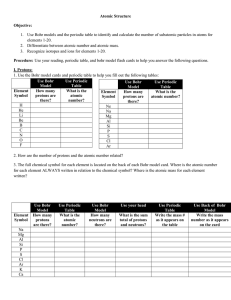
How many protons, electrons and neutrons are in an atom of krypton
... charged electrons. Atoms must have equal numbers of protons and electrons. In our example, an atom of krypton must contain 36 electrons since it contains 36 protons. Electrons are arranged around atoms in a special way. If you need to know how the electrons are arranged around an atom, take a look ...
... charged electrons. Atoms must have equal numbers of protons and electrons. In our example, an atom of krypton must contain 36 electrons since it contains 36 protons. Electrons are arranged around atoms in a special way. If you need to know how the electrons are arranged around an atom, take a look ...
Atomic Structure PowerPoint Presentation
... the average mass of all the isotopes found in nature. No element exists with only one possible isotope. Hydrogen has the smallest number of isotopes: 1H protium, 2H deuterium, 3H tritium. Its atomic mass is 1.0079 amu (atomic mass units). The atomic mass is calculated by adding the % of 1H mass foun ...
... the average mass of all the isotopes found in nature. No element exists with only one possible isotope. Hydrogen has the smallest number of isotopes: 1H protium, 2H deuterium, 3H tritium. Its atomic mass is 1.0079 amu (atomic mass units). The atomic mass is calculated by adding the % of 1H mass foun ...
Chapter 10 - MrsDoughertys
... Atoms are made up of protons, neutrons and electrons. Protons and neutrons have an electric charge. Electrons and protons are found in the nucleus of an atom. If you change the number of neutrons in the center of an atom, you change the type of ...
... Atoms are made up of protons, neutrons and electrons. Protons and neutrons have an electric charge. Electrons and protons are found in the nucleus of an atom. If you change the number of neutrons in the center of an atom, you change the type of ...
Symbols of Elements - Chemistry with Mr. Patmos
... Are tiny particles of matter. of an element are similar and different from other elements. of two or more different elements combine in whole number ratios to form compounds. are separated, joined, or rearranged in a chemical reaction. ...
... Are tiny particles of matter. of an element are similar and different from other elements. of two or more different elements combine in whole number ratios to form compounds. are separated, joined, or rearranged in a chemical reaction. ...
Elements of Chemical Structure and Inorganic Nomenclature
... wall may be constructed from a basic unit, the brick. In trying to find this basic unit, they separated matter by all the methods (chemical and physical) available to them until they could not separate it any further. They felt this separation must result in the building block of matter, which they ...
... wall may be constructed from a basic unit, the brick. In trying to find this basic unit, they separated matter by all the methods (chemical and physical) available to them until they could not separate it any further. They felt this separation must result in the building block of matter, which they ...
Balancing Reaction Equations Oxidation State Reduction
... Oxidation: Loss of electrons from an element. Oxidation number increases Reduction: Gain of electrons by an element. Oxidation number decreases ...
... Oxidation: Loss of electrons from an element. Oxidation number increases Reduction: Gain of electrons by an element. Oxidation number decreases ...
Chapter 4 Atomic Structure
... – A carbon atom has 6 protons in its nucleus – A neutral carbon atom has 6 electrons • A carbon atom also contains neutrons, but unlike electrons and protons the number of neutrons can change from carbon atom to carbon atom • Some carbon atoms have 6 neutrons, others have 7 neutrons and still others ...
... – A carbon atom has 6 protons in its nucleus – A neutral carbon atom has 6 electrons • A carbon atom also contains neutrons, but unlike electrons and protons the number of neutrons can change from carbon atom to carbon atom • Some carbon atoms have 6 neutrons, others have 7 neutrons and still others ...
04_Lecture Atoms and Elements
... • When an atom gains or loses electrons, it becomes an ion. • Positively charged ions are called cations. • Negatively charged ions are called anions. • Cations and anions occur together so that matter is chargeneutral. Isotopes: • Atoms of the same element with different numbers of neutrons are cal ...
... • When an atom gains or loses electrons, it becomes an ion. • Positively charged ions are called cations. • Negatively charged ions are called anions. • Cations and anions occur together so that matter is chargeneutral. Isotopes: • Atoms of the same element with different numbers of neutrons are cal ...
04_Lecture Atoms and Elements
... • When an atom gains or loses electrons, it becomes an ion. • Positively charged ions are called cations. • Negatively charged ions are called anions. • Cations and anions occur together so that matter is chargeneutral. Isotopes: • Atoms of the same element with different numbers of neutrons are cal ...
... • When an atom gains or loses electrons, it becomes an ion. • Positively charged ions are called cations. • Negatively charged ions are called anions. • Cations and anions occur together so that matter is chargeneutral. Isotopes: • Atoms of the same element with different numbers of neutrons are cal ...
U1 Atoms, Periodic Table, Variables, Conversions Unit 1
... 17. A certain atom has 20 electrons, 21 neutrons, and 20 protons. What is the atomic mass of the ...
... 17. A certain atom has 20 electrons, 21 neutrons, and 20 protons. What is the atomic mass of the ...
ch14 lecture 7e
... Zeff increases for the larger 3A elements due to poor shielding by d and f electrons. The larger 3A elements have smaller atomic radii and larger ionization energies and electronegativities than expected. These properties influence the physical and chemical behavior of these elements. ...
... Zeff increases for the larger 3A elements due to poor shielding by d and f electrons. The larger 3A elements have smaller atomic radii and larger ionization energies and electronegativities than expected. These properties influence the physical and chemical behavior of these elements. ...
4.1 Experiencing Atoms at Tiburon 4.1 Experiencing Atoms
... • The nuclei of some isotopes of a given element are not stable. • These atoms emit a few energetic subatomic particles from their nuclei and change into different isotopes of different elements. • The emitted subatomic particles are called nuclear radiation. • The isotopes that emit them are te ...
... • The nuclei of some isotopes of a given element are not stable. • These atoms emit a few energetic subatomic particles from their nuclei and change into different isotopes of different elements. • The emitted subatomic particles are called nuclear radiation. • The isotopes that emit them are te ...
Chapter 4 Atoms and Elements
... • When an atom gains or loses electrons, it becomes an ion. • Positively charged ions are called cations. • Negatively charged ions are called anions. • Cations and anions occur together so that matter is chargeneutral. Isotopes: • Atoms of the same element with different numbers of neutrons are cal ...
... • When an atom gains or loses electrons, it becomes an ion. • Positively charged ions are called cations. • Negatively charged ions are called anions. • Cations and anions occur together so that matter is chargeneutral. Isotopes: • Atoms of the same element with different numbers of neutrons are cal ...
the Language of Chemistry
... He was the first person to make the distinction between organic and inorganic compounds. He introduced the classical system of chemical symbols in 1811, in which elements are abbreviated by one or two letters to make a distinct abbreviation from their Latin names. He developed the radical theory of ...
... He was the first person to make the distinction between organic and inorganic compounds. He introduced the classical system of chemical symbols in 1811, in which elements are abbreviated by one or two letters to make a distinct abbreviation from their Latin names. He developed the radical theory of ...
Chapter #4 Section Assessment #1 - 33
... *Elements with high electronegativies and ionization energies tend to be clustered in the upper right hand corner of the periodic table. [Don’t burden your brain too much with these examples just yet. We’ll be looking at them more closely in chapter 6.] ...
... *Elements with high electronegativies and ionization energies tend to be clustered in the upper right hand corner of the periodic table. [Don’t burden your brain too much with these examples just yet. We’ll be looking at them more closely in chapter 6.] ...
Make a large atom with p:95, n:146, e:95 - TSDCurriculum
... 5. READ: This simulation only lets you to build atoms that exist in nature or have been made by scientists. If you can’t build it, it can't be made in the real world. Scientists use the word isotope to distinguish between atoms that have the same number of protons but different numbers of neutrons. ...
... 5. READ: This simulation only lets you to build atoms that exist in nature or have been made by scientists. If you can’t build it, it can't be made in the real world. Scientists use the word isotope to distinguish between atoms that have the same number of protons but different numbers of neutrons. ...
George Mason University General Chemistry 211 Chapter 2 The
... (compounds) physically intermingled, but not chemically combined ...
... (compounds) physically intermingled, but not chemically combined ...
Matter and Measurement
... Most metals react with non-metals to give ionic compounds. The metals are oxidized and the non-metals reduced. Example: 2 Na(s) + Cl2(g) --> 2 NaCl(s) When the metal can form ions of different charges, different products are possible depending on the experimental ...
... Most metals react with non-metals to give ionic compounds. The metals are oxidized and the non-metals reduced. Example: 2 Na(s) + Cl2(g) --> 2 NaCl(s) When the metal can form ions of different charges, different products are possible depending on the experimental ...
Atoms, Elements, and Compounds
... could spontaneously decay into another element by expelling a piece of the atom. This was surprising to the scientific community—the idea that atoms could change into other atoms had been scorned as alchemy. ...
... could spontaneously decay into another element by expelling a piece of the atom. This was surprising to the scientific community—the idea that atoms could change into other atoms had been scorned as alchemy. ...
Name
... • Isotopes are atoms of the same element that have different masses. • The isotopes of a particular element all have the same number of protons and electrons but different numbers of neutrons. • Most of the elements consist of mixtures of isotopes. Mass Number • The mass number is the total number o ...
... • Isotopes are atoms of the same element that have different masses. • The isotopes of a particular element all have the same number of protons and electrons but different numbers of neutrons. • Most of the elements consist of mixtures of isotopes. Mass Number • The mass number is the total number o ...
Atomic Structure_Bohr Flashcards
... 8. a. Compare the number of protons and neutrons in H-1 to H-2 to H-3. List any similarities and differences. b. Compare the number of protons and neutrons in C-12 to C-13 to C-14. List any similarities and differences. ...
... 8. a. Compare the number of protons and neutrons in H-1 to H-2 to H-3. List any similarities and differences. b. Compare the number of protons and neutrons in C-12 to C-13 to C-14. List any similarities and differences. ...
Atoms, Isotopes and Relative Atomic Masses
... The Group 7 element bromine was discovered by Balard in 1826. Bromine gets its name from the Greek bromos meaning stench. Bromine consists of a mixture of two isotopes, 79Br and 81Br. ...
... The Group 7 element bromine was discovered by Balard in 1826. Bromine gets its name from the Greek bromos meaning stench. Bromine consists of a mixture of two isotopes, 79Br and 81Br. ...
Electrochemistry Oxidation – Reduction and Oxidation Numbers
... Rules for assigning oxidation numbers: 1. Elements in their most abundant naturally occurring form are assigned an oxidation number of zero. e.g. Na, Fe, Cl2, O2 2. The sum of the oxidation numbers for a compound or formula unit is zero. 3. For a polyatomic ion, the oxidation numbers of the constit ...
... Rules for assigning oxidation numbers: 1. Elements in their most abundant naturally occurring form are assigned an oxidation number of zero. e.g. Na, Fe, Cl2, O2 2. The sum of the oxidation numbers for a compound or formula unit is zero. 3. For a polyatomic ion, the oxidation numbers of the constit ...
14.1 Structure of the Atom
... Sometimes the mass number for an element is included in its symbol. When the symbol is written in this way, we call it isotope notation. The isotope notation for carbon-12 is shown to the right. How many neutrons does an atom of carbon-12 have? To find out, simply take the mass number and subtract t ...
... Sometimes the mass number for an element is included in its symbol. When the symbol is written in this way, we call it isotope notation. The isotope notation for carbon-12 is shown to the right. How many neutrons does an atom of carbon-12 have? To find out, simply take the mass number and subtract t ...
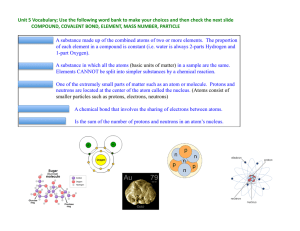
Review Unit 5
... CHEMICALLY STABLE: Elements that are nonreactive because their last electron shell is completely filled with 8 electrons. (e.g. Neon, Argon, Krypton.) ISOTOPE: ...
... CHEMICALLY STABLE: Elements that are nonreactive because their last electron shell is completely filled with 8 electrons. (e.g. Neon, Argon, Krypton.) ISOTOPE: ...