Unit 3 Notes, Practice, and Review
... 6. Because neutral atoms contain the same number of negative elections and positive protons, so they cancel out. 7. The number of protons in that atom. 8. a. 15, b. 42, c. 13, d. 48, e. 24, f. 82 ...
... 6. Because neutral atoms contain the same number of negative elections and positive protons, so they cancel out. 7. The number of protons in that atom. 8. a. 15, b. 42, c. 13, d. 48, e. 24, f. 82 ...
A2 Module 2814: Chains, Rings and Spectroscopy
... For the elements up to Ca the 3d orbitals are higher in energy than the 4s orbital. Therefore, after argon (element 18), the 4s orbital is filled: Ca has electron configuration [Ar] 4s2. From scandium on, the 3d orbitals are filled, until they have ten electrons at zinc. The term “d-block elements” ...
... For the elements up to Ca the 3d orbitals are higher in energy than the 4s orbital. Therefore, after argon (element 18), the 4s orbital is filled: Ca has electron configuration [Ar] 4s2. From scandium on, the 3d orbitals are filled, until they have ten electrons at zinc. The term “d-block elements” ...
Students will review concepts from their quiz and then correct it at
... The atoms are ___chemically___ combined in some way. Often times (but not always) they come together to form groups of atoms called molecules. A compound is always homogeneous (uniform). Compounds ___cannot___ be separated by physical means. Separating a compound requires a chemical reaction. The pr ...
... The atoms are ___chemically___ combined in some way. Often times (but not always) they come together to form groups of atoms called molecules. A compound is always homogeneous (uniform). Compounds ___cannot___ be separated by physical means. Separating a compound requires a chemical reaction. The pr ...
Unit 2- The Atom
... Joseph Priestley (1733‐1804) discovered oxygen and found to support combustion. He also found carbon dioxide in a fermentation of grain ...
... Joseph Priestley (1733‐1804) discovered oxygen and found to support combustion. He also found carbon dioxide in a fermentation of grain ...
mack atoms - McClymonds Chemistry
... Looking for Patterns: The Periodic Law and the Periodic Table • Mendeleev’s periodic law was based on observation. • Like all scientific laws, the periodic law summarized many observations but did not give the underlying reason for the observation—only theories do that. • For now, we accept the per ...
... Looking for Patterns: The Periodic Law and the Periodic Table • Mendeleev’s periodic law was based on observation. • Like all scientific laws, the periodic law summarized many observations but did not give the underlying reason for the observation—only theories do that. • For now, we accept the per ...
Atomic Structure 3: ISOTOPES
... element have the same number of protons, as given by the atomic number. You also know that the number of electrons in an uncharged atom equals the number of protons. The number of neutrons in the nucleus of an atom is not so predictable. The periodic table helps us out, though, by giving the atomic ...
... element have the same number of protons, as given by the atomic number. You also know that the number of electrons in an uncharged atom equals the number of protons. The number of neutrons in the nucleus of an atom is not so predictable. The periodic table helps us out, though, by giving the atomic ...
atom`s - Hauppauge School District
... Modern Atomic or Wave Mechanical Model • These locations are based on where they are most likely found, ________________ • We call this arrangement an _____________________ • ______________ are a three dimensional representation of principal energy levels ...
... Modern Atomic or Wave Mechanical Model • These locations are based on where they are most likely found, ________________ • We call this arrangement an _____________________ • ______________ are a three dimensional representation of principal energy levels ...
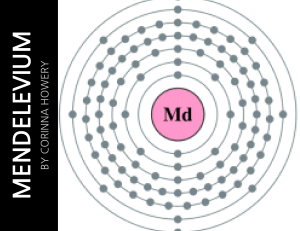
Mendelevium
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
Entry Task
... • Nonmetals– elements on the right of the periodic table – Properties are opposite of metals – Their properties vary more from element to element than the properties of metals – All are gas in their natural state at room temperature except for bromine which is a liquid – Poor conductors of heat and ...
... • Nonmetals– elements on the right of the periodic table – Properties are opposite of metals – Their properties vary more from element to element than the properties of metals – All are gas in their natural state at room temperature except for bromine which is a liquid – Poor conductors of heat and ...
OCR AS LEVEL CHEMISTRY A 1.1.1 ATOMS 1.2.1 ELECTRON
... State which two elements from the first twenty elements of the modern Periodic Table are not arranged in order of increasing atomic mass. ...
... State which two elements from the first twenty elements of the modern Periodic Table are not arranged in order of increasing atomic mass. ...
Atomic
... • Atomic # = # Protons = # Electrons • Because the number of positive charges equals the number of negative charges, the atom is neutral (no charge) ...
... • Atomic # = # Protons = # Electrons • Because the number of positive charges equals the number of negative charges, the atom is neutral (no charge) ...
File
... isotope. Atomic molar mass is given under the name for each element in the periodic table. b. The atomic molar mass of hydrogen is 1.01 g/mol. It is not exactly 1 because it is the average mass of the three naturally occurring isotopes of hydrogen on Earth. 22. The magnesium ion is a cation, since i ...
... isotope. Atomic molar mass is given under the name for each element in the periodic table. b. The atomic molar mass of hydrogen is 1.01 g/mol. It is not exactly 1 because it is the average mass of the three naturally occurring isotopes of hydrogen on Earth. 22. The magnesium ion is a cation, since i ...
Atoms and Elements: Are they Related?
... • There are 92 naturally occurring elements on the periodic table. This means they are in nature and not made in a laboratory by scientists. • As of this year (2011) there are 118 confirmed elements. There are 4 more under investigation and scientific testing. ...
... • There are 92 naturally occurring elements on the periodic table. This means they are in nature and not made in a laboratory by scientists. • As of this year (2011) there are 118 confirmed elements. There are 4 more under investigation and scientific testing. ...
Redox Flash Cards - No Brain Too Small
... The ox. number of The ox. number of oxygen (in hydrogen (in If its oxidation compounds/ions) compounds/ions) number increases, is __ , except in is __ , except in the element has ...
... The ox. number of The ox. number of oxygen (in hydrogen (in If its oxidation compounds/ions) compounds/ions) number increases, is __ , except in is __ , except in the element has ...
Atoms and Elements
... • Each element has a _________________________number of protons in its nucleus. All carbon atoms have 6 protons in their nuclei. The ______________________in the nucleus of an atom is called the atomic number. ________is the short-hand designation for the________________________. Because each ...
... • Each element has a _________________________number of protons in its nucleus. All carbon atoms have 6 protons in their nuclei. The ______________________in the nucleus of an atom is called the atomic number. ________is the short-hand designation for the________________________. Because each ...
Matching - hrsbstaff.ednet.ns.ca
... Identify the choice that best completes the statement or answers the question. ____ 11. Who was the man who lived from 460B.C.–370B.C. and was among the first to suggest the idea of atoms? a. Atomos c. Democritus b. Dalton d. Thomson ____ 12. The smallest particle of an element that retains the prop ...
... Identify the choice that best completes the statement or answers the question. ____ 11. Who was the man who lived from 460B.C.–370B.C. and was among the first to suggest the idea of atoms? a. Atomos c. Democritus b. Dalton d. Thomson ____ 12. The smallest particle of an element that retains the prop ...
Chapter 3 Notes - Scarsdale Schools
... powers. He placed iodine (I) after tellurium (Te), for example, because it had similar properties with chlorine (Cl) and bromine (Br) despite the fact that it had a smaller atomic mass. The key to Mendeleev’s Periodic Table, and the reason he is acknowledged as the discoverer of the Periodic Tab ...
... powers. He placed iodine (I) after tellurium (Te), for example, because it had similar properties with chlorine (Cl) and bromine (Br) despite the fact that it had a smaller atomic mass. The key to Mendeleev’s Periodic Table, and the reason he is acknowledged as the discoverer of the Periodic Tab ...
Unit 3-The Atom Chapter Packet
... ____________, that matter could not be created or destroyed. Then ___________ proposed, in his law of _________ _________, that the ratio of the masses of elements in any given compound is always the same. The law of ____________ ___________ , proposed soon after, states that the masses of one eleme ...
... ____________, that matter could not be created or destroyed. Then ___________ proposed, in his law of _________ _________, that the ratio of the masses of elements in any given compound is always the same. The law of ____________ ___________ , proposed soon after, states that the masses of one eleme ...
lecture CH2 chem121pikul
... Bryson in A Short History of Nearly Everything, carbon is described as “the party animal of the atomic world, latching on to many other atoms (including itself) and holding tight, forming molecular conga lines of hearty robustness—the very trick of nature necessary to build proteins and DNA.” As a r ...
... Bryson in A Short History of Nearly Everything, carbon is described as “the party animal of the atomic world, latching on to many other atoms (including itself) and holding tight, forming molecular conga lines of hearty robustness—the very trick of nature necessary to build proteins and DNA.” As a r ...
Build An Atom - ChemConnections
... b) An atom with 12 protons and 14 neutrons, 10 electrons: _____________ c) An atom with 14 protons and 13 neutrons, 14 electrons: _____________ ...
... b) An atom with 12 protons and 14 neutrons, 10 electrons: _____________ c) An atom with 14 protons and 13 neutrons, 14 electrons: _____________ ...
atom
... atom or chemically bonded group of atoms by adding or removing one or more electrons. A cation is a positively charged ion formed by losing one or more electrons. A cation is named by its element name followed by the word ion. For example, Na+ is the sodium ion, etc. Under the definition of anion ad ...
... atom or chemically bonded group of atoms by adding or removing one or more electrons. A cation is a positively charged ion formed by losing one or more electrons. A cation is named by its element name followed by the word ion. For example, Na+ is the sodium ion, etc. Under the definition of anion ad ...
lecture CH2 chem121pikul
... Nucleus and structure of atom Atomic number, Mass Number, Isotopes, Atomic Weight, & Atomic Mass Periodic Table: groups & periods: similar properties within groups Electron structure: valence electrons and electron dot symbols Periodic trends: atomic size and ionization energy ...
... Nucleus and structure of atom Atomic number, Mass Number, Isotopes, Atomic Weight, & Atomic Mass Periodic Table: groups & periods: similar properties within groups Electron structure: valence electrons and electron dot symbols Periodic trends: atomic size and ionization energy ...