CHAPTER 1 Practice Exercises 1.1 12.3 g Cd 1.3 26.9814 u 1.5
... An orbital is a region in space where there is a non-zero probability of finding an electron. ...
... An orbital is a region in space where there is a non-zero probability of finding an electron. ...
Chemistry Fall-2016 Final
... AA. any metal in Group 2A of the periodic table; generally harder, denser, stronger, and have higher melting points than alkali metals ...
... AA. any metal in Group 2A of the periodic table; generally harder, denser, stronger, and have higher melting points than alkali metals ...
The Periodic Table HL Page 1 of 3 G. Galvin Name: Periodic Table
... -list the numbers of electrons in each main energy level in atoms of numbers 1-20 -build up the electronic structure of the first 36 elements -derive the electronic configurations of ions of s- and p-block elements only -describe the arrangement of electrons in individual orbitals of p-block atom ...
... -list the numbers of electrons in each main energy level in atoms of numbers 1-20 -build up the electronic structure of the first 36 elements -derive the electronic configurations of ions of s- and p-block elements only -describe the arrangement of electrons in individual orbitals of p-block atom ...
Atomic Number
... -Elements within the same group have similar properties EX. Au, Ag, Cu -Each horizontal row is called a ____________________ -Properties of the elements gradually change when you move through a period -Elements get smaller when you move from _________________ to ______________. ...
... -Elements within the same group have similar properties EX. Au, Ag, Cu -Each horizontal row is called a ____________________ -Properties of the elements gradually change when you move through a period -Elements get smaller when you move from _________________ to ______________. ...
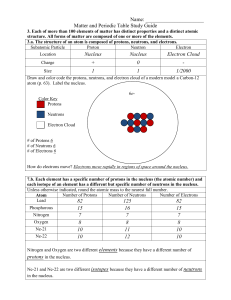
Matter and the Periodic Table Study Guide Answer Key
... 3.b. Compounds are formed by combining two or more different elements and compounds have properties that are different from their constituent elements. 3.f. Use the periodic table to identify elements in simple compounds. Compound List the number of each type of atom making up the compound NH4 1 Nit ...
... 3.b. Compounds are formed by combining two or more different elements and compounds have properties that are different from their constituent elements. 3.f. Use the periodic table to identify elements in simple compounds. Compound List the number of each type of atom making up the compound NH4 1 Nit ...
Structure-Prop of Matter session
... Carbon-14 and Carbon-13 atoms’ are not as stable as carbon-12 and easily break down. If an isotope has too many or too few neutrons compared to the number of protons, it is unstable and will undergo radioactive decay. These radioactive isotopes become different elements in an effort to become more s ...
... Carbon-14 and Carbon-13 atoms’ are not as stable as carbon-12 and easily break down. If an isotope has too many or too few neutrons compared to the number of protons, it is unstable and will undergo radioactive decay. These radioactive isotopes become different elements in an effort to become more s ...
Chapter 5 Review
... As a result of the discovery of the nucleus by Rutherford, what model described an atom? Describe the nucleus of an atom. Know the properties of the electron. Atoms have what charge, and how many protons and electrons? ...
... As a result of the discovery of the nucleus by Rutherford, what model described an atom? Describe the nucleus of an atom. Know the properties of the electron. Atoms have what charge, and how many protons and electrons? ...
The New Alchemy
... Protons – one of the parts of an atom. Protons have a (+) charge and are found in the nucleus. Neutrons – one of the parts of an atom. Neutrons have no charge and are found in the nucleus. Nucleus – found in the center of an atom. It contains protons and neutrons. Nuclei is the plural of nucleus. Nu ...
... Protons – one of the parts of an atom. Protons have a (+) charge and are found in the nucleus. Neutrons – one of the parts of an atom. Neutrons have no charge and are found in the nucleus. Nucleus – found in the center of an atom. It contains protons and neutrons. Nuclei is the plural of nucleus. Nu ...
Ch. 5 Outline Notes
... Ch. 5 Lecture Notes Atomic Structure and the Periodic Table 5-1 Atoms A. Early Models of the Atom 1. ____________________ – 4th century BC – world made up of empty space and tiny particles called _______________ (atomos) ‘indivisible’ a. Hypothesized _________________ using experiments B. __________ ...
... Ch. 5 Lecture Notes Atomic Structure and the Periodic Table 5-1 Atoms A. Early Models of the Atom 1. ____________________ – 4th century BC – world made up of empty space and tiny particles called _______________ (atomos) ‘indivisible’ a. Hypothesized _________________ using experiments B. __________ ...
Atoms - ChemistryatBiotech
... What elements are made by small stars? What additional elements are made by large ...
... What elements are made by small stars? What additional elements are made by large ...
Physical Science Notes–Ch. 17-Glencoe
... An element is matter that is composed of one type of atom, which is the ________________________________________________________ of the element. ...
... An element is matter that is composed of one type of atom, which is the ________________________________________________________ of the element. ...
I CAN write Chemical formulas
... the oxidation numbers and write the oxidation number (without plus or minus) of one element as the subscript of the other element. 3. Reduce the subscripts (number of atoms) to their simplest form, if needed. WHAT IS THE CHEMICAL FORMULA FOR CALCIUM CHLORIDE? ...
... the oxidation numbers and write the oxidation number (without plus or minus) of one element as the subscript of the other element. 3. Reduce the subscripts (number of atoms) to their simplest form, if needed. WHAT IS THE CHEMICAL FORMULA FOR CALCIUM CHLORIDE? ...
Extra Credit Test Review
... 1. There are more than 110 known elements on the Periodic Table, but no element with an atomic number greater than 92 is found naturally in measurable quantities on Earth. The remaining elements are artificially produced in a laboratory setting. 2. What did Rutherford contribute to the Atomic Theory ...
... 1. There are more than 110 known elements on the Periodic Table, but no element with an atomic number greater than 92 is found naturally in measurable quantities on Earth. The remaining elements are artificially produced in a laboratory setting. 2. What did Rutherford contribute to the Atomic Theory ...
Chapter 4
... 1. All matter is made up of very tiny, indivisible particles (atoms). 2. All atoms of a given element have the same chemical properties. 3. Compounds are made up of two or more different kinds of atoms. A compound has the same relative numbers and types of atoms. ...
... 1. All matter is made up of very tiny, indivisible particles (atoms). 2. All atoms of a given element have the same chemical properties. 3. Compounds are made up of two or more different kinds of atoms. A compound has the same relative numbers and types of atoms. ...
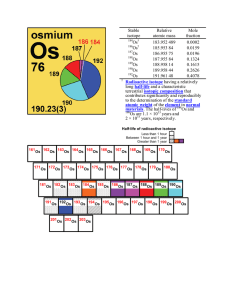
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... radiation, neutrons or protons to reach a final stable energy state. radioisotope (radioactive isotope) – an atom for which radioactive decay has been experimentally measured (also see half-life). [return] radiogenic – produced by the decay of a radioactive isotope, but which itself may or may not b ...
... radiation, neutrons or protons to reach a final stable energy state. radioisotope (radioactive isotope) – an atom for which radioactive decay has been experimentally measured (also see half-life). [return] radiogenic – produced by the decay of a radioactive isotope, but which itself may or may not b ...
Chemistry Notes
... • There are more metals on the Periodic Table than non-metals. The white is non-metals. The metals are everything that isn’t white. ...
... • There are more metals on the Periodic Table than non-metals. The white is non-metals. The metals are everything that isn’t white. ...
Chemistry for Changing Times
... Groups of atoms chemically bonded together H represents a hydrogen atom H2 represents a hydrogen molecule How many atoms of O are in H2O2? Be careful when writing formulas for ...
... Groups of atoms chemically bonded together H represents a hydrogen atom H2 represents a hydrogen molecule How many atoms of O are in H2O2? Be careful when writing formulas for ...
Getting to Know: Periodic Table
... element do not have the same number of neutrons. For example, most atoms of the element carbon, C, have 6 neutrons in their nucleus. There are some atoms of carbon that have 7 or even 8 neutrons in their nucleus. Atoms of the same element that have a different number of neutrons are called isotopes. ...
... element do not have the same number of neutrons. For example, most atoms of the element carbon, C, have 6 neutrons in their nucleus. There are some atoms of carbon that have 7 or even 8 neutrons in their nucleus. Atoms of the same element that have a different number of neutrons are called isotopes. ...
Document
... 2. Three transitional metals in Group 12 of the periodic table are _______________. 3. Given the compound Li3N , what is the oxidation number for nitrogen? 4. A combustion reaction must have________________ included in the reactants. 5. The elements that make up a compound and the exact number of at ...
... 2. Three transitional metals in Group 12 of the periodic table are _______________. 3. Given the compound Li3N , what is the oxidation number for nitrogen? 4. A combustion reaction must have________________ included in the reactants. 5. The elements that make up a compound and the exact number of at ...
Matter: A) Homogeneous Matter • Uniform and in 1 phase • Even
... Use stock system if necessary Ex: Fe(NO3)2 – Iron (II) Nitrate or Ferrous Nitrate Binary Molecular Compounds: Use prefixes, but don’t use mono on the first element The second element also ends in “ide” Ex: N2O3 (Dinitrogen trioxide) If there are two conflicting vowels right next to each othe ...
... Use stock system if necessary Ex: Fe(NO3)2 – Iron (II) Nitrate or Ferrous Nitrate Binary Molecular Compounds: Use prefixes, but don’t use mono on the first element The second element also ends in “ide” Ex: N2O3 (Dinitrogen trioxide) If there are two conflicting vowels right next to each othe ...
What does an elements atomic # tell us about the element?
... Smallest part of a compound that still has all the properties of that compound Compound can have properties entirely unlike the elements of which it is made ...
... Smallest part of a compound that still has all the properties of that compound Compound can have properties entirely unlike the elements of which it is made ...