Atoms, Molecules, and Ions Chapter 2 Handout 1 The Atom Dalton`s
... 1. Each element is composed of extremely small particles called atoms. 2. All atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of all the other elements. 3. Atoms are neither created nor destroyed in chem ...
... 1. Each element is composed of extremely small particles called atoms. 2. All atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of all the other elements. 3. Atoms are neither created nor destroyed in chem ...
The Atom - Taylorsville
... Five main points of Dalton's atomic theory 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. ...
... Five main points of Dalton's atomic theory 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. ...
Another look at chemical reactions HYDROGEN PEROXIDE WATER
... Example: Helium has an atomic number of 2. Every helium atom has two protons in its nucleus. - MASS NUMBER: The number of protons PLUS the number of neutrons in the atomic nucleus, Atoms of the same element may have DIFFERENT mass numbers. - ISOTOPES: are atoms of the same element with different mas ...
... Example: Helium has an atomic number of 2. Every helium atom has two protons in its nucleus. - MASS NUMBER: The number of protons PLUS the number of neutrons in the atomic nucleus, Atoms of the same element may have DIFFERENT mass numbers. - ISOTOPES: are atoms of the same element with different mas ...
S1-2-02: What is the basic subatomic structure of an atom?
... 10. Which one of the following is a physical change? a) Acid damages the surface of a car. b) The car burns up gasoline on a trip. c) The car explodes in a collision. d) The auto-wrecker crushes the car into a tiny cube. ...
... 10. Which one of the following is a physical change? a) Acid damages the surface of a car. b) The car burns up gasoline on a trip. c) The car explodes in a collision. d) The auto-wrecker crushes the car into a tiny cube. ...
Atoms, Elements, and the Periodic Table Part 1: The Atomic Model
... column predict share the properties similar chemical of the unknown properties. elements. ...
... column predict share the properties similar chemical of the unknown properties. elements. ...
Biochemistry I (CHE 418 / 5418)
... – Excited state when electrons is pushed into an orbit farther from the nucleus. • When electrons move from an excited state (higher energy level) to the ground state (lower energy level), the energy produced is given off often as light. • Worked well to explain the emision spectrum of hydrogen, but ...
... – Excited state when electrons is pushed into an orbit farther from the nucleus. • When electrons move from an excited state (higher energy level) to the ground state (lower energy level), the energy produced is given off often as light. • Worked well to explain the emision spectrum of hydrogen, but ...
Atoms, Elements, and the Periodic Table Part 1: The Atomic Model
... column predict share the properties similar chemical of the unknown properties. elements. ...
... column predict share the properties similar chemical of the unknown properties. elements. ...
ATOMIC THEORY
... The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different. What makes atoms of different elements different? The fundamental characteristic that all atoms of the same element share is the number of protons . All atoms of hydrogen have on ...
... The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different. What makes atoms of different elements different? The fundamental characteristic that all atoms of the same element share is the number of protons . All atoms of hydrogen have on ...
Kentucky newspapers 1949 look at the city, part 5
... All the phases of atomic energy are, of course, based on the atom, the basis of all substance and most or all energy in this world of ours. What is an atom? Well, it resembles the solar system. It is a particle so small that man has not been able to see it; and he doesn’t even hope ever to be able t ...
... All the phases of atomic energy are, of course, based on the atom, the basis of all substance and most or all energy in this world of ours. What is an atom? Well, it resembles the solar system. It is a particle so small that man has not been able to see it; and he doesn’t even hope ever to be able t ...
AP Chemistry
... 2.5.5 Nonmetals—upper right side of table, range of properties 2.5.6 “Stairway to heaven” separates metals from nonmetals; elements along this are called metalloids 2.6 Molecules and Molecular Compounds 2.6.1 Molecule = assembly of two or more atoms tightly bound together 2.6.1.1 Diatomic molecule: ...
... 2.5.5 Nonmetals—upper right side of table, range of properties 2.5.6 “Stairway to heaven” separates metals from nonmetals; elements along this are called metalloids 2.6 Molecules and Molecular Compounds 2.6.1 Molecule = assembly of two or more atoms tightly bound together 2.6.1.1 Diatomic molecule: ...
STURCTURES AND PROPERTIES OF MATTER
... Carbon-14 and Carbon-13 atoms’ are not as stable as carbon-12 and easily break down. If an isotope has too many or too few neutrons compared to the number of protons, it is unstable and will undergo radioactive decay. These radioactive isotopes become different elements in an effort to become more s ...
... Carbon-14 and Carbon-13 atoms’ are not as stable as carbon-12 and easily break down. If an isotope has too many or too few neutrons compared to the number of protons, it is unstable and will undergo radioactive decay. These radioactive isotopes become different elements in an effort to become more s ...
Terminology 1
... (The chemical identity of an atom can be determined solely by it’s atomic number) When the atom is neutral, i.e. not electrically charged, the atomic number equals the number of electrons in its shells ...
... (The chemical identity of an atom can be determined solely by it’s atomic number) When the atom is neutral, i.e. not electrically charged, the atomic number equals the number of electrons in its shells ...
Chemical Equations - Warren County Schools
... results: a small portion of the particles were deflected, indicating a small, concentrated positive charge. Note that the image is not to scale; in reality the nucleus is vastly smaller than the electron shell. ...
... results: a small portion of the particles were deflected, indicating a small, concentrated positive charge. Note that the image is not to scale; in reality the nucleus is vastly smaller than the electron shell. ...
atomic number
... distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed but only exchanged between ...
... distinguished by their different masses – Compounds are combinations of atoms of different elements and possess properties different from those of their component elements – In chemical reactions, atoms are neither created nor destroyed but only exchanged between ...
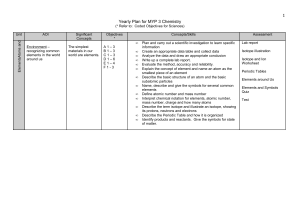
Yearly Plan for MYP 1 Science
... Create an appropriate data table and collect data Analyze the data and draw an appropriate conclusion Write up a complete lab report. Evaluate the method, accuracy and reliability. Explain the concept of element and name an atom as the smallest piece of an element Describe the basic structure of an ...
... Create an appropriate data table and collect data Analyze the data and draw an appropriate conclusion Write up a complete lab report. Evaluate the method, accuracy and reliability. Explain the concept of element and name an atom as the smallest piece of an element Describe the basic structure of an ...
AP Chapter 2 Outline 2014
... (1) Each element is made of extremely small particles (atoms). (2) All atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of other elements. (3) Atoms of an element are not changed into atoms of a different el ...
... (1) Each element is made of extremely small particles (atoms). (2) All atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of other elements. (3) Atoms of an element are not changed into atoms of a different el ...
Chem 200 Dr. Saidane
... a) The Law of Conservation of Mass, which states that mass is neither destroyed nor created during ordinary chemical reactions. b) The Law of Definite Proportions, which states that a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the s ...
... a) The Law of Conservation of Mass, which states that mass is neither destroyed nor created during ordinary chemical reactions. b) The Law of Definite Proportions, which states that a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the s ...
Chemistry Scavenger Hunt
... Go to the “States of Matter” area (left side) to find the answers to these questions. 1. Matter is anything occupying _______________ and having ______________; it is the material of the _______________. 2. There are three main phases of matter: _____________, ________________, and _____________. Th ...
... Go to the “States of Matter” area (left side) to find the answers to these questions. 1. Matter is anything occupying _______________ and having ______________; it is the material of the _______________. 2. There are three main phases of matter: _____________, ________________, and _____________. Th ...
1. Of the three major categories of elements (metals, non
... They are called groups or families. 12. What are the horizontal rows on the periodic table called? They are called periods. 13. Explain the relationship between elements in the same group. They have similar chemical and physical properties because each one has the same number of valence electrons. ...
... They are called groups or families. 12. What are the horizontal rows on the periodic table called? They are called periods. 13. Explain the relationship between elements in the same group. They have similar chemical and physical properties because each one has the same number of valence electrons. ...
Chapter 5
... o Using carbon-12 as the standard, an atomic mass unit (amu) is defined as one twelfth of the mass of a carbon-12 atom o In nature, most elements occur as a mixture of two or more isotopes o Atomic mass is the average mass of the atoms in a naturally occurring sample of the element - To calculate th ...
... o Using carbon-12 as the standard, an atomic mass unit (amu) is defined as one twelfth of the mass of a carbon-12 atom o In nature, most elements occur as a mixture of two or more isotopes o Atomic mass is the average mass of the atoms in a naturally occurring sample of the element - To calculate th ...
Present - Images
... For fun, the Radium Girls painted their nails, teeth and faces with the deadly paint produced at the factory, sometimes to surprise their boyfriends when the lights went out. They mixed glue, water and radium powder, and then used camel hair brushes to apply the glowing paint onto dial numbers. The ...
... For fun, the Radium Girls painted their nails, teeth and faces with the deadly paint produced at the factory, sometimes to surprise their boyfriends when the lights went out. They mixed glue, water and radium powder, and then used camel hair brushes to apply the glowing paint onto dial numbers. The ...
Chapter 3.1 PPT
... the same proportions by mass regardless of the size of the sample or source of the compound • Law of multiple proportions: if two or more different compounds are composed of the same two elements, then the ratio of the masses of the second element combined with a certain mass of the first element is ...
... the same proportions by mass regardless of the size of the sample or source of the compound • Law of multiple proportions: if two or more different compounds are composed of the same two elements, then the ratio of the masses of the second element combined with a certain mass of the first element is ...
Isotopes
... • Isotopes are atoms that have the same number of protons, but different numbers of neutrons. • They can be a radioactive form of an element. – Atoms of the same element all have the same number of protons. – Isotopes of the element have different numbers of neutrons. ...
... • Isotopes are atoms that have the same number of protons, but different numbers of neutrons. • They can be a radioactive form of an element. – Atoms of the same element all have the same number of protons. – Isotopes of the element have different numbers of neutrons. ...
Atomic Theory, Nomenclature, and Balancing - Ars
... A theory (or model) of the way matter works goes back to the ancient Greeks. Two competing theories at that time were the idea that matter is continuous and that matter is composed of indivisible particles called atoms. The first states that however finely matter is divided it is the same. The secon ...
... A theory (or model) of the way matter works goes back to the ancient Greeks. Two competing theories at that time were the idea that matter is continuous and that matter is composed of indivisible particles called atoms. The first states that however finely matter is divided it is the same. The secon ...