Pdf - Text of NPTEL IIT Video Lectures
... This is definition of the problem apart from that there is another thing. Assuming the temperature difference of 11.1 degree Celsius exists between the working fluid, it is very important, and the river water on the one hand and the required room temperature on the other that means when the heat pum ...
... This is definition of the problem apart from that there is another thing. Assuming the temperature difference of 11.1 degree Celsius exists between the working fluid, it is very important, and the river water on the one hand and the required room temperature on the other that means when the heat pum ...
3 CO 2 (g) + 4 H 2 O(l)
... Enthalpy is not stored as heat. Rather enthalpy is a measure of the capacity of the system to supply energy as heat when a certain change occurs. The enthalpy of a substance will depend on its physical state (solid, liquid or vapour) and its temperature (it will also depend upon the pressure but thi ...
... Enthalpy is not stored as heat. Rather enthalpy is a measure of the capacity of the system to supply energy as heat when a certain change occurs. The enthalpy of a substance will depend on its physical state (solid, liquid or vapour) and its temperature (it will also depend upon the pressure but thi ...
Scorpion - Dunn Labortechnik GmbH
... Multiple tip sizes can be used in one protocol. Suitable pipette tips: see separate price list. Liquid transfers can be defined as volume, concentration or pH. Possibility to set up multiple plates with varying pH, different additives and varying concentration e. g normalization of salt screens. The ...
... Multiple tip sizes can be used in one protocol. Suitable pipette tips: see separate price list. Liquid transfers can be defined as volume, concentration or pH. Possibility to set up multiple plates with varying pH, different additives and varying concentration e. g normalization of salt screens. The ...
Engines and the Second Law of Thermodynamics
... It is easy to produce thermal energy using work, but how does one produce work using thermal energy? This is a heat engine; mechanical energy can be obtained from thermal energy only when heat can flow from a higher temperature to a lower temperature. ...
... It is easy to produce thermal energy using work, but how does one produce work using thermal energy? This is a heat engine; mechanical energy can be obtained from thermal energy only when heat can flow from a higher temperature to a lower temperature. ...
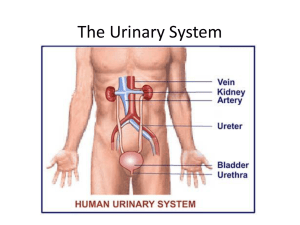
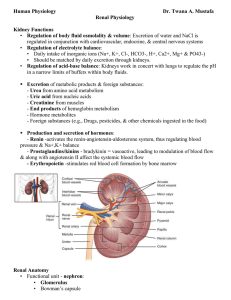
Structure and Function of the Kidneys and Nephrons Note
... 1. Forms a shell around medulla 2. Projects into medulla between renal pyramids, forming ________________________. 5. Describe 4 functions of the kidneys. ...
... 1. Forms a shell around medulla 2. Projects into medulla between renal pyramids, forming ________________________. 5. Describe 4 functions of the kidneys. ...
Why is S(H2O(l) > S(H20(g)? It is better to speak of entropy as a
... equilibrium and in a low entropy (improbable) state, where the equilibrium state would be simpler, less complex, and higher entropy. From: http://www.structuralcommunication.com/sc/thermalphysics/sc_thermalphysics_unit1.html: ...
... equilibrium and in a low entropy (improbable) state, where the equilibrium state would be simpler, less complex, and higher entropy. From: http://www.structuralcommunication.com/sc/thermalphysics/sc_thermalphysics_unit1.html: ...
Phase changes
... one may write hLhL0=cL(TT0)+(pp0)/L and hVhV0=cp(TT0), but both hL0 and hV0 cannot be chosen as zero because hV0hL0=hLV0. The reference state is a free choice to the user; a liquid-state reference is preferable to work with positive numbers most of the time, but several different temperatures ...
... one may write hLhL0=cL(TT0)+(pp0)/L and hVhV0=cp(TT0), but both hL0 and hV0 cannot be chosen as zero because hV0hL0=hLV0. The reference state is a free choice to the user; a liquid-state reference is preferable to work with positive numbers most of the time, but several different temperatures ...
Thermodynamics and Kinetics
... Heat capacity - The amount of heat needed to raise the temperature of a defined amount of a pure substance by one degree. Specific heat - The amount of heat needed to raise the temperature of one gram of a substance by 1 C (or 1 K) •SI unit for specific heat is joules per gram-1 Kelvin-1 (J/g-K) Cal ...
... Heat capacity - The amount of heat needed to raise the temperature of a defined amount of a pure substance by one degree. Specific heat - The amount of heat needed to raise the temperature of one gram of a substance by 1 C (or 1 K) •SI unit for specific heat is joules per gram-1 Kelvin-1 (J/g-K) Cal ...
Countercurrent exchange

Countercurrent exchange is a mechanism occurring in nature and mimicked in industry and engineering, in which there is a crossover of some property, usually heat or some component, between two flowing bodies flowing in opposite directions to each other. The flowing bodies can be liquids, gases, or even solid powders, or any combination of those. For example, in a distillation column, the vapors bubble up through the downward flowing liquid while exchanging both heat and mass.The maximum amount of heat or mass transfer that can be obtained is higher with countercurrent than co-current (parallel) exchange because countercurrent maintains a slowly declining difference or gradient (usually temperature or concentration difference). In cocurrent exchange the initial gradient is higher but falls off quickly, leading to wasted potential. For example, in the diagram at the right, the fluid being heated (exiting top) has a higher exiting temperature than the cooled fluid (exiting bottom) that was used for heating. With cocurrent or parallel exchange the heated and cooled fluids can only approach one another. The result is that countercurrent exchange can achieve a greater amount of heat or mass transfer than parallel under otherwise similar conditions. See: flow arrangement.Countercurrent exchange when set up in a circuit or loop can be used for building up concentrations, heat, or other properties of flowing liquids. Specifically when set up in a loop with a buffering liquid between the incoming and outgoing fluid running in a circuit, and with active transport pumps on the outgoing fluid's tubes, the system is called a Countercurrent multiplier, enabling a multiplied effect of many small pumps to gradually build up a large concentration in the buffer liquid.Other countercurrent exchange circuits where the incoming and outgoing fluids touch each other are used for retaining a high concentration of a dissolved substance or for retaining heat, or for allowing the external buildup of the heat or concentration at one point in the system.Countercurrent exchange circuits or loops are found extensively in nature, specifically in biologic systems. In vertebrates, they are called a Rete mirabile, originally the name of an organ in fish gills for absorbing oxygen from the water. It is mimicked in industrial systems. Countercurrent exchange is a key concept in chemical engineering thermodynamics and manufacturing processes, for example in extracting sucrose from sugar beet roots.Countercurrent multiplication is a similar but different concept where liquid moves in a loop followed by a long length of movement in opposite directions with an intermediate zone. The tube leading to the loop passively building up a gradient of heat (or cooling) or solvent concentration while the returning tube has a constant small pumping action all along it, so that a gradual intensification of the heat or concentration is created towards the loop. Countercurrent multiplication has been found in the kidneys as well as in many other biological organs.