Chemistry Unit Study Guide Key
... how balanced equations support the law of conservation of mass – The ...
... how balanced equations support the law of conservation of mass – The ...
Ch. 5 Outline Notes
... Ch. 5 Lecture Notes Atomic Structure and the Periodic Table 5-1 Atoms A. Early Models of the Atom 1. ____________________ – 4th century BC – world made up of empty space and tiny particles called _______________ (atomos) ‘indivisible’ a. Hypothesized _________________ using experiments B. __________ ...
... Ch. 5 Lecture Notes Atomic Structure and the Periodic Table 5-1 Atoms A. Early Models of the Atom 1. ____________________ – 4th century BC – world made up of empty space and tiny particles called _______________ (atomos) ‘indivisible’ a. Hypothesized _________________ using experiments B. __________ ...
PowerPoint Presentation - Introduction to Atoms & Nuclei
... -Silicon -Germanium -Tin -Lead • Silicon is used to make semiconductors for computers and other electronics . • Diamond and Graphite are two forms of carbon. ...
... -Silicon -Germanium -Tin -Lead • Silicon is used to make semiconductors for computers and other electronics . • Diamond and Graphite are two forms of carbon. ...
Understanding the Atom GN
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
Alkaline earth metals
... All In the second row Don’t occur as free elements Most commonly are found occurring as the carbonates, phosphates silicates, and sulfates Atoms loose 2 electrons Most are insoluble or slightly soluble Very Reactive ...
... All In the second row Don’t occur as free elements Most commonly are found occurring as the carbonates, phosphates silicates, and sulfates Atoms loose 2 electrons Most are insoluble or slightly soluble Very Reactive ...
Chapter 4 Study Guide Physical Science 1. The word atom comes
... ____________________.” 2. Halogens are very reactive elements located in Group _______of the periodic table. 3. The nucleus of an atom has a(n) ____________________ electric charge. 4. Carbon is found in group ______ of the periodic table. 5. Bohr’s model of the atom compares electrons to __________ ...
... ____________________.” 2. Halogens are very reactive elements located in Group _______of the periodic table. 3. The nucleus of an atom has a(n) ____________________ electric charge. 4. Carbon is found in group ______ of the periodic table. 5. Bohr’s model of the atom compares electrons to __________ ...
Name Period _____ Chemistry Review
... Indicate whether the sentence or statement is true or false. If false, change the identified word or phrase to make the sentence or statement true. ____ 13. A change that produces one or more new substances is called a physical change. _________________________ ____ 14. A(n) pure substance is made o ...
... Indicate whether the sentence or statement is true or false. If false, change the identified word or phrase to make the sentence or statement true. ____ 13. A change that produces one or more new substances is called a physical change. _________________________ ____ 14. A(n) pure substance is made o ...
Life Cycles of Stars
... Composition is > 98% hydrogen and helium. 1/3 of the hydrogen is converted to helium. ...
... Composition is > 98% hydrogen and helium. 1/3 of the hydrogen is converted to helium. ...
Stellar Nucleosynthesis
... • After 1 million years matter began to cool enough to form atoms- Hydrogen- the building block of stars ...
... • After 1 million years matter began to cool enough to form atoms- Hydrogen- the building block of stars ...
Classification of Matter
... 1. Elements are the simplest form of matter. 2. Elements are the building blocks of all substances and cannot be easily divided into smaller subunits by ordinary chemical processes. 3. Elements are organized by atomic number on the periodic table. 4. Elements are identified by their symbols. ...
... 1. Elements are the simplest form of matter. 2. Elements are the building blocks of all substances and cannot be easily divided into smaller subunits by ordinary chemical processes. 3. Elements are organized by atomic number on the periodic table. 4. Elements are identified by their symbols. ...
Learning Standards vocab chemical basis and molecules of life 09
... Given the number of protons, identify the element using a Periodic Table. Explain the arrangement of the elements on the Periodic Table, including the significant relationships among elements in a given column or row. Explain how ions and ionic bonds are formed (e.g., sodium atoms lose an elec ...
... Given the number of protons, identify the element using a Periodic Table. Explain the arrangement of the elements on the Periodic Table, including the significant relationships among elements in a given column or row. Explain how ions and ionic bonds are formed (e.g., sodium atoms lose an elec ...
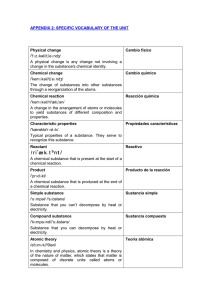
specific vocabulary of the unit
... They inform you of the number of molecules or estequiométricos atoms that take part in a chemical reaction. reaction Acid rain /'æsɪd//reɪn/ ...
... They inform you of the number of molecules or estequiométricos atoms that take part in a chemical reaction. reaction Acid rain /'æsɪd//reɪn/ ...
CH 5 Periodic Law
... Salt: a compound composed of positive and negative ions arranged in a regular 3D pattern - most reactive group of nonmetals - varying physical properties, similar chemical properties ...
... Salt: a compound composed of positive and negative ions arranged in a regular 3D pattern - most reactive group of nonmetals - varying physical properties, similar chemical properties ...
Slides from Lecture09
... How is Energy Generated? (and how much energy?) • Mass is converted into energy! • The total mass of the nuclei (or nucleus) produced by a reaction, Moutput, is less than the total mass of the nuclei (or nucleus) that start(s) the reaction, Minput. • E = (DM)c2 – Here, the quantity, DM = (Minput – ...
... How is Energy Generated? (and how much energy?) • Mass is converted into energy! • The total mass of the nuclei (or nucleus) produced by a reaction, Moutput, is less than the total mass of the nuclei (or nucleus) that start(s) the reaction, Minput. • E = (DM)c2 – Here, the quantity, DM = (Minput – ...
matter and its reactivity. Objects in the universe are composed of
... 3.1a Substances have characteristic properties. Some of these properties include color, odor, phase, density, solubility, heat and electrical conductivity, and boiling and freezing points. 3.1b Solubility can be affected by the nature of the solute and solvent, temperature, and pressure. The rate of ...
... 3.1a Substances have characteristic properties. Some of these properties include color, odor, phase, density, solubility, heat and electrical conductivity, and boiling and freezing points. 3.1b Solubility can be affected by the nature of the solute and solvent, temperature, and pressure. The rate of ...
Chapter 2: Elements are the building blocks of matter
... • Organizes the elements according to their physical and chemical properties • The one we use was developed by Dmitri Mendeleev in 1867 ...
... • Organizes the elements according to their physical and chemical properties • The one we use was developed by Dmitri Mendeleev in 1867 ...
Atoms, Molecules and Ions In This Chapter
... • After a very short time (matter of seconds) that the universe was produced by the Big Bang the only elements present were hydrogen and helium. • After millions of years the cooling of the universe caused the hydrogen and helium to collect together and form large clouds, due to gravity. • These clo ...
... • After a very short time (matter of seconds) that the universe was produced by the Big Bang the only elements present were hydrogen and helium. • After millions of years the cooling of the universe caused the hydrogen and helium to collect together and form large clouds, due to gravity. • These clo ...
Timeline Assignment
... Early Greek philosophers believed all matter was made up of four “elements” earth, air, water, and fire ...
... Early Greek philosophers believed all matter was made up of four “elements” earth, air, water, and fire ...
Atoms: The Building Blocks of Matter
... to show that matter was not created or destroyed during chemical reactions – Law of Conservation of Matter (Mass). • Joseph Proust (1799)- showed that compounds always contain exactly the same proportion of elements by mass– Law of constant composition ...
... to show that matter was not created or destroyed during chemical reactions – Law of Conservation of Matter (Mass). • Joseph Proust (1799)- showed that compounds always contain exactly the same proportion of elements by mass– Law of constant composition ...
Abundance of the chemical elements

The abundance of a chemical element measures how common is the element relative to all other elements in a given environment. Abundance is measured in one of three ways: by the mass-fraction (the same as weight fraction); by the mole-fraction (fraction of atoms by numerical count, or sometimes fraction of molecules in gases); or by the volume-fraction. Volume-fraction is a common abundance measure in mixed gases such as planetary atmospheres, and is similar in value to molecular mole-fraction for gas mixtures at relatively low densities and pressures, and ideal gas mixtures. Most abundance values in this article are given as mass-fractions. For example, the abundance of oxygen in pure water can be measured in two ways: the mass fraction is about 89%, because that is the fraction of water's mass which is oxygen. However, the mole-fraction is 33.3333...% because only 1 atom of 3 in water, H2O, is oxygen. As another example, looking at the mass-fraction abundance of hydrogen and helium in both the Universe as a whole and in the atmospheres of gas-giant planets such as Jupiter, it is 74% for hydrogen and 23-25% for helium; while the (atomic) mole-fraction for hydrogen is 92%, and for helium is 8%, in these environments. Changing the given environment to Jupiter's outer atmosphere, where hydrogen is diatomic while helium is not, changes the molecular mole-fraction (fraction of total gas molecules), as well as the fraction of atmosphere by volume, of hydrogen to about 86%, and of helium to 13%.