1000 - Paint Valley Local Schools
... shell. Therefore, they are ready to lose that one electron in ionic bonding with other elements very easily. This makes them _______ _________ especially with group 17 because they have 7 valence electrons and when the two combine they make the magic number “8”. ...
... shell. Therefore, they are ready to lose that one electron in ionic bonding with other elements very easily. This makes them _______ _________ especially with group 17 because they have 7 valence electrons and when the two combine they make the magic number “8”. ...
1 Chapter 4 Atomic Structure 4.1 Defining the Atom Early Models of
... element are different from those of any other element. 3. Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. 4. Chemical reactions occur when _____________ are separated, joined or rearranged. Atoms of one element, howev ...
... element are different from those of any other element. 3. Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. 4. Chemical reactions occur when _____________ are separated, joined or rearranged. Atoms of one element, howev ...
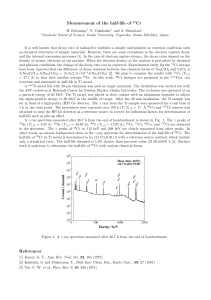
Measurement of the half-life of
... It is well known that decay rate of radioactive nuclides is usually independent on external conditions such as chemical structures of sample materials. However, there are some exceptions in the electron capture decay and the internal conversion processes [1]. In the case of electron capture decays, ...
... It is well known that decay rate of radioactive nuclides is usually independent on external conditions such as chemical structures of sample materials. However, there are some exceptions in the electron capture decay and the internal conversion processes [1]. In the case of electron capture decays, ...
Study Island Copyright © 2012 Study Island
... 28. Sea salt, which is mostly chloride and sodium, makes up about 3.5% of the world's oceans. Which chemical makes up the remaining 96.5% of the world's oceans? A. CH4 B. O2 C. CO2 D. H2O ...
... 28. Sea salt, which is mostly chloride and sodium, makes up about 3.5% of the world's oceans. Which chemical makes up the remaining 96.5% of the world's oceans? A. CH4 B. O2 C. CO2 D. H2O ...
T1 Final Study Guide - District 196 e
... a. The basketball is orange Intensive b. The diameter of the basketball is 31 centimeters Extensive c. The surface of the basketball has indented seams. Intensive d. The density of copper is 8.92 g/cm3. Intensive 7. Determine the number of significant figures in each of the following measurement res ...
... a. The basketball is orange Intensive b. The diameter of the basketball is 31 centimeters Extensive c. The surface of the basketball has indented seams. Intensive d. The density of copper is 8.92 g/cm3. Intensive 7. Determine the number of significant figures in each of the following measurement res ...
2-1 Chemistry of life
... energy levels of the six electrons in a carbon atom. Carbon will have 2 electrons in the first energy level and four electrons in the second level How many valence electrons does carbon have to share with other atoms? ...
... energy levels of the six electrons in a carbon atom. Carbon will have 2 electrons in the first energy level and four electrons in the second level How many valence electrons does carbon have to share with other atoms? ...
Chapter Two - Alfred State College intranet site
... in order of atomic weight, they could be placed in horizontal rows, one row under the other, so that the elements in vertical columns have similar properties! ...
... in order of atomic weight, they could be placed in horizontal rows, one row under the other, so that the elements in vertical columns have similar properties! ...
Inside the Atom
... B. Periodic Table – Chart that organizes and displays information about the elements. 1. Atomic number – the top number in the element’s periodic table block a. Tells the number of protons in the nucleus of each atom of that element b. The number of protons remains constant in every atom of an elem ...
... B. Periodic Table – Chart that organizes and displays information about the elements. 1. Atomic number – the top number in the element’s periodic table block a. Tells the number of protons in the nucleus of each atom of that element b. The number of protons remains constant in every atom of an elem ...
ATOMS AND ELEMENTS
... 1. Capitalize the first letter of the element name, like C for carbon. 2. Capitalize the first letter of the element name and then make the second letter of the element’s name small case, like Ca for calcium. 3. Sometimes the element’s symbol does not seem to fit the element’s name at all, like Fe f ...
... 1. Capitalize the first letter of the element name, like C for carbon. 2. Capitalize the first letter of the element name and then make the second letter of the element’s name small case, like Ca for calcium. 3. Sometimes the element’s symbol does not seem to fit the element’s name at all, like Fe f ...
7th Grade Atomic Structure and Periodic Table of Elements
... Metalloids – acts a a semiconductor, at high temperatures they are good conductors of electricity, at low temperatures stops electricity from flowing 5. What significance do the elements play in our daily life? Chemistry is all around us. We use it in the kitchen while cooking (chemical reactions), ...
... Metalloids – acts a a semiconductor, at high temperatures they are good conductors of electricity, at low temperatures stops electricity from flowing 5. What significance do the elements play in our daily life? Chemistry is all around us. We use it in the kitchen while cooking (chemical reactions), ...
Name Period ______ Unit 4 Study Guide A common isotope of iron
... If an atom becomes an ion by gaining three electrons, what is its oxidation number (charge)? List the Alkali Metals. Which group will commonly form compounds with oxygen in a one-to-one ratio? Which metals have a tendency to gain two electrons in order to be stable? Most non-metals on the periodic t ...
... If an atom becomes an ion by gaining three electrons, what is its oxidation number (charge)? List the Alkali Metals. Which group will commonly form compounds with oxygen in a one-to-one ratio? Which metals have a tendency to gain two electrons in order to be stable? Most non-metals on the periodic t ...
03.03a Atomic Number, Mass Number, and Isotopes
... IONS: Just as isotopes have different numbers of neutrons, ions have different numbers of electrons. Because protons are positively charged, and electrons are negatively charged, atoms are considered neutral because the numbers of each are equal. However, ions have different numbers of protons and e ...
... IONS: Just as isotopes have different numbers of neutrons, ions have different numbers of electrons. Because protons are positively charged, and electrons are negatively charged, atoms are considered neutral because the numbers of each are equal. However, ions have different numbers of protons and e ...
ATOMS, MOLECULES and IONS
... Chemical properties of elements depend on the atomic number of the element. A complete Periodic Table lists the elements, their symbols and atomic numbers as well as atomic masses. The Periodic Table is arranged into rows, called periods and columns, which are called groups. The first period consist ...
... Chemical properties of elements depend on the atomic number of the element. A complete Periodic Table lists the elements, their symbols and atomic numbers as well as atomic masses. The Periodic Table is arranged into rows, called periods and columns, which are called groups. The first period consist ...
PS 2.2 - S2TEM Centers SC
... Introduction to the lesson: Isotopes have the same atomic number and hence nearly identical chemical behavior but different atomic masses. Most elements found in nature are mixtures of several isotopes; tin, for example, has 10 isotopes. In most cases, only stable isotopes of elements are found in n ...
... Introduction to the lesson: Isotopes have the same atomic number and hence nearly identical chemical behavior but different atomic masses. Most elements found in nature are mixtures of several isotopes; tin, for example, has 10 isotopes. In most cases, only stable isotopes of elements are found in n ...
Trends in the Periodic Table
... Name: _________________________________ Period: ____ Date: ___ / ___ / ___ ...
... Name: _________________________________ Period: ____ Date: ___ / ___ / ___ ...
Elements, Compounds, Mixtures
... 1862, Alexandre-Emile de Chancourtois, the first notion of periodicity by increasing atomic weight, “screw” periodic table 1864, Julius Lothar Meyer, table based on valency 1863-1866, John Newlands, Law of Octaves ...
... 1862, Alexandre-Emile de Chancourtois, the first notion of periodicity by increasing atomic weight, “screw” periodic table 1864, Julius Lothar Meyer, table based on valency 1863-1866, John Newlands, Law of Octaves ...
Name Date Class Period ______
... F. Proton G. M shell H. Electron I. Nucleus J. Shells K. Atom L. Element M. Subatomic particles ...
... F. Proton G. M shell H. Electron I. Nucleus J. Shells K. Atom L. Element M. Subatomic particles ...
atomic number
... distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed but only exchanged between ...
... distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed but only exchanged between ...
No Slide Title
... distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed but only exchanged between ...
... distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed but only exchanged between ...
Chemical Element
... During the early phases of the Big Bang, nucleosynthesis of hydrogen nuclei resulted in the production of hydrogen and helium isotopes, as well as very minuscule amounts (on the order of 10-10) of lithium and beryllium. No heavier elements were produced. As a result, the primordial abundance of ato ...
... During the early phases of the Big Bang, nucleosynthesis of hydrogen nuclei resulted in the production of hydrogen and helium isotopes, as well as very minuscule amounts (on the order of 10-10) of lithium and beryllium. No heavier elements were produced. As a result, the primordial abundance of ato ...
Subatomic Heavyweights
... Atoms of the same element will ALWAYS have the same number of protons • Atomic weight: the weighted average atomic mass of the naturally occurring isotopes (the # on the periodic table) ...
... Atoms of the same element will ALWAYS have the same number of protons • Atomic weight: the weighted average atomic mass of the naturally occurring isotopes (the # on the periodic table) ...
PS-CC-2test - Edquest Science
... 12. As you move across the periodic table the properties of the elements change. The most reactive metals include … A. sodium and lithium B. iron and copper C. aluminum and carbon D. lead and zinc 13. The periodic table is organized by the patterns of the properties of the elements. The rows in the ...
... 12. As you move across the periodic table the properties of the elements change. The most reactive metals include … A. sodium and lithium B. iron and copper C. aluminum and carbon D. lead and zinc 13. The periodic table is organized by the patterns of the properties of the elements. The rows in the ...
Chapter 3 notes
... • Orbital- or energy shell/level is a region in an atom where there is a high probability of finding electrons. • Valence electron- an electron in the outermost energy level of an atom. So in Al valence 3. ...
... • Orbital- or energy shell/level is a region in an atom where there is a high probability of finding electrons. • Valence electron- an electron in the outermost energy level of an atom. So in Al valence 3. ...