Isopropanol oxidation by pure metal oxide
... reduction of the metal oxide surface can usually take place. Other methods involve the adsorption of CO2 and NH3 . CO2 and NH3 do not measure all the surface sites. CO2 only adsorbs on basic OH groups on the surface and NH3 only adsorbs on Lewis and Bronsted acid sites. Furthermore, the above method ...
... reduction of the metal oxide surface can usually take place. Other methods involve the adsorption of CO2 and NH3 . CO2 and NH3 do not measure all the surface sites. CO2 only adsorbs on basic OH groups on the surface and NH3 only adsorbs on Lewis and Bronsted acid sites. Furthermore, the above method ...
Thermal and Mechanical Characterizations of W-armoured
... figure 4-a, and it can be observed that the adapted theoretical relation does not fit at all to the experimental results. As a consequence, an adaption of the Hall-Petch relation by fitting the experimental data has been carried out, leading to a reassessment of parameters ‘a’ and ‘b’ at 65.5 and 7 ...
... figure 4-a, and it can be observed that the adapted theoretical relation does not fit at all to the experimental results. As a consequence, an adaption of the Hall-Petch relation by fitting the experimental data has been carried out, leading to a reassessment of parameters ‘a’ and ‘b’ at 65.5 and 7 ...
1 Assignment 5 Hydrogen – The Unique Element
... Both molecular and saline hydrides are quite reactive. Group 1 and 2 hydrides react vigorously with water to produce hydrogen gas and a metal hydroxide. This means that they can be used as drying agents for solvents – the most commonly used in this regard is CaH2. p-Block molecular hydrides have dif ...
... Both molecular and saline hydrides are quite reactive. Group 1 and 2 hydrides react vigorously with water to produce hydrogen gas and a metal hydroxide. This means that they can be used as drying agents for solvents – the most commonly used in this regard is CaH2. p-Block molecular hydrides have dif ...
1 Assignment 4 Hydrogen – The Unique Element
... Both molecular and saline hydrides are quite reactive. Group 1 and 2 hydrides react vigorously with water to produce hydrogen gas and a metal hydroxide. This means that they can be used as drying agents for solvents – the most commonly used in this regard is CaH2. p-Block molecular hydrides have dif ...
... Both molecular and saline hydrides are quite reactive. Group 1 and 2 hydrides react vigorously with water to produce hydrogen gas and a metal hydroxide. This means that they can be used as drying agents for solvents – the most commonly used in this regard is CaH2. p-Block molecular hydrides have dif ...
study material class X (science)
... Ans: We know that copper is more reactive than silver, so it will displace silver from its salt solution: Cu(s) + 2AgNO3(aq) Cu(NO3)2(aq)+2Ag(s) So the solution will turn blue due to the formation of copper nitrate. ...
... Ans: We know that copper is more reactive than silver, so it will displace silver from its salt solution: Cu(s) + 2AgNO3(aq) Cu(NO3)2(aq)+2Ag(s) So the solution will turn blue due to the formation of copper nitrate. ...
New Materials from Metal Vapour Chemistry
... Another interesting example of cluster growth in the liquid phase that has recently found application in the fabrication of fuel cell electrodes involves the interaction of e.g. Ag, Pd and Pt atoms with ether and aromatic solvents, liquid oligoand poly-ethers and olefins, in the presence of various ...
... Another interesting example of cluster growth in the liquid phase that has recently found application in the fabrication of fuel cell electrodes involves the interaction of e.g. Ag, Pd and Pt atoms with ether and aromatic solvents, liquid oligoand poly-ethers and olefins, in the presence of various ...
Chapter 1 Introduction
... variety of coordinated metal ions is expressed by the vast number of references that can be found for metal-salen complexes in chemical databases: for each of about 20 different metals more ...
... variety of coordinated metal ions is expressed by the vast number of references that can be found for metal-salen complexes in chemical databases: for each of about 20 different metals more ...
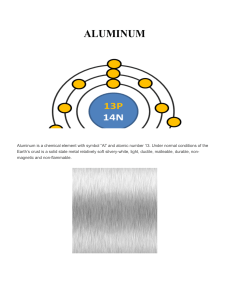
ALUMINUM
... Aluminum is highly reactive and it is very rare to find natively on the crust, so it is a very versatile, which can constitute about 300 alloys and a lot of variants. Despite this, aluminum does not ignite instantly, so it has many applications in handling flammable or explosive materials. Aluminum ...
... Aluminum is highly reactive and it is very rare to find natively on the crust, so it is a very versatile, which can constitute about 300 alloys and a lot of variants. Despite this, aluminum does not ignite instantly, so it has many applications in handling flammable or explosive materials. Aluminum ...
Descriptive Chemistry of Elements d-Block
... [CoBr3(NH3)3] leaves three positive charges on cobalt. Therefore, the oxidation number of cobalt in [CoBr3(NH3)3] is +3 or +III. Note that the sum of charges inside the square bracket [ ] is equal to the charge of the complex ion [ ]n+ or [ ]n (written outside the bracket). For a neutral complex li ...
... [CoBr3(NH3)3] leaves three positive charges on cobalt. Therefore, the oxidation number of cobalt in [CoBr3(NH3)3] is +3 or +III. Note that the sum of charges inside the square bracket [ ] is equal to the charge of the complex ion [ ]n+ or [ ]n (written outside the bracket). For a neutral complex li ...
Unit - II Electrochemistry
... The gas collects in gaps and voids and causes blisters and fissures. Consequently the metal becomes weak. By other gases : Gases like, SO2, CO2, H2S, F2 and Cl2 are also corrosive. ...
... The gas collects in gaps and voids and causes blisters and fissures. Consequently the metal becomes weak. By other gases : Gases like, SO2, CO2, H2S, F2 and Cl2 are also corrosive. ...
Characterization of various zinc oxide catalysts
... oxide catalysts were more selective for isobutene, as the predominant dehydrated product. The single oxide catalyst exhibited higher selectivity towards isobutyraldehyde as the dehydrogenated product, and the selectivity increased with increasing reaction temperature. Keywords: metal oxides; acid–ba ...
... oxide catalysts were more selective for isobutene, as the predominant dehydrated product. The single oxide catalyst exhibited higher selectivity towards isobutyraldehyde as the dehydrogenated product, and the selectivity increased with increasing reaction temperature. Keywords: metal oxides; acid–ba ...
Preparation and Properties of Hydrogen
... If hydrogen is heated without oxygen, no reaction will take place. This means that hydrogen does not support combustion; it is the fuel. REACTIONS OF METAL WITH AN ACID TO PRODUCE HYDROGEN Various metals react with acids to produce hydrogen gas (H2) and metallic salts. The rate at which the acid rea ...
... If hydrogen is heated without oxygen, no reaction will take place. This means that hydrogen does not support combustion; it is the fuel. REACTIONS OF METAL WITH AN ACID TO PRODUCE HYDROGEN Various metals react with acids to produce hydrogen gas (H2) and metallic salts. The rate at which the acid rea ...
Semiconductor Manufacturing Process
... deposited oxide must be used rather than the grown oxides. Deposited oxides can be produced by various reactions between gaseous silicon compounds and gaseous oxidizers. Deposited oxides tend to possess low densities and large numbers of defect sites. Not suitable for use as gate dielectrics for M ...
... deposited oxide must be used rather than the grown oxides. Deposited oxides can be produced by various reactions between gaseous silicon compounds and gaseous oxidizers. Deposited oxides tend to possess low densities and large numbers of defect sites. Not suitable for use as gate dielectrics for M ...
738 influence of the oxide layer in the erosion of metals caused by
... Single-discharge erosion experiments were done in pure metals samples so as in pre-oxidized samples of RuAl intermetallic compounds. The discharges were produced in air at 9 bar and room temperature using the samples as the cathode. The volume of the craters produced by the discharge were measured u ...
... Single-discharge erosion experiments were done in pure metals samples so as in pre-oxidized samples of RuAl intermetallic compounds. The discharges were produced in air at 9 bar and room temperature using the samples as the cathode. The volume of the craters produced by the discharge were measured u ...
Lab: size of the atom
... The size of an atom is too small to imagine. Counting the number of atoms in even a microscopic amount of material is an impossible task—it would take a billion years! It turns out, however, that it is possible to apply a thin surface coating of metal atoms onto another metal. This is done to change ...
... The size of an atom is too small to imagine. Counting the number of atoms in even a microscopic amount of material is an impossible task—it would take a billion years! It turns out, however, that it is possible to apply a thin surface coating of metal atoms onto another metal. This is done to change ...
acids - WordPress.com
... with a measuring cylinder. The metal, oxide, hydroxide or carbonate is weighed out and added in small portions to the acid in the beaker with stirring. The mixture maybe heated to speed up the reaction. When no more of the solid dissolves it means the acid is neutralised. The hot solution is filtere ...
... with a measuring cylinder. The metal, oxide, hydroxide or carbonate is weighed out and added in small portions to the acid in the beaker with stirring. The mixture maybe heated to speed up the reaction. When no more of the solid dissolves it means the acid is neutralised. The hot solution is filtere ...
INTRODUCTION The HSAB concept is an acronym for `hard and soft
... The Hard Soft [Lewis] Acid Base Principle Ralph Pearson introduced the Hard Soft [Lewis] Acid Base (HSAB) principle in the early nineteen sixties, and in doing so attempted to unify inorganic and organic reaction chemistry. The impact of the new idea was immediate, however over time the HSAB princip ...
... The Hard Soft [Lewis] Acid Base Principle Ralph Pearson introduced the Hard Soft [Lewis] Acid Base (HSAB) principle in the early nineteen sixties, and in doing so attempted to unify inorganic and organic reaction chemistry. The impact of the new idea was immediate, however over time the HSAB princip ...
Structures and Bonding
... Here are some common alloys and their main uses and metals: Brass – copper and zinc Amalgam mercury ...
... Here are some common alloys and their main uses and metals: Brass – copper and zinc Amalgam mercury ...
Structure and magnetic behaviour of mononuclear and dinuclear Cu(II)/Zn(II) monocarboxylate-pyridine
... In contrast to the copper(II) complexes, the crystal structures of c and d contain no coordinated water molecules and, thus, no water hydrogen bonding. These structures are simply constructed by the intermolecular interactions between one mononuclear or dinuclear unit and the surrounding mononuclear ...
... In contrast to the copper(II) complexes, the crystal structures of c and d contain no coordinated water molecules and, thus, no water hydrogen bonding. These structures are simply constructed by the intermolecular interactions between one mononuclear or dinuclear unit and the surrounding mononuclear ...
Lecture 4 Oxidation (applies to Si and SiC only) Reading: Chapter 4
... Halogen species (Cl, F, etc...) are often introduced to getter metallic impurities from the tube during an oxidation. This also tends to increase the oxidation rate for thin oxides (linear term). HCL is the safest to use (bubbled into the furnace as described in the diffusion discussion) but is high ...
... Halogen species (Cl, F, etc...) are often introduced to getter metallic impurities from the tube during an oxidation. This also tends to increase the oxidation rate for thin oxides (linear term). HCL is the safest to use (bubbled into the furnace as described in the diffusion discussion) but is high ...
MEMS Processing
... Ions: Reactive ion etching (RIE), focused ion beams (FIB) Laser drilling: using high powered lasers (CO2/YAG) ...
... Ions: Reactive ion etching (RIE), focused ion beams (FIB) Laser drilling: using high powered lasers (CO2/YAG) ...
Balancing Redox Equations
... Metal hydrides e.g. NaH, CaH2 H2 can act as either: Oxidizing agent when it combines with metals. Reducing agent when it combines with nonmetals. ...
... Metal hydrides e.g. NaH, CaH2 H2 can act as either: Oxidizing agent when it combines with metals. Reducing agent when it combines with nonmetals. ...
KS4-Chemical-Reactions
... heating copper sulphate. Note the double arrow symbol in the chemical equation ...
... heating copper sulphate. Note the double arrow symbol in the chemical equation ...
advanced high density interconnect materials and techniques
... Along with interconnect materials, it is also essential that we have new interconnect techniques to form interconnects between multiple stacked layers. ...
... Along with interconnect materials, it is also essential that we have new interconnect techniques to form interconnects between multiple stacked layers. ...
Flux (metallurgy)

In metallurgy, a flux (derived from Latin fluxus meaning “flow”) is a chemical cleaning agent, flowing agent, or purifying agent. Fluxes may have more than one function at a time. They are used in both extractive metallurgy and metal joining.Some of the earliest known fluxes were carbonate of soda, potash, charcoal, coke, borax, lime, lead sulfide and certain minerals containing phosphorus. Iron ore was also used as a flux in the smelting of copper. These agents served various functions, the simplest being a reducing agent which prevented oxides from forming on the surface of the molten metal, while others absorbed impurities into the slag which could be scraped off the molten metal.As cleaning agents, fluxes facilitate soldering, brazing, and welding by removing oxidation from the metals to be joined. Common fluxes are: ammonium chloride or rosin for soldering tin; hydrochloric acid and zinc chloride for soldering galvanized iron (and other zinc surfaces); and borax for brazing or braze-welding ferrous metals. In the process of smelting, inorganic chlorides, fluorides (see fluorite), limestone and other materials are designated as ""fluxes"" when added to the contents of a smelting furnace or a cupola for the purpose of purging the metal of chemical impurities such as phosphorus, and of rendering slag more liquid at the smelting temperature. The slag is a liquid mixture of ash, flux, and other impurities. This reduction of slag viscosity with temperature, increasing the flow of slag in smelting, is the original origin of the word flux in metallurgy. Fluxes are also used in foundries for removing impurities from molten nonferrous metals such as aluminum, or for adding desirable trace elements such as titanium.In high-temperature metal joining processes (welding, brazing and soldering), the primary purpose of flux is to prevent oxidation of the base and filler materials. Tin-lead solder (e.g.) attaches very well to copper, but poorly to the various oxides of copper, which form quickly at soldering temperatures. Flux is a substance which is nearly inert at room temperature, but which becomes strongly reducing at elevated temperatures, preventing the formation of metal oxides. Additionally, flux allows solder to flow easily on the working piece rather than forming beads as it would otherwise.The role of a flux in joining processes is typically dual: dissolving of the oxides on the metal surface, which facilitates wetting by molten metal, and acting as an oxygen barrier by coating the hot surface, preventing its oxidation. In some applications molten flux also serves as a heat transfer medium, facilitating heating of the joint by the soldering tool or molten solder.Fluxes for soft soldering are typically of organic nature, though inorganic fluxes, usually based on halogenides and/or acids, are also used in non-electronics applications. Fluxes for brazing operate at significantly higher temperatures and are therefore mostly inorganic; the organic compounds tend to be of supplementary nature.