Periodic Table of Elements
... 1. It was not valid for elements that had atomic masses higher than Ca. 2. It was assumed by Newlands that only 56 elements existed in nature and no more elements would be discovered in the future. But, later on, several new elements were discovered, whose properties did not fit into the Law of Octa ...
... 1. It was not valid for elements that had atomic masses higher than Ca. 2. It was assumed by Newlands that only 56 elements existed in nature and no more elements would be discovered in the future. But, later on, several new elements were discovered, whose properties did not fit into the Law of Octa ...
Periodicity of Elements and Periodic Table CHAPTER – 4
... Properties of Group I 1. They are mono atomic. 2. They exist in solid metallic state. 3. Outer most shell of these elements is incomplete having one electron. 4. Elements of this group are highly reactive. 5. Elements of this group have large tendency to form compounds. 6. Elements of this group are ...
... Properties of Group I 1. They are mono atomic. 2. They exist in solid metallic state. 3. Outer most shell of these elements is incomplete having one electron. 4. Elements of this group are highly reactive. 5. Elements of this group have large tendency to form compounds. 6. Elements of this group are ...
the pdf of this lesson!
... sodium alginate. That is why it comes out of solution. It starts to clump. As it does this, it traps water from the solution. The Calcium Connection You may have noticed that the wormy things skin turns blue. That is because calcium chloride itself is slightly basic, or alkaline. As Algy hits the Ca ...
... sodium alginate. That is why it comes out of solution. It starts to clump. As it does this, it traps water from the solution. The Calcium Connection You may have noticed that the wormy things skin turns blue. That is because calcium chloride itself is slightly basic, or alkaline. As Algy hits the Ca ...
chem - CBSE Guess
... Ans. Element B with atomic number 12 and electronic configuration 2,8,2 represents metal. Q.Define the term Ore, Mineral. 1 Ans.The elementary state or the compounds of metal occur in nature along with impurities are known as minerals. The minerals from which metals can be extracted conveniently and ...
... Ans. Element B with atomic number 12 and electronic configuration 2,8,2 represents metal. Q.Define the term Ore, Mineral. 1 Ans.The elementary state or the compounds of metal occur in nature along with impurities are known as minerals. The minerals from which metals can be extracted conveniently and ...
chemical-peiodicity
... period. because there are more protons and electrons, which create greater attraction. b. How does the size of the atom vary as you move down the periodic table? Why? The size increase, because increasing number of electron shells. where the inner shells reduce the attractive forces of the nucleus a ...
... period. because there are more protons and electrons, which create greater attraction. b. How does the size of the atom vary as you move down the periodic table? Why? The size increase, because increasing number of electron shells. where the inner shells reduce the attractive forces of the nucleus a ...
Periodic classificatiion of elements
... A. R is used to represent any of the elements in the group. 46. Why are inert gases discovered late? A. Inert gases are discovered very late because they are very inert and present in extremely low concentrations in our atmosphere. 47. Why is no fixed position be given to hydrogen in the periodic ta ...
... A. R is used to represent any of the elements in the group. 46. Why are inert gases discovered late? A. Inert gases are discovered very late because they are very inert and present in extremely low concentrations in our atmosphere. 47. Why is no fixed position be given to hydrogen in the periodic ta ...
Honors Chemistry Worksheet on Periodic Table
... someday be discovered. How was he able to do this? There was an opening or blank spot left in gallium’s position on the table he had constructed. He recognized that no known element had the properties needed to fit into the open “slot”, hence he believed it had yet to be discovered. 26. After Mendel ...
... someday be discovered. How was he able to do this? There was an opening or blank spot left in gallium’s position on the table he had constructed. He recognized that no known element had the properties needed to fit into the open “slot”, hence he believed it had yet to be discovered. 26. After Mendel ...
Periodic Table Packet
... b. Lose electrons from two outermost energy levels c. Ions form colored solutions Comparing Metals and Nonmetals 8. Metals a. Chemical properties - tend to lose electrons easily " have low ionization energy (energy needed to remove electrons) i. have low electron affinity (attraction for electrons) ...
... b. Lose electrons from two outermost energy levels c. Ions form colored solutions Comparing Metals and Nonmetals 8. Metals a. Chemical properties - tend to lose electrons easily " have low ionization energy (energy needed to remove electrons) i. have low electron affinity (attraction for electrons) ...
Section 1 How Are Elements Organized
... • The transition metals constitute Groups 3 through 12 and are sometimes called the d-block elements because of their position in the periodic table. • A transition metal is one of the metals that can use the inner shell before using the outer shell to bond. • A transition metal may lose one, two, o ...
... • The transition metals constitute Groups 3 through 12 and are sometimes called the d-block elements because of their position in the periodic table. • A transition metal is one of the metals that can use the inner shell before using the outer shell to bond. • A transition metal may lose one, two, o ...
Document
... • The periodic law states that when the elements are arranged by increasing atomic number, there is a periodic repetition of their chemical and physical properties. • The periodic table organizes the elements into periods (rows) and groups (columns); elements with similar properties are in the same ...
... • The periodic law states that when the elements are arranged by increasing atomic number, there is a periodic repetition of their chemical and physical properties. • The periodic table organizes the elements into periods (rows) and groups (columns); elements with similar properties are in the same ...
File
... Example: Barium forms the basic oxide BaO: Reacts with water: BaO (s) + H2O (ℓ) Ba2+ (aq) + 2 OH– (aq) Reacts with acid: BaO (s) + H+ (aq) Ba2+ (aq) + H2O (ℓ) Other examples of basic oxides: ...
... Example: Barium forms the basic oxide BaO: Reacts with water: BaO (s) + H2O (ℓ) Ba2+ (aq) + 2 OH– (aq) Reacts with acid: BaO (s) + H+ (aq) Ba2+ (aq) + H2O (ℓ) Other examples of basic oxides: ...
The Modern Periodic Table (cont.)
... • Metals are elements that are generally shiny when smooth and clean, solid at room temperature, and good conductors of heat and electricity. • Alkali metals are all the elements in group 1 except hydrogen, and are very reactive. • Alkaline earth metals are in group 2, and are also highly reactive. ...
... • Metals are elements that are generally shiny when smooth and clean, solid at room temperature, and good conductors of heat and electricity. • Alkali metals are all the elements in group 1 except hydrogen, and are very reactive. • Alkaline earth metals are in group 2, and are also highly reactive. ...
The Periodic Table and Chemical Properties
... Chemical families or groups are arranged in vertical columns in the periodic table. Elements in the same chemical family have similar physical and chemical properties. The families are in numbered columns 1 to 18 of the table. Four well-known groups are the alkali metals, the alkaline earth metals, ...
... Chemical families or groups are arranged in vertical columns in the periodic table. Elements in the same chemical family have similar physical and chemical properties. The families are in numbered columns 1 to 18 of the table. Four well-known groups are the alkali metals, the alkaline earth metals, ...
Atomic structure and periodic table
... A compound is a combination of two or more elements in fixed proportions. The ratio of the atoms making a compound is called the chemical formulae. Elements combine together to form a compound depending on their combining power. The combining power of atoms in an element is called Valency.Valency of ...
... A compound is a combination of two or more elements in fixed proportions. The ratio of the atoms making a compound is called the chemical formulae. Elements combine together to form a compound depending on their combining power. The combining power of atoms in an element is called Valency.Valency of ...
GROUP 13 ELEMENTS -THE BORON FAMILY -
... Gallium has the chemical symbol Ga and atomic number 31. It has the electron configuration [Ar]2s2 2p1 and +3 oxidation state. The melting point is 29.8º C and therefore melts by increasing room temperature by a little. Gallium is important because it forms gallium arsenide (GaAs), which can convert ...
... Gallium has the chemical symbol Ga and atomic number 31. It has the electron configuration [Ar]2s2 2p1 and +3 oxidation state. The melting point is 29.8º C and therefore melts by increasing room temperature by a little. Gallium is important because it forms gallium arsenide (GaAs), which can convert ...
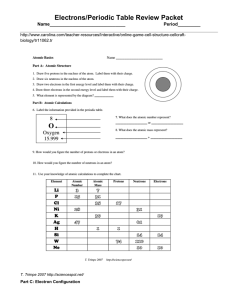
Electrons/Periodic Table Review Packet Name______________________________ Period_________
... 12. How many electrons can each level hold? 1st = _____ 2nd = _____ 3rd = _____ 13. What term is used for the electrons in the outermost shell or energy level? ____________________ 14. Scientists use two types of diagrams to show the electron configuration for atoms. What are they? 15. Calculate the ...
... 12. How many electrons can each level hold? 1st = _____ 2nd = _____ 3rd = _____ 13. What term is used for the electrons in the outermost shell or energy level? ____________________ 14. Scientists use two types of diagrams to show the electron configuration for atoms. What are they? 15. Calculate the ...
What Are Compounds?
... and the anion from an acid is often referred to as a salt. – examples: • Table salt, NaCl, contains the anion from hydrochloric acid, HCl. • Calcium sulfate, CaSO4, is a salt containing the anion from sulfuric acid, H2SO4. ...
... and the anion from an acid is often referred to as a salt. – examples: • Table salt, NaCl, contains the anion from hydrochloric acid, HCl. • Calcium sulfate, CaSO4, is a salt containing the anion from sulfuric acid, H2SO4. ...
Instructional-Objectives
... State the basic postulates of Bohr's theory. Describe the first model, the Bohr model of arrangement of electrons in shells (orbits) around a nucleus of an atom. Describe how Bohr models explain absorption and emission of light from atoms. Compare and contrast Bohr's theory and the more soph ...
... State the basic postulates of Bohr's theory. Describe the first model, the Bohr model of arrangement of electrons in shells (orbits) around a nucleus of an atom. Describe how Bohr models explain absorption and emission of light from atoms. Compare and contrast Bohr's theory and the more soph ...
Instructional Objectives 3. Atomic Structure and the Periodic Table
... • State the basic postulates of Bohr's theory. • Describe the first model, the Bohr model of arrangement of electrons in shells (orbits) around a nucleus of an atom. • Describe how Bohr models explain absorption and emission of light from atoms. • Compare and contrast Bohr's theory and the more soph ...
... • State the basic postulates of Bohr's theory. • Describe the first model, the Bohr model of arrangement of electrons in shells (orbits) around a nucleus of an atom. • Describe how Bohr models explain absorption and emission of light from atoms. • Compare and contrast Bohr's theory and the more soph ...
IT`S ATOMIC
... this zigzag line are considered to be metals. Elements that are to the right of the zigzag line are considered to be nonmetals. Metals and nonmetals have very different properties. As a result, metals and nonmetals will combine to form Group Figure 1 new substances. In addition to the zigzag line, t ...
... this zigzag line are considered to be metals. Elements that are to the right of the zigzag line are considered to be nonmetals. Metals and nonmetals have very different properties. As a result, metals and nonmetals will combine to form Group Figure 1 new substances. In addition to the zigzag line, t ...
Atomic Structure - RHS Encore Academy
... 16. What are metalloids? (page 136) 17. What major change occurs as you move from left to right across the periodic table? (page 138) 18. What is a valence electron? Draw a SQUARE around each valence electron in the picture! (page 139) ...
... 16. What are metalloids? (page 136) 17. What major change occurs as you move from left to right across the periodic table? (page 138) 18. What is a valence electron? Draw a SQUARE around each valence electron in the picture! (page 139) ...
Moles and Equations
... eg 2. Iron reacts with oxygen to make iron (III) oxide. Calculate the mass of iron and the volume of oxygen required to produce 3 grammes of iron (III) oxide. (Ar values are Fe: 56 O:16) ...
... eg 2. Iron reacts with oxygen to make iron (III) oxide. Calculate the mass of iron and the volume of oxygen required to produce 3 grammes of iron (III) oxide. (Ar values are Fe: 56 O:16) ...
Periodic Table Review
... They have 8 valence electrons They have a full outermost electron shell ...
... They have 8 valence electrons They have a full outermost electron shell ...
Periodic Trends
... Reactivity increases as you go down the columns of metallic elements. Reactivity decreases as you go down the columns of non-metallic elements. Watch the video to see what that means. ...
... Reactivity increases as you go down the columns of metallic elements. Reactivity decreases as you go down the columns of non-metallic elements. Watch the video to see what that means. ...