World of
... Rules for Writing Formulas 1.Each atom present is represented by its element symbol. 2.The number of each type of atom is indicated by a subscript written to the right of the element symbol. 3.When only one atom of a given type is present, the subscript 1 is not written. Copyright© by Houghton Miff ...
... Rules for Writing Formulas 1.Each atom present is represented by its element symbol. 2.The number of each type of atom is indicated by a subscript written to the right of the element symbol. 3.When only one atom of a given type is present, the subscript 1 is not written. Copyright© by Houghton Miff ...
JJ Thompson Webquest
... Definite Proportions (when atoms combine to form a particular compound, they always combine in the same ratios by weight) and Proust’s Law of Constant Compostion (States that in a pure compound, the elements are always present in the same definite proportion by mass).” Proposed an "atomic theory" wi ...
... Definite Proportions (when atoms combine to form a particular compound, they always combine in the same ratios by weight) and Proust’s Law of Constant Compostion (States that in a pure compound, the elements are always present in the same definite proportion by mass).” Proposed an "atomic theory" wi ...
t2 images part 1
... If the Universe is expanding, it stands that at some point in the past everything in the Universe was all concentrated at the same point and began expanding outward. This point in time is called the “Big Bang” The Big Bang happened somewhere between 1016 Billion years ago ...
... If the Universe is expanding, it stands that at some point in the past everything in the Universe was all concentrated at the same point and began expanding outward. This point in time is called the “Big Bang” The Big Bang happened somewhere between 1016 Billion years ago ...
neutron star - Adams State University
... Other reactions create elements up to iron. (There are a lot more reactions than are listed here.) ...
... Other reactions create elements up to iron. (There are a lot more reactions than are listed here.) ...
Ch2 lecture outline - OnCourse Systems For Education
... • The _____________ of an electron • And then later • The _____________ of the electron 11. What was used to discover protons? ...
... • The _____________ of an electron • And then later • The _____________ of the electron 11. What was used to discover protons? ...
File
... Substances formed when two or more different elements chemically combine—these have properties that differ from the elements that make them up ...
... Substances formed when two or more different elements chemically combine—these have properties that differ from the elements that make them up ...
DO NOT WRITE ON THIS PAPER Standard 1 Objective 1 Study
... 15.The time it takes for half of a sample of a radioactive isotope to decay is called a Halflife. 16.Radioactive dating is used to determine the absolute age of rocks because radioactive decay happens at a relatively constant rate. 17.Radiometric dating is determining the age of a substance by compa ...
... 15.The time it takes for half of a sample of a radioactive isotope to decay is called a Halflife. 16.Radioactive dating is used to determine the absolute age of rocks because radioactive decay happens at a relatively constant rate. 17.Radiometric dating is determining the age of a substance by compa ...
File
... Matter is anything that has mass and takes up space. Mass is the amount of matter (how much stuff) an object contains. The mass of an object will not change if the force of gravity on it changes. For mass, the SI unit is kilogram (kg). The amount of space that matter occupies is the volume. Common u ...
... Matter is anything that has mass and takes up space. Mass is the amount of matter (how much stuff) an object contains. The mass of an object will not change if the force of gravity on it changes. For mass, the SI unit is kilogram (kg). The amount of space that matter occupies is the volume. Common u ...
投影片 - 中正大學化生系
... 3. The arrangement of the elements in groups of elements in the order of their atomic weights corresponds to their so-called valencies, as well as, to some extent, to their distinctive chemical properties; as is apparent among other series in that of Li, Be, B, C, N, O, and F. 4. The magnitude of th ...
... 3. The arrangement of the elements in groups of elements in the order of their atomic weights corresponds to their so-called valencies, as well as, to some extent, to their distinctive chemical properties; as is apparent among other series in that of Li, Be, B, C, N, O, and F. 4. The magnitude of th ...
希臘 - 中正大學化生系
... 3. The arrangement of the elements in groups of elements in the order of their atomic weights corresponds to their so-called valencies, as well as, to some extent, to their distinctive chemical properties; as is apparent among other series in that of Li, Be, B, C, N, O, and F. 4. The magnitude of th ...
... 3. The arrangement of the elements in groups of elements in the order of their atomic weights corresponds to their so-called valencies, as well as, to some extent, to their distinctive chemical properties; as is apparent among other series in that of Li, Be, B, C, N, O, and F. 4. The magnitude of th ...
What is a mixture?
... Identifying Elements • Elements are categorized by unique properties on the Periodic Table. • They are arranged in order by their number of protons. (More on this later!) • Each element has unique properties like melting point, boiling point, and whether it is metal, nonmetal or metalloid. ...
... Identifying Elements • Elements are categorized by unique properties on the Periodic Table. • They are arranged in order by their number of protons. (More on this later!) • Each element has unique properties like melting point, boiling point, and whether it is metal, nonmetal or metalloid. ...
Slide 1 - Effingham County Schools
... • Aristotle was wrong. However, his theory persisted for 2000 years. ...
... • Aristotle was wrong. However, his theory persisted for 2000 years. ...
The Atom Power point - Effingham County Schools
... • Aristotle was wrong. However, his theory persisted for 2000 years. ...
... • Aristotle was wrong. However, his theory persisted for 2000 years. ...
Chapter 16: The Origin of the Solar System RQ 16
... was near to the center of the system), that only matters with high “boiling points” were able to condense. Therefore, only metals and then also silicates could condense to form solids. At these inner orbits, material with lower boiling points (lighter elements) could not be collected, since it still ...
... was near to the center of the system), that only matters with high “boiling points” were able to condense. Therefore, only metals and then also silicates could condense to form solids. At these inner orbits, material with lower boiling points (lighter elements) could not be collected, since it still ...
Elements, Compounds, Mixtures
... • Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. • Atoms of different elements combine in simple whole-number ratios to form chemical compounds. • 1803-1805: Dalton published his first table of relat ...
... • Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. • Atoms of different elements combine in simple whole-number ratios to form chemical compounds. • 1803-1805: Dalton published his first table of relat ...
THE INCREDIBLE ORIGIN OF THE CHEMICAL ELEMENTS
... amounts of matter into space, including all the elements they have built up. Stars several times as massive as our Sun, or heavier, shrink catastrophically, forming "neutron stars1" or even "black holes2". In many cases an enormous explosion (known as a "supernova") results, throwing much of the ou ...
... amounts of matter into space, including all the elements they have built up. Stars several times as massive as our Sun, or heavier, shrink catastrophically, forming "neutron stars1" or even "black holes2". In many cases an enormous explosion (known as a "supernova") results, throwing much of the ou ...
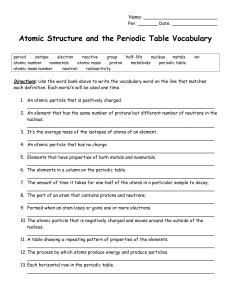
Atomic Structure and the Periodic Table Vocabulary
... Choose the vocabulary word that matches each description by circling it. Use the bolded words in the sentences as clues. 19. Sometimes this is called a family of elements because these elements seem to be ...
... Choose the vocabulary word that matches each description by circling it. Use the bolded words in the sentences as clues. 19. Sometimes this is called a family of elements because these elements seem to be ...
Lecture 16 - Yet More Evolution of Stars
... • Imagine we have a cluster of stars that were all formed at the same time, but have a variety of different masses • Using what we know about stellar evolution is there a way to determine the age of the star cluster? ...
... • Imagine we have a cluster of stars that were all formed at the same time, but have a variety of different masses • Using what we know about stellar evolution is there a way to determine the age of the star cluster? ...
Periodic Table Vocab page 7
... Mass number: the number of protons and neutrons in the nucleus of an atom. Mass number = protons + neutrons Valence #: The number of outermost electrons in an atom. Periodic Table: An arrangement of elements in order of increasing atomic numbers that demonstrates the periodic patterns that occur amo ...
... Mass number: the number of protons and neutrons in the nucleus of an atom. Mass number = protons + neutrons Valence #: The number of outermost electrons in an atom. Periodic Table: An arrangement of elements in order of increasing atomic numbers that demonstrates the periodic patterns that occur amo ...
Chemistry Study Guide
... position on the table will show many of its general properties Periods- The table is arranged in horizontal rows called periods. The period tells you how many electron energy levels the atom has. Groups- Vertical columns whose members of the same group have similar chemical and physical properti ...
... position on the table will show many of its general properties Periods- The table is arranged in horizontal rows called periods. The period tells you how many electron energy levels the atom has. Groups- Vertical columns whose members of the same group have similar chemical and physical properti ...
Chemistry Study Guide
... position on the table will show many of its general properties Periods- The table is arranged in horizontal rows called periods. The period tells you how many electron energy levels the atom has. Groups- Vertical columns whose members of the same group have similar chemical and physical properti ...
... position on the table will show many of its general properties Periods- The table is arranged in horizontal rows called periods. The period tells you how many electron energy levels the atom has. Groups- Vertical columns whose members of the same group have similar chemical and physical properti ...
Maybe We Are Alone in the Universe, After All
... another by the emptiness of interstellar space. Just for Earth's own galaxy, the Milky Way, experts have estimated that there might be up to one million advanced societies. Now, two prominent scientists say the conventional wisdom is wrong. The alien search, they add, is likely to fail. The two argu ...
... another by the emptiness of interstellar space. Just for Earth's own galaxy, the Milky Way, experts have estimated that there might be up to one million advanced societies. Now, two prominent scientists say the conventional wisdom is wrong. The alien search, they add, is likely to fail. The two argu ...
Abundance of the chemical elements

The abundance of a chemical element measures how common is the element relative to all other elements in a given environment. Abundance is measured in one of three ways: by the mass-fraction (the same as weight fraction); by the mole-fraction (fraction of atoms by numerical count, or sometimes fraction of molecules in gases); or by the volume-fraction. Volume-fraction is a common abundance measure in mixed gases such as planetary atmospheres, and is similar in value to molecular mole-fraction for gas mixtures at relatively low densities and pressures, and ideal gas mixtures. Most abundance values in this article are given as mass-fractions. For example, the abundance of oxygen in pure water can be measured in two ways: the mass fraction is about 89%, because that is the fraction of water's mass which is oxygen. However, the mole-fraction is 33.3333...% because only 1 atom of 3 in water, H2O, is oxygen. As another example, looking at the mass-fraction abundance of hydrogen and helium in both the Universe as a whole and in the atmospheres of gas-giant planets such as Jupiter, it is 74% for hydrogen and 23-25% for helium; while the (atomic) mole-fraction for hydrogen is 92%, and for helium is 8%, in these environments. Changing the given environment to Jupiter's outer atmosphere, where hydrogen is diatomic while helium is not, changes the molecular mole-fraction (fraction of total gas molecules), as well as the fraction of atmosphere by volume, of hydrogen to about 86%, and of helium to 13%.