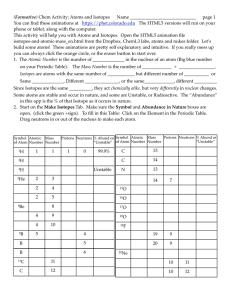
wahideh chemistry eportfolio hw
... Sodium is a member of the alkali metals family. The alkali family consists of elements in Group 1 (IA) of the periodic table. The periodic table is a chart that shows how chemical elements are related to one another. Other Group 1 (IA) elements are lithium, potassium, rubidium, cesium, and francium. ...
... Sodium is a member of the alkali metals family. The alkali family consists of elements in Group 1 (IA) of the periodic table. The periodic table is a chart that shows how chemical elements are related to one another. Other Group 1 (IA) elements are lithium, potassium, rubidium, cesium, and francium. ...
Section 2 Oxidation Numbers
... • In general when assigning oxidation numbers, shared electrons are assumed to “belong” to the more electronegative atom in each bond. • More-specific rules are provided by the following guidelines. ...
... • In general when assigning oxidation numbers, shared electrons are assumed to “belong” to the more electronegative atom in each bond. • More-specific rules are provided by the following guidelines. ...
Slides Chapter 2 File
... • The Greek symbol indicates summing of terms. © 2013 Pearson Education, Inc. ...
... • The Greek symbol indicates summing of terms. © 2013 Pearson Education, Inc. ...
Powerpoint slides
... • The Greek symbol indicates summing of terms. © 2013 Pearson Education, Inc. ...
... • The Greek symbol indicates summing of terms. © 2013 Pearson Education, Inc. ...
Chapter 4
... ____ 50. Which of the following equals one atomic mass unit? a. the mass of one electron b. the mass of one helium-4 atom c. the mass of one carbon-12 atom d. one-twelfth the mass of one carbon-12 atom ____ 51. Which of the following statements is NOT true? a. Protons have a positive charge. b. Elec ...
... ____ 50. Which of the following equals one atomic mass unit? a. the mass of one electron b. the mass of one helium-4 atom c. the mass of one carbon-12 atom d. one-twelfth the mass of one carbon-12 atom ____ 51. Which of the following statements is NOT true? a. Protons have a positive charge. b. Elec ...
Chapter 4 Early Atomic Theory
... To understand why the periodic table has this set up we must understand the structure of the atom. The original idea of the atom came from Ancient Greece (400 BC). The Greek philosophers (lovers of knowledge) tried to make sense of their world by observing their surroundings. Greek society was slave ...
... To understand why the periodic table has this set up we must understand the structure of the atom. The original idea of the atom came from Ancient Greece (400 BC). The Greek philosophers (lovers of knowledge) tried to make sense of their world by observing their surroundings. Greek society was slave ...
Atom 3 Isotopes - Solon City Schools
... Atoms with the same number of protons & electrons but a different number of neutrons. They are the same element, are chemically identical and undergo the exact same chemical reactions They have different masses (different mass number). All isotopes are used to calculate average atomic mass (this mas ...
... Atoms with the same number of protons & electrons but a different number of neutrons. They are the same element, are chemically identical and undergo the exact same chemical reactions They have different masses (different mass number). All isotopes are used to calculate average atomic mass (this mas ...
Once scientists concluded that all matter contains negatively
... After a hypothesis has been formed, scientists conduct experiments to test its validity. Experiments are systematic observations or measurements, preferably made under controlled conditions—that is, under conditions in which a single variable changes. For example, in our extinction scenario, iridiu ...
... After a hypothesis has been formed, scientists conduct experiments to test its validity. Experiments are systematic observations or measurements, preferably made under controlled conditions—that is, under conditions in which a single variable changes. For example, in our extinction scenario, iridiu ...
1.1 - cloudfront.net
... If we were to place a sample of carbon into a mass spectrometer and analyze its mass, we would find that some of the carbon atoms have a relative mass of 12, while other atoms have a relative mass of 13, and still others have a relative mass of 14. The mass spectrometer measures the percent abundanc ...
... If we were to place a sample of carbon into a mass spectrometer and analyze its mass, we would find that some of the carbon atoms have a relative mass of 12, while other atoms have a relative mass of 13, and still others have a relative mass of 14. The mass spectrometer measures the percent abundanc ...
Atoms and nukes packet 2016
... open. (click the green +sign). To fill in this Table: Click on the Element in the Periodic Table. Drag neutrons in or out of the nucleus to make each atom. ...
... open. (click the green +sign). To fill in this Table: Click on the Element in the Periodic Table. Drag neutrons in or out of the nucleus to make each atom. ...
Redox
... (rusting, tarnishing, etc.) and burning is an even more recent development. Reactions of substances with oxygen, whether they were the explosive combustion of gunpowder, the burning of wood, or the slow corrosion of iron came to be called oxidation. It soon became apparent that oxygen was not the on ...
... (rusting, tarnishing, etc.) and burning is an even more recent development. Reactions of substances with oxygen, whether they were the explosive combustion of gunpowder, the burning of wood, or the slow corrosion of iron came to be called oxidation. It soon became apparent that oxygen was not the on ...
Scandium and Yttrium - Mercyhurst University
... reaction.4 At room temperature, scandium metal turns a light yellow or pink color in air.1 It is not obvious what is causing the color as Sc3+ would be expected to be colorless (white) due to the lack of d-electrons. Purification and Properties The most interesting thing about these two elements is ...
... reaction.4 At room temperature, scandium metal turns a light yellow or pink color in air.1 It is not obvious what is causing the color as Sc3+ would be expected to be colorless (white) due to the lack of d-electrons. Purification and Properties The most interesting thing about these two elements is ...
Chapter 04s
... Dalton’s Law of Atomic Theory 1. All matter is composed of extremely small particles called atoms. 2. Atoms of a given element are identical in their physical and chemical properties, while atoms of different elements differ in their physical and chemical properties. http://www.kjemi.uio.no/softwar ...
... Dalton’s Law of Atomic Theory 1. All matter is composed of extremely small particles called atoms. 2. Atoms of a given element are identical in their physical and chemical properties, while atoms of different elements differ in their physical and chemical properties. http://www.kjemi.uio.no/softwar ...
Answers to Chapter Diagnostic Test
... fixed proportions (2.1) chemical reaction rearrangement of atoms present in reacting substances to give new chemical combinations present in the substances formed (2.1) atomic symbol element (2.1) ...
... fixed proportions (2.1) chemical reaction rearrangement of atoms present in reacting substances to give new chemical combinations present in the substances formed (2.1) atomic symbol element (2.1) ...
105
... I The combustion of ammonia in oxygen to form nitrogen dioxide and water vapour involves covalent molecules in the gas phase. The oxidation number method for balancing the equation was shown in an example in this section. Devise a half-reaction method for balancing the equation. Describe the assumpt ...
... I The combustion of ammonia in oxygen to form nitrogen dioxide and water vapour involves covalent molecules in the gas phase. The oxidation number method for balancing the equation was shown in an example in this section. Devise a half-reaction method for balancing the equation. Describe the assumpt ...
Redox I
... Mg got oxidized. Fe2+ was the oxidizing agent. •Fe goes from an ion to an element: Fe2+ Fe Fe2+ got reduced. Mg was the reducing agent. ...
... Mg got oxidized. Fe2+ was the oxidizing agent. •Fe goes from an ion to an element: Fe2+ Fe Fe2+ got reduced. Mg was the reducing agent. ...
Redox
... (Cl-) and chlorate(V) ions (ClO3-). Deduce the two half-equations for this reaction, and hence derive an overall equation. Chlorine is changing its oxidation state from 0 in Cl2 to -1 in Cl- and therefore needs to gain 1e-. ½ Cl2 + eClChlorine is changing its oxidation state from 0 in Cl2 to +5 in C ...
... (Cl-) and chlorate(V) ions (ClO3-). Deduce the two half-equations for this reaction, and hence derive an overall equation. Chlorine is changing its oxidation state from 0 in Cl2 to -1 in Cl- and therefore needs to gain 1e-. ½ Cl2 + eClChlorine is changing its oxidation state from 0 in Cl2 to +5 in C ...
Jeopardy - SchoolRack
... Use the CRT Thomson determined that there the cathode rays were negative but he knew that not all matter was negative so then he determined that there must be a positive charge. ...
... Use the CRT Thomson determined that there the cathode rays were negative but he knew that not all matter was negative so then he determined that there must be a positive charge. ...
PowerPoint
... 1- How many protons, neutrons, and electrons are in an atom of chlorine-37? Chlorine has an atomic number of 17. Protons = 17 (atomic number) Electron = 17 (atomic number) Neutrons = 20 (neutrons = mass number (37)—atomic number (17) 2- How many protons, neutrons, and electrons are in an atom of Br ...
... 1- How many protons, neutrons, and electrons are in an atom of chlorine-37? Chlorine has an atomic number of 17. Protons = 17 (atomic number) Electron = 17 (atomic number) Neutrons = 20 (neutrons = mass number (37)—atomic number (17) 2- How many protons, neutrons, and electrons are in an atom of Br ...
Period:______ Table Number
... 56. On the periodic table of elements those elements which have an atomic number of ____ (Hydrogen) to ________ (Uranium) are naturally occuring elements which can be found to exist somewhere in the earth’s land, water, or air. 57. On the periodic table of elements those elements which have an atomi ...
... 56. On the periodic table of elements those elements which have an atomic number of ____ (Hydrogen) to ________ (Uranium) are naturally occuring elements which can be found to exist somewhere in the earth’s land, water, or air. 57. On the periodic table of elements those elements which have an atomi ...
Name
... 56. On the periodic table of elements those elements which have an atomic number of ______(Hydrogen) to ___________(Uranium) are naturally occuring elements which can be found to exist somewhere in the earth’s land, water, or air. 57. On the periodic table of elements those elements which have an at ...
... 56. On the periodic table of elements those elements which have an atomic number of ______(Hydrogen) to ___________(Uranium) are naturally occuring elements which can be found to exist somewhere in the earth’s land, water, or air. 57. On the periodic table of elements those elements which have an at ...
Annotation - Origin of the Elements - Student
... Most astronomers today theorize that the Universe as we know it started from a massive “explosion” called the Big Bang. Evidence leading to this unusual theory was first discovered in 1929, when Dr. Edwin Hubble had made a startling announcement that he had found that all of the distant galaxies in ...
... Most astronomers today theorize that the Universe as we know it started from a massive “explosion” called the Big Bang. Evidence leading to this unusual theory was first discovered in 1929, when Dr. Edwin Hubble had made a startling announcement that he had found that all of the distant galaxies in ...
Chapter 4 Atoms and Elements
... • The properties of atoms determine the properties of matter. • An atom is the smallest identifiable unit of an element. • An element is a substance that cannot be broken down into simpler substances. • There are about 91 different elements in nature, and consequently about 91 different kinds of ato ...
... • The properties of atoms determine the properties of matter. • An atom is the smallest identifiable unit of an element. • An element is a substance that cannot be broken down into simpler substances. • There are about 91 different elements in nature, and consequently about 91 different kinds of ato ...
Period:______ Table Number
... 83. The number and arrangement of the electrons found in the electron cloud of an atom determines nearly all of an element’s CHEMICAL properties. P. 125, VCR: Atoms and Molecules 84. The total number of electrons found in the electron cloud of an atom adds very little to the mass of an atom since it ...
... 83. The number and arrangement of the electrons found in the electron cloud of an atom determines nearly all of an element’s CHEMICAL properties. P. 125, VCR: Atoms and Molecules 84. The total number of electrons found in the electron cloud of an atom adds very little to the mass of an atom since it ...