metals. Metals are strong, sonorous (ring), malleable (can be bent into shape) and are good conductors of heat and electricity. ...
L.O.
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
Made in the Stars Notes
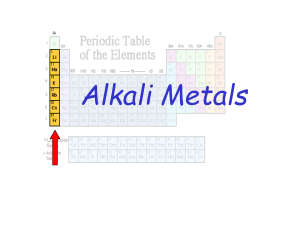
... The Periodic Table This is a way of organising the elements. Each element has its own chemical symbol, e.g. copper is Cu and Iron is Fe. The horizontal rows in the periodic table are called Periods. The vertical columns are called Groups: Group 1 Alkali metals Group 2 Alkali earth metals Group 7 Hal ...
... The Periodic Table This is a way of organising the elements. Each element has its own chemical symbol, e.g. copper is Cu and Iron is Fe. The horizontal rows in the periodic table are called Periods. The vertical columns are called Groups: Group 1 Alkali metals Group 2 Alkali earth metals Group 7 Hal ...
Stars Answers - Science Skool!
... 8. What happens to massive stars after the red super giant stage? Outer layers are thrown into space which scatters gas and dust into space and distributes the elements throughout space. The core left behind forms a neutron star or black hole if sufficient mass is left behind 9. Why do scientists be ...
... 8. What happens to massive stars after the red super giant stage? Outer layers are thrown into space which scatters gas and dust into space and distributes the elements throughout space. The core left behind forms a neutron star or black hole if sufficient mass is left behind 9. Why do scientists be ...
C2- Topic 1: Atomic structure and the periodic table. Assessable
... - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
... - arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
C2 Topic 1 Can Do Sheet
... a arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds b used his table to predict the existence and properties of some elements not then discovered 1.2 Classify elements as metals or non-metals according to their position in the pe ...
... a arranged the elements, known at that time, in a periodic table by using properties of these elements and their compounds b used his table to predict the existence and properties of some elements not then discovered 1.2 Classify elements as metals or non-metals according to their position in the pe ...
Periodic Table Puzzle
... The code letters A to Z have been assigned to represent the first 26 representative elements in the Periodic Table. The letters do not relate to the actual chemical symbols for these elements. Your challenge is to put the code letters in the correct boxes in the Periodic Table, based on the ...
... The code letters A to Z have been assigned to represent the first 26 representative elements in the Periodic Table. The letters do not relate to the actual chemical symbols for these elements. Your challenge is to put the code letters in the correct boxes in the Periodic Table, based on the ...
Chapter 6 Vocabulary crossword puzzle
... 3. Elements in which the highest occupied s and p sublevels are partially filled 6. Measures the ability of an atom to attract electrons when the atom is in a compound; the element named Cesium has the lowest amount, while the element named Fluorine has the highest amount 7. Term that refers to a se ...
... 3. Elements in which the highest occupied s and p sublevels are partially filled 6. Measures the ability of an atom to attract electrons when the atom is in a compound; the element named Cesium has the lowest amount, while the element named Fluorine has the highest amount 7. Term that refers to a se ...
CHAPTER6_MEET_THE_ELEMENTS
... 1. What are the 4 main gases found in our atmosphere? 2. Why is photosynthesis vital for life on Earth? 3. What is the hydrosphere? 4. List one element and 4 compounds found in our hydrosphere? 5. What is the importance of dissolved 02 and CO2 in our hydrosphere? 6. List the four most common element ...
... 1. What are the 4 main gases found in our atmosphere? 2. Why is photosynthesis vital for life on Earth? 3. What is the hydrosphere? 4. List one element and 4 compounds found in our hydrosphere? 5. What is the importance of dissolved 02 and CO2 in our hydrosphere? 6. List the four most common element ...
The Chemical Basis of Life Chapter 4
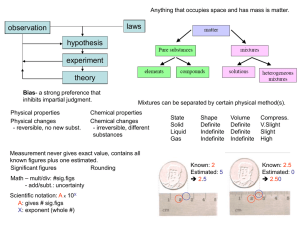
... How is Matter Organized? •Divided into pure substances or mixtures based on composition. –Pure substance •Uniform composition with same properties throughout •Can be classified as either an element or a compound ...
... How is Matter Organized? •Divided into pure substances or mixtures based on composition. –Pure substance •Uniform composition with same properties throughout •Can be classified as either an element or a compound ...
Earth - Capital High School
... they looked like millions of years ago – it takes one year for light to travel 5+ trillion miles (a light year) ...
... they looked like millions of years ago – it takes one year for light to travel 5+ trillion miles (a light year) ...
Chapter 14 Review
... 9. What information can be obtained by knowing the atomic number of an element? ...
... 9. What information can be obtained by knowing the atomic number of an element? ...
Homework Geochem Test Review
... 2. What is smallest part of an element that has all the properties of that element? ...
... 2. What is smallest part of an element that has all the properties of that element? ...
Nineteenth lecture
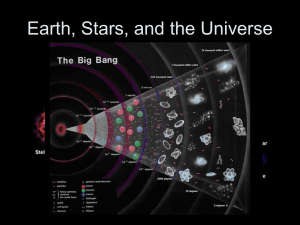
... …. creating a new nebula that included much heavier elements than the old original star elements above iron in the Periodic Table of the Elements (the transferric elements). Much of our hydrogen and helium was lost to space, only some remaining in what would become the outer planets (Jupiter, Saturn ...
... …. creating a new nebula that included much heavier elements than the old original star elements above iron in the Periodic Table of the Elements (the transferric elements). Much of our hydrogen and helium was lost to space, only some remaining in what would become the outer planets (Jupiter, Saturn ...
chapters 1-4
... A compound is a distinct substance that contains two or more elements combined in a definite proportion by weight. Compounds can be decomposed chemically into simpler substances – that is, into simpler compounds or elements. ...
... A compound is a distinct substance that contains two or more elements combined in a definite proportion by weight. Compounds can be decomposed chemically into simpler substances – that is, into simpler compounds or elements. ...
Chemical Basis of Life
... Title: The Chemical Basis of Life 1- Introduction: Your body is an elaborate chemical system. Chemical reactions power all of the body’s activities. At the most basic level, life is about chemicals and how they interact with each other. 2- Matter – Matter is anything that has mass and occupies space ...
... Title: The Chemical Basis of Life 1- Introduction: Your body is an elaborate chemical system. Chemical reactions power all of the body’s activities. At the most basic level, life is about chemicals and how they interact with each other. 2- Matter – Matter is anything that has mass and occupies space ...
Abundance of the chemical elements

The abundance of a chemical element measures how common is the element relative to all other elements in a given environment. Abundance is measured in one of three ways: by the mass-fraction (the same as weight fraction); by the mole-fraction (fraction of atoms by numerical count, or sometimes fraction of molecules in gases); or by the volume-fraction. Volume-fraction is a common abundance measure in mixed gases such as planetary atmospheres, and is similar in value to molecular mole-fraction for gas mixtures at relatively low densities and pressures, and ideal gas mixtures. Most abundance values in this article are given as mass-fractions. For example, the abundance of oxygen in pure water can be measured in two ways: the mass fraction is about 89%, because that is the fraction of water's mass which is oxygen. However, the mole-fraction is 33.3333...% because only 1 atom of 3 in water, H2O, is oxygen. As another example, looking at the mass-fraction abundance of hydrogen and helium in both the Universe as a whole and in the atmospheres of gas-giant planets such as Jupiter, it is 74% for hydrogen and 23-25% for helium; while the (atomic) mole-fraction for hydrogen is 92%, and for helium is 8%, in these environments. Changing the given environment to Jupiter's outer atmosphere, where hydrogen is diatomic while helium is not, changes the molecular mole-fraction (fraction of total gas molecules), as well as the fraction of atmosphere by volume, of hydrogen to about 86%, and of helium to 13%.