Chemistry Spell check on
... 1 Check that the answer sheet provided is for Chemistry Intermediate 2 (Section A). 2 For this section of the examination you must use an HB pencil and, where necessary, an eraser. 3 Check that the answer sheet you have been given has your name, date of birth, SCN (Scottish Candidate Number ...
... 1 Check that the answer sheet provided is for Chemistry Intermediate 2 (Section A). 2 For this section of the examination you must use an HB pencil and, where necessary, an eraser. 3 Check that the answer sheet you have been given has your name, date of birth, SCN (Scottish Candidate Number ...
Preparation of Sm–Ru bimetallic alloy films on Ru(0001) surface by
... The general method of preparing RM/TM surface alloy films is the deposition of the vapour obtained by evaporation of pure rare earth samples in ultrahigh vacuum. It is, however, difficult to prepare clean rare earth-based alloy films because of high reactivity of rare earth metals towards oxygen and ...
... The general method of preparing RM/TM surface alloy films is the deposition of the vapour obtained by evaporation of pure rare earth samples in ultrahigh vacuum. It is, however, difficult to prepare clean rare earth-based alloy films because of high reactivity of rare earth metals towards oxygen and ...
Chap 9 Redox Review Q`s
... (ii) State the products formed and give equations showing the reactions at each electrode. ...
... (ii) State the products formed and give equations showing the reactions at each electrode. ...
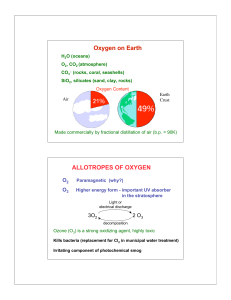
The representative Elements: Groups 1A – 4A
... • Lightweight metal (density = 2.70 g/cm3); • Forms strong, lightweight alloys with copper and magnesium for aircraft bodies and parts; • High resistance to corrosion - extensively used to make beverage containers (soda drinks cans); • Also used as reducing agent in the fuel during space shuttle lau ...
... • Lightweight metal (density = 2.70 g/cm3); • Forms strong, lightweight alloys with copper and magnesium for aircraft bodies and parts; • High resistance to corrosion - extensively used to make beverage containers (soda drinks cans); • Also used as reducing agent in the fuel during space shuttle lau ...
Reaction of niobium with water
... Current methodology involves the separation of tantalum from these acid solutions using a liquid-liquid extraction technique. In this process tantalum salts are extracted into the ketone MIBK (methyl isobutyl ketone, 4-methyl pentan-2-one). The niobium remains in the HF solution. This solvent extrac ...
... Current methodology involves the separation of tantalum from these acid solutions using a liquid-liquid extraction technique. In this process tantalum salts are extracted into the ketone MIBK (methyl isobutyl ketone, 4-methyl pentan-2-one). The niobium remains in the HF solution. This solvent extrac ...
Stud bump bonding
... Al can be shaped into fine wires applied for wedgewedge Al bonding. Al immediately forms oxides in air. Aluminum is not a readily solderable surface, neither wettable nor bondable by most solders. Aluminum may corrode over time when not protected from the environment. Low melting temperature (660°C) ...
... Al can be shaped into fine wires applied for wedgewedge Al bonding. Al immediately forms oxides in air. Aluminum is not a readily solderable surface, neither wettable nor bondable by most solders. Aluminum may corrode over time when not protected from the environment. Low melting temperature (660°C) ...
CHEMISTRY OF MAIN GROUP ELEMENTS Classification -1 s
... Like alkali metals they also predominantly form ionic compounds. Be is slightly covalent in nature due to the small size of the atom therefore the polarising power is high. Be resembles Al due to diagonal relationships ...
... Like alkali metals they also predominantly form ionic compounds. Be is slightly covalent in nature due to the small size of the atom therefore the polarising power is high. Be resembles Al due to diagonal relationships ...
Jay Shri Harsiddhi Mataji Pick the date
... without Whiteware having good strength, translucency and very low porosity can be obtained by firing the products at 1450-15000C. Whitewares contain refractory body and glassy coating known as the glaze. There is a corresponding variation in the degree vitrification due to different amount of fluxes ...
... without Whiteware having good strength, translucency and very low porosity can be obtained by firing the products at 1450-15000C. Whitewares contain refractory body and glassy coating known as the glaze. There is a corresponding variation in the degree vitrification due to different amount of fluxes ...
chapter 8 ceramic/metal nanocomposites
... the system by converting it into ice in a condenser, operating at very low temperatures, outside the freeze drying chamber. Supercritical drying (superheated steam drying) involves steam drying of products containing water. This process is feasible because water in the product is boiled off, and joi ...
... the system by converting it into ice in a condenser, operating at very low temperatures, outside the freeze drying chamber. Supercritical drying (superheated steam drying) involves steam drying of products containing water. This process is feasible because water in the product is boiled off, and joi ...
Heat Treating of Non Ferous Alloys
... Dispersion hardening Conventional strengthening mechanisms, such as cold working and precipitation hardening, are ineffective at high temperature, owing to the effects of recrystallization, and particle coarsening and dissolution respectively. Applications require materials with a high thermal condu ...
... Dispersion hardening Conventional strengthening mechanisms, such as cold working and precipitation hardening, are ineffective at high temperature, owing to the effects of recrystallization, and particle coarsening and dissolution respectively. Applications require materials with a high thermal condu ...
CC-80 art 6
... Kaolinite´s formula is Al2Si2O5(OH)4. Humans have been using this material in different ways from time immemorial. In the fifteen century, porcelain made of ceramics with a high content of kaolin acquired great fame among the nobility. Nowadays, the main consumer of kaolin is the paper industry, whi ...
... Kaolinite´s formula is Al2Si2O5(OH)4. Humans have been using this material in different ways from time immemorial. In the fifteen century, porcelain made of ceramics with a high content of kaolin acquired great fame among the nobility. Nowadays, the main consumer of kaolin is the paper industry, whi ...
Sec 7.3
... All metals except for gold and copper are silver-grey in color. Malleable – when a substance can be hammered or shaped without breaking Ductile – substances that can be drawn into wires Conductors – can carry electrons or electricity usually well and can also transfer heat well Solid- all metals (ex ...
... All metals except for gold and copper are silver-grey in color. Malleable – when a substance can be hammered or shaped without breaking Ductile – substances that can be drawn into wires Conductors – can carry electrons or electricity usually well and can also transfer heat well Solid- all metals (ex ...
periodic table - Mesa Community College
... OBJECTIVES: To develop the skills needed to write correct formulas from names and correct names from formulas. DISCUSSION: In the course of your study of chemistry you will encounter numerous compounds that you will have to recognize by formula and/or name. Either you are going to have to spend a gr ...
... OBJECTIVES: To develop the skills needed to write correct formulas from names and correct names from formulas. DISCUSSION: In the course of your study of chemistry you will encounter numerous compounds that you will have to recognize by formula and/or name. Either you are going to have to spend a gr ...
Open Access proceedings Journal of Physics: Conference series
... Cr(III) – 3p63d3). When a core electron vacancy is made by photoionization, there can be coupling between the unpaired electron in the core with the unpaired electrons in the outer shell. This can create a number of final states which will be seen in the photoelectron spectrum. For some materials, w ...
... Cr(III) – 3p63d3). When a core electron vacancy is made by photoionization, there can be coupling between the unpaired electron in the core with the unpaired electrons in the outer shell. This can create a number of final states which will be seen in the photoelectron spectrum. For some materials, w ...
CHM 212 - The Federal University of Agriculture, Abeokuta
... COURSE DESCRIPTION CHM 212: Basic Inorganic Chemistry II ...
... COURSE DESCRIPTION CHM 212: Basic Inorganic Chemistry II ...
Lecture 2
... Hard acids or bases are small and non-polarizable Hard acids are cations with high positive charge (3+ or greater), or cations with d electrons not available for π-bonding Soft acids are cations with a moderate positive charge (2+ or lower), Or cations with d electrons readily availbale for π-bondin ...
... Hard acids or bases are small and non-polarizable Hard acids are cations with high positive charge (3+ or greater), or cations with d electrons not available for π-bonding Soft acids are cations with a moderate positive charge (2+ or lower), Or cations with d electrons readily availbale for π-bondin ...
Biologically Important Inorganic Elements Occurrence and Availability
... Bioinorganic chemistry is concerned with the roles of inorganic elements in biological processes. In CHEM 646, we will apply fundamental principles of inorganic chemistry, particularly transition metal coordination chemistry and ligand field theory, to understand the structure and function of me ...
... Bioinorganic chemistry is concerned with the roles of inorganic elements in biological processes. In CHEM 646, we will apply fundamental principles of inorganic chemistry, particularly transition metal coordination chemistry and ligand field theory, to understand the structure and function of me ...
p-Block Elements, Part 1
... Silicate Minerals: [Si2O52-]n, SiO44Sand: SiO2 (this is also quartz). With aluminum in aluminosilicates (clay, feldspars). Prepared by: SiO2(s) + 2C(s) → Si(l) + 2CO(g) sand coke ...
... Silicate Minerals: [Si2O52-]n, SiO44Sand: SiO2 (this is also quartz). With aluminum in aluminosilicates (clay, feldspars). Prepared by: SiO2(s) + 2C(s) → Si(l) + 2CO(g) sand coke ...
ch14
... Carbon forms predominantly covalent bonds, but the larger members of the group form bonds with increasing ionic character. Elements of this group also exhibit multiple oxidation states. Lower oxidation states become more prominent down the group. Pb and Sn show more metallic character in their lower ...
... Carbon forms predominantly covalent bonds, but the larger members of the group form bonds with increasing ionic character. Elements of this group also exhibit multiple oxidation states. Lower oxidation states become more prominent down the group. Pb and Sn show more metallic character in their lower ...
Project_FEA.doc
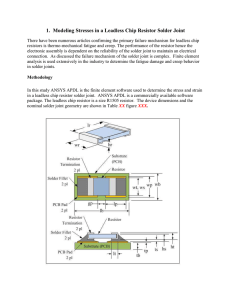
... 1. Modeling Stresses in a Leadless Chip Resistor Solder Joint There have been numerous articles confirming the primary failure mechanism for leadless chip resistors is thermo-mechanical fatigue and creep. The performance of the resistor hence the electronic assembly is dependent on the reliability o ...
... 1. Modeling Stresses in a Leadless Chip Resistor Solder Joint There have been numerous articles confirming the primary failure mechanism for leadless chip resistors is thermo-mechanical fatigue and creep. The performance of the resistor hence the electronic assembly is dependent on the reliability o ...
pblock - Chemistry Courses
... Silicate Minerals: [Si2O52-]n, SiO44Sand: SiO2 (this is also quartz). With aluminum in aluminosilicates (clay, feldspars). Prepared by: SiO2(s) + 2C(s) Si(l) + 2CO(g) sand coke ...
... Silicate Minerals: [Si2O52-]n, SiO44Sand: SiO2 (this is also quartz). With aluminum in aluminosilicates (clay, feldspars). Prepared by: SiO2(s) + 2C(s) Si(l) + 2CO(g) sand coke ...
Interfacial Reaction between Alumina and Cu
... filler metals, it is very important to understand which reaction occurs and how the reaction products grow at the interface. In this research, the interfacial reaction and the kinetics of reaction products growth in alumina brazing utilizing Cu-Ti filler metals were investigated. Cu-Ti filler metals ...
... filler metals, it is very important to understand which reaction occurs and how the reaction products grow at the interface. In this research, the interfacial reaction and the kinetics of reaction products growth in alumina brazing utilizing Cu-Ti filler metals were investigated. Cu-Ti filler metals ...
Date - PetyaPisanScienceAQ
... Physical property: Traditionally, metals have certain characteristic physical properties: they are usually shiny (they have metallic luster), have a high density, are ductile and malleable, usually have a high melting point, are usually hard, are usually a solid at room temperature and conduct elect ...
... Physical property: Traditionally, metals have certain characteristic physical properties: they are usually shiny (they have metallic luster), have a high density, are ductile and malleable, usually have a high melting point, are usually hard, are usually a solid at room temperature and conduct elect ...
Formation of intermetallic compounds upon cooling of Sn1
... Upon cooling below the liquidus temperature the crystalline phase appears. The analysis of the diffraction patterns recorded at a temperature corresponding to a two-phase mixture and at room temperature revealed the crystallization of ZrSn2 and tin. These two phases exist also at room temperature, w ...
... Upon cooling below the liquidus temperature the crystalline phase appears. The analysis of the diffraction patterns recorded at a temperature corresponding to a two-phase mixture and at room temperature revealed the crystallization of ZrSn2 and tin. These two phases exist also at room temperature, w ...
Flux (metallurgy)

In metallurgy, a flux (derived from Latin fluxus meaning “flow”) is a chemical cleaning agent, flowing agent, or purifying agent. Fluxes may have more than one function at a time. They are used in both extractive metallurgy and metal joining.Some of the earliest known fluxes were carbonate of soda, potash, charcoal, coke, borax, lime, lead sulfide and certain minerals containing phosphorus. Iron ore was also used as a flux in the smelting of copper. These agents served various functions, the simplest being a reducing agent which prevented oxides from forming on the surface of the molten metal, while others absorbed impurities into the slag which could be scraped off the molten metal.As cleaning agents, fluxes facilitate soldering, brazing, and welding by removing oxidation from the metals to be joined. Common fluxes are: ammonium chloride or rosin for soldering tin; hydrochloric acid and zinc chloride for soldering galvanized iron (and other zinc surfaces); and borax for brazing or braze-welding ferrous metals. In the process of smelting, inorganic chlorides, fluorides (see fluorite), limestone and other materials are designated as ""fluxes"" when added to the contents of a smelting furnace or a cupola for the purpose of purging the metal of chemical impurities such as phosphorus, and of rendering slag more liquid at the smelting temperature. The slag is a liquid mixture of ash, flux, and other impurities. This reduction of slag viscosity with temperature, increasing the flow of slag in smelting, is the original origin of the word flux in metallurgy. Fluxes are also used in foundries for removing impurities from molten nonferrous metals such as aluminum, or for adding desirable trace elements such as titanium.In high-temperature metal joining processes (welding, brazing and soldering), the primary purpose of flux is to prevent oxidation of the base and filler materials. Tin-lead solder (e.g.) attaches very well to copper, but poorly to the various oxides of copper, which form quickly at soldering temperatures. Flux is a substance which is nearly inert at room temperature, but which becomes strongly reducing at elevated temperatures, preventing the formation of metal oxides. Additionally, flux allows solder to flow easily on the working piece rather than forming beads as it would otherwise.The role of a flux in joining processes is typically dual: dissolving of the oxides on the metal surface, which facilitates wetting by molten metal, and acting as an oxygen barrier by coating the hot surface, preventing its oxidation. In some applications molten flux also serves as a heat transfer medium, facilitating heating of the joint by the soldering tool or molten solder.Fluxes for soft soldering are typically of organic nature, though inorganic fluxes, usually based on halogenides and/or acids, are also used in non-electronics applications. Fluxes for brazing operate at significantly higher temperatures and are therefore mostly inorganic; the organic compounds tend to be of supplementary nature.