* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Unit - II Electrochemistry
Patch clamp wikipedia , lookup
Oil refinery wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Multielectrode array wikipedia , lookup
Double layer forces wikipedia , lookup
Coordination complex wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Flux (metallurgy) wikipedia , lookup
Metalloprotein wikipedia , lookup
Metallic bonding wikipedia , lookup
Surface properties of transition metal oxides wikipedia , lookup
History of electrochemistry wikipedia , lookup
Cathodic protection wikipedia , lookup
Electrolysis of water wikipedia , lookup
Liquid-feed flame spray pyrolysis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
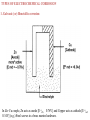
CY6251-Unit – II ELECTROCHEMISTRY AND CORROSION Lecturer-II /I Electrochemical cells and its types. Electrode potential –origin – oxidation and reduction potent INTRODUCTION • Electrochemistry is a branch of chemistry. • It deals with the chemical reactions produced by passing electric current through an electrolyte or production of electric current through a chemical reaction Conductors Conductor is a material which allows free flow of electricity. Example: All metals, graphite, fused salts, solution of electrolytes Non-conductors (Insulators) Insulators are materials which donot conduct electrical current Example: Wood, plastics, most of non metals. Types of conductors (i) Metallic conductors : The solid material, which conduct electric current due to the movement of electron from one end to the other end without producing chemical reaction. Examples : All metals & graphite. (ii) Electrolytic conductors : They conduct electric current due to the movement of ions from one electrode to another electrode in solution or in fused state. This process is accompanied by a chemical reaction. Examples : Metal ions dissolved solvent Cell Terminology 1. Current: Flow of electrons through a conductor. 2. Electrode: Electrode is a material (rod, bar, strip) which conducts electrons. 3. Anode: Electrode at which oxidation occurs. 4. Cathode: Electrode at which reduction occurs. 5. Electrolyte: Water soluble substance forming ions in solution and conducts electric current 6. Anode compartment: Compartment of the cell in which the oxidation half reaction occurs. It contains the anode 7. Cathode compartment: Compartment of the cell in which the reduction half reaction occurs. It contains the cathode 8. Half–cell: It is the part of a cell, which contains an electrode dipped in an electrolyte. If oxidation occurs in this half-cell, then it is called the oxidation half cell. If reduction occurs at the cell, it is called the reduction half-cell. 9. Cell: Device consisting of two half cell. The two half cells are connected through one wire. 10.Salt bridge: Contains solutions of a salt (KNO3 or NH4NO3) that literally serve as a bridge to completed the circuit, maintain electro neutrality of electrolyte and minimize. For precise measurement of potential a salt bridge is used. TYPES OF CELLS A cell is a device consisting of two half cells. Each half cell consists of an electrode dipped in an electrolyte solution. The two half cells are connected through one wire. S.No Electrolytic cell Electrochemical cell 1 Electrical energy converted to Chemical energy converted to electrical chemical energy. energy. Example: Electrolysis, electroplating. Example: Daniel Cell 2 3 4 5 6 Anode carries +ve charge. Cathode carries – ve charge. Electrons are supplied to the cell from an external source. Amount of electricity is measured by coulometer Extent of chemical change is governed by Faraday’s laws. Anode carries –ve charge Cathode carries +ve charge. Electrons are drawn from the cell Emf produced is measured by potentiometer Emf of the cell depends on the concentration of the electrolyte and the nature of the electrode. Electrolytic cell - Example : Electrolysis of HCl. Mechanism At anode : 2Cl– Cl2 + 2e (oxidation) At cathode : 2H+ + 2e– H2 (reduction) Electrochemical cell - Example : Daniel cell Electromotive Force (emf) (at anode) (at cathode) Cu2+ + Zn Zn2+ + Cu (net cell reaction) • At anode : Oxidation of Zn to Zn2+ place with the liberation of electrons. • At cathode : Reduction of Cu2+ to Cu place by the acceptance of electrons. The electrons liberated in oxidation reaction flow through external wire and are consumed by the copper ions at the cathode. • Salt bridge : It consists of a U-tube containing a saturated solution of KCl or (NH4)2NO3 agar–agar gel. It connects the two half cells. Functions i. It eliminates liquid junction potential. ii. It provides a path for the flow of electrons between two half cells. Representation (Cell diagram) i. Galvanic cell consists of two electrodes, anode and cathode ii. Anode is written on the LHS and cathode on RHS iii. The anode is written with the metal first and then the electrolyte which are separated by a vertical line Examples : Zn/Zn2+ (or) Zn/ZnSO4 iv. The cathode is written with the electrolyte first and then the metal. Examples : Cu2+/Cu (or) CuSO4/Cu v. The two half cells are separated by a salt bridge, which is indicated by two vertical lines. Cell is represented as Zn/ZnSO4 (1M) // ) CuSO4 (1M) /Cu ELECTRODE POTENTIAL ORIGIN OF ELECTRODE POTENTIAL When a metal (M) is placed in a solution of its own salt (Mn+) one of the two processes are possible (i) Metal atoms go into solution in the form of ions. M Mn+ + ne- (Oxidation) Example : e- (ii) Metal ions from solution may deposit on the metal Mn+ + ne- M (reduction) e- Zn Cu Zn Cu 2+ Zn2+ + Cu2+ Zn 2++ 2e- 2eCu Electrode Potential At equilibrium, the potential difference becomes a constant value which is known as the electrode potential of the metal. Thus the tendency of the electrode to lose electrons is called Oxidation potential and tendency of an electrode to gain electrons is called reduction potential. Single electrode potential (E) : It is the tendency of a metallic electrode to lose or gain electrons when it is in contact with a solution of its own salt. Standard electrode potential (Eo) : It is the tendency of a metallic electrode to lose or gain electrons when it is in contact with a solution of its own salt of 1M concentration at 25oC. Lecturer-II /II Measurement Of Single Electrode Potential and its applications. Electro chemical series and its significances MEASUREMENT OF SINGLE ELECTRODE POTENTIAL It is impossible to evaluate the absolute value of a single electrode potential. Using reference electrode. Reference (or) Standard electrode The potential of unknown electrode can be measured by coupling it with another electrode, called reference electrode whose electrode potential is already known. Examples : Standard hydrogen electrode, Standard calomel electrodes. Standard hydrogen electrode (SHE) It is also called as Primary reference electrode because. The potential developed by this electrode is arbitrarily fixed as zero Construction • It consists of a platinum foil that is connected to a platinum wire sealed in a glass tube. •The Pt foil is dipped in 1M HCl. H2 gas of 1 atm pressure is passed through the side of glass tube. • The standard electrode potential of SHE is arbitrarily fix as zero Pt , H2 (1atm) / H+(1M) ; E0 = 0V H 2 (g) 2H + 2e Limitations (or) drawbacks of SHE • It is difficult to get pure hydrogen gas. • The pressure of hydrogen is to be kept 1 atm all the time. • It is difficult to set up and transport. • Hydrogen gas reduces many ions like Ag+ and affects compounds of Hg, Ag etc • A large volume of test solution is required. • It cannot be used in solutions of redox systems, the solution may poison platinum surface. Saturated calomel electrode (SCE) (Secondary reference electrode) • Glass tube containing pure Hg at the bottom over which mercurous chloride is placed. The remaining portion of the tube is filled with saturated solution of KCl. • The bottom of the tube is sealed with a platinum wire. The side tube is used for making electrical contact with a salt bridge. Hg | Hg2 Cl2(s) | KC | (Saturated, Solution) Eº = 0.2422V 2Hg(l ) + 2Cl Hg 2Cl2 (s )+ 2e HgCl2 +2e 2 Hg +Cl KCl (v) 0.1N 0.3335 V 1N 0.281 V Saturated 0.2422 V Measurement of single electrode potential using a reference electrode (saturated calomel electrode) Hg 2 Cl 2 (s) 2Hg ( ) 2Cl The emf of the cell is measured using a potentiometer. The value of Ecell = 1.0025 volt. E = E oright----- Eoleft E = E calo --- EoZn E oZn = E ocal ---- Eocell = + 0.2422 – 1.0025 = – 0.7603 V ELECTROCHEMICAL SERIES (OR) EMF SERIES Significance of emf series (or) Application of electrochemical series • Calculation of Standard emf of a Cell : We can calculate the standard emf of a cell, if the standard electrode potential values are known (Ecell =ERHE –ELHE) • Relative ease of oxidation or reduction (a) Fluorine has higher +ve value of standard reduction potential (+2.87V) and shows higher tendency for reduction. (b) Lithium has highest – ve value (–3.02V) and shows higher tendency towards oxidation. • Displacement of one element by the other Metals with a lower reduction potential will displace metals with a higher reduction potential from their salt solution (Copper will displace silver from its solution). Example : Zn(-0.76V) will displace copper (+0.34V) from its solution Zn + CuSO4ZnSO4 + Cu • Determination of equilibrium constant (K) for a reaction Standard electrode potentials are used to determine the equilibrium constants as follows: Go = ln K 2.303 RT log K log 𝐾 = 𝐺0 2.303 𝑅𝑇 = 𝑛𝐹𝐸0 [𝐺0𝑛𝐹𝐸0] 2.303 𝑅𝑇 • Hydrogen displacement behavior Metals with negative reduction potential (metals placed above H2) in emf series will displace hydrogen from dilute acids solutions. Example: Zn (-0.76 V) will displace H2 from dilute acids whereas, silver (0.8) cannot • Zn + H2SO4 ZnSO4 + H2 • Ag + H2SO4 No reaction • Predicting the spontaneity of redox reactions If Eo of a cell is positive the reaction is spontaneous. If Eo of a cell is negative the reaction is not feasible. Lecturer-II / III NERNST EQUATION FOR ELECTRODE POTENTIAL The potential of any electrode system (E) depends on (i) nature of the metal / element (ii) temperature and concentration of the electrolyte. The functional dependence of potential of any electrode system (E) on these factors is given by Nernst equation E = E 0 + (RT / nF) ( log Mn+) Potential evolves from the inter-conversion between chemical and electrical forms of energy. The equation pertaining to these two forms of energy is G = G0 + RT ln K ----- (1) where G, G0, K and R are respectively The free energy change for any chemical to electrical energy conversion process is given by the equation G = - nFE ----- (2) where E is the potential of the electrode system, F – Faraday’s constant = 96496 or 96500 coulombs and ‘n’ is the number of electrons transferred between the element and the ion which are in equilibrium. Combining equations (1) and (2), we get -nFE = - nFE0 + RT ln K ----- (3) where E0 is the standard electrode potential of the electrode system. Consider the reduction reaction Mn+ + n e → M Equation (3) becomes E = E0 - (RT / nF) ln (aproducts / areactants ) ----- (4) where ‘a’ is the activity of the species. The activity of a (homogenous / uniform) solid is taken as unity, that of the electrolyte expressed in terms of the concentration and that of a gas (or gaseous mixture) expressed in terms of pressure (or partial pressure) of the gas. Equation (4) can be written as E = E0 - (RT / nF) ln (aM / aMn+ ) (or) E = E0 + (RT / nF) ln [Mn+] ----- (4) where [Mn+] is the concentration of the electrolyte / metal ion in solution. i.e. E = E0 + (RT / nF) ln [Mn+] ----- (5) substituting the values of R= 8.314 Joules, F = 96500 Coulombs and introducing the factor 2.303 to convert natural logarithms to common logarithms, equation (5) becomes E = E0 + (0.059 / n) log [Mn+] ----- (6) Equations (5) and (6) are the two forms of Nernst equation which gives the dependence of electrode potential on the factors mentioned Applications : • It is used to calculate the electrode potential of unknown metal. • To know the emf and polarity of electrodes in an electrochemical cell. • The corrosion tendency of metals in a given set of environmental • conditions can be predicted. Lecturer-II /IV Corrosion-Introduction, types Mechanism of dry and wet corrosion “Corrosion is defined as the gradual destruction of metals or alloys by the chemical or electrochemical reaction with its environment.” Causes of corrosion occurs Most of the metals (except noble metals) naturally exist in combined form. During metallurgy the metal are extracted from their ores by reduction process. In the pure metallic state, the metals are unstable and considered to be in the excited (higher energy) state. Therefore, the extracted metals have a tendency to go to thermodynamically stable (lower energy) state, which is otherwise known as corrosion. Thus, corrosion is a process “reverse of extraction of metals.” Consequences of corrosion • The efficiency of machine will be lost due to the loss of useful properties of metal. • The products gets contaminated due to corrosion. • Increase in maintenance and production cost. • Preventive maintenance like metallic (or) organic coating is required. • Toxic products are released. TYPES OF CORROSION Based on the environment, corrosion is classified into two types. (i) Dry (or) Chemical corrosion (ii) Wet or electro chemical corrosion Dry (or) Chemical corrosion • It is due to the direct chemical attack on metals by atmospheric gases such as oxygen, carbon-di-oxide, hydrogen sulphide, etc. • It follows adsorption mechanism. Corrosion product accumulate in the same spot of corrosion. Example : Tarnishing of silver in H2S gas, Action of dry HCl on iron surface. There are three main types of chemical corrosion. • Oxidation corrosion • Corrosion by hydrogen • Liquid–Metal corrosion Oxidation corrosion 2M 2Mn+ + 2ne– At the oxide scale/environment interface n O + 2ne– nO2– 2 2 The over all reaction 2M + n2 O2 2Mn+ + nO2– Metal – Oxide scale The nature of oxide film formed on the metal surface plays an important role in oxidation corrosion. Nature of oxide layer and future course of corrosion 1. Stable : The oxide layer formed in some cases stick firmly to the parent metal surface. Such layers naturally do not allow penetration of oxygen to the underlying metal surface and thus act as protective films. Example : Al, Sn, Pb, Cu, etc. 2. Unstable : In case of some metals, elemental or uncombined state is naturally more stable than the combined state such as oxide, sulphide, sulphate, etc. Metal oxide Metal + Oxygen Only forward reaction is favoured. So, oxidation corrosion is not possible in those metals. Example : Ag, Au and Pt 3. Volatile : In some metals the oxide layers formed are volatile. They leave the metal surface as soon as they are formed. That means, the fresh metal surface is kept exposed all the time for further attack. This makes the corrosion continuous and rapid. Example : Mo (MoO3 is volatile). 4. Porous : The oxide layer formed in some cases are porous. Atmospheric oxygen gets free access to underlying metal surface. Consequently corrosion goes non-stop till the entire metal is converted into its oxide. Example : Alkali metals (Li, Na, Ka etc.) Pilling–Bed worth rule An oxide layer is protective (or) non-porous, if the volume of the metal oxide formed is atleast as great as the volume of the metal from which it is formed. Protective or non-porous : Example : Al, Sn, Pb, Cu, etc. An oxide layer is non-protective (or) porous, if the volume of the metal oxide formed is less than the volume of the metal from which it is formed. VM 2 O n VM Non-protective or Porous : Example :Alkali metals : Li, Na, K, etc. Alkaline earth metals : Mg, Ca, Si etc. Corrosion by hydrogen Atomic hydrogen (H) can more easily penetrate steel and other metals than molecular hydrogen (H2), which is chemically more active. (i) Hydrogen embrittlement : At ordinary temperatures, some reactions produce atomic hydrogen which attack metals and reduce their strength. Example : Aqueous solution of H2S liberates atomic hydrogen at iron-surfaces. Fe + H2S FeS + 2H (ii) Decarburization: At high temperatures, atomic hydrogen is produced by thermal dissociation. H2 H + H 4H + C (in steel) CH4 The gas collects in gaps and voids and causes blisters and fissures. Consequently the metal becomes weak. By other gases : Gases like, SO2, CO2, H2S, F2 and Cl2 are also corrosive. c) Liquid–Metal corrosion This is brought about by chemical action of flowing liquid metals at high temperatures over solid metal or alloy. The corrosion involves either dissolution of a solid metal by a liquid metal or internal penetration of the liquid metal into the solid metal. Example : Coolant (sodium metal) leads to corrosion of cadmium in nuclear reactor. Wet (or) Electrochemical Corrosion This type of corrosion occurs when, Metal is in contact with electrolyte or varying concentration of oxygen. Two dissimilar metals or alloys are immersed or partially dipped in a solution. At anode – Oxidation takes place (loss of electron) At Cathode – reduction takes place (gain of electron) i.e. It involves flow of current between anodic and cathodic areas. Mechanism of electrochemical corrosion Acidic : 2H+ + 2e– H2 Basic: 2 O2 + 2e– + H2O 2OH– The Overall reaction is Fe + 2H+ Fe + H2 ii) Absorption of oxygen (Rusting of Iron) type corrosion This type of corrosion occurs when metal comes in contact with neutral or alkaline medium. Over all : Fe2+ + 2OH– Fe(OH)2 (Ferrous hydroxide) 4Fe(OH)2 + O2 + 2H2O 4 Fe(OH)3 (Ferric hydroxide)(Rust) TYPES OF ELECTROCHEMICAL CORROSION 1. Galvanic (or) Bimetallic corrosion In Zn–Cu couple, Zn acts as anode [E oZn 0.76V] and Copper acts as cathode [E ored 0.34V] (e.g.) Steel screws in a brass marine hardware. 2. Differential aeration (or) Concentration cell corrosion This type of corrosion occurs when metal is exposed to varying concentration of oxygen or an electrolyte. At anode (less aerated part) corrosion occurs M M2+ + 2e– At cathode (more aerated part) OH– ions are produced. 1 O + H O + 2e– 2OH– 2 2 2 Pitting corrosion Pitting is a localised attack, resulting in the formation of a hole around which the metal is relatively unaffected. The mechanism involved is differential aeration corrosion. Fe Fe 2 2e [Anode] 1 2 O2 + 2e + H2O 2OH [Cathode] Fe +1/2 O2 + H2O Fe(OH)2 [Over all Reaction] 4) Water line corrosion Difference between Chemical and Electrochemical corrosion S.No 1. 2. 3. 4. 5. Chemical or Dry corrosion Electrochemical or Wet corrosion It occurs in the presence ofmoisture or It occurs in dry condition electrolyte. It involves the direct chemical It involves the setting up of large number of attack of metalsby dry gases. electrochemical cells. Corrosion products accumulatein Corrosion occurs at the anode, products the same spot of corrosion. gather at the cathode. Corrosion is uniform throughout Corrosion is not uniform, which depends on the surface anodic and cathodic. It follows mechanism of electrochemical It follows adsorption mechanism. reactions Lecturer-II / V FACTORS INFLUENCING THE RATE OF CORROSION The rate and extent of corrosion depends on 1. 2. Nature of the metal Nature of the environment 1. Nature of the metal a. Position in galvanic series b. Over voltage c. Purity of metal d. Relative areas of anode and cathode e. Nature of oxide film (surface film) f. Nature of corrosion product g. Physical state of metal a) Position in galvonic series (b) Over voltage If hydrogen overvoltage of the cathodic metal is very high, corrosion rate is lesser. When the voltage in a circuit of part of it raised above its upper design limit - over voltage. 1/ corrosion (i.e.) Over voltage of a metal (e.g.) When Zn dipped in 1 N H2SO4, the hydrogen over voltage is 0.70 volts. c) Purity of metal: The 100% pure metal will not undergo any types of corrosion. e.g. %Purity of Zn Corrosion rate 99.9999 1 99.99 99.95 2650 5000 (d) Relative areas of anode and cathode : Corrosion is severe, if the anodic area is smaller than cathodic area. (e) Nature of oxide film (surface film): The nature of surface film formed decides the extent of corrosion. (f) Nature of corrosion product The corrosion product formed is soluble in corroding medium, the corrosion rate will be greater. Similarly, if the corrosion product is volatile (e.g.) MoO3, SnCl4 both are volatile corrosion rate will be faster. (g) Physical state of metal The rate of corrosion depends on physical state of metal (i.e) grain size, stress, orientation of crystals. The smaller the grain size of metal or alloy greater will be its solubility and corrosion. Areas under stress (bends, joints, rivets) tend to be anodic and leads to corrosion. Nature of the environment (a) Temperature: The rate of corrosion is directly proportional to temperature. (b) Humidity: The greater is the humidity, the greater is the rate and extent of corrosion. This is due to the fact that moisture acts as a solvent for O2, H2S, SO2 and NaCl etc., resulting an electrochemical cell. (c) Presence of impurity in atmosphere: Presence of gases like CO2, H2S, SO2 and fumes of HCl, H2SO4 makes the environment acidic so corrosion is accelerated. (d) Presence of suspended particles: If the environment contains chemically active particles (NaCl, (NH4)2SO4) they absorb moisture and acts as strong electrolytes. So rate of corrosion is high. (e) Nature of ions present: Presence of silicate (anion) in the medium leads to insoluble products (silica gel), which prevents corrosion. Presence of Cl ions in electrolyte destroys the protective films and enhances corrosion. If ammonium salts are present in corroding medium, that will lead to corrosion of many metals. (f) Effect of pH: Generally acidic medium is more corrosive than alkaline medium. The corrosion of iron in oxygen free water is very slow upto pH = 5. But in presence of oxygen the corrosion rate of iron is very high at pH = 5. But at pH = 4 the corrosion rate of iron is considerably increased due to the change in oxidation states of iron from Fe2+ to Fe3+. Zinc which is readily corroded in acidic solution suffers very less corrosion in alkaline medium i.e. pH = 11. Aluminium has less corrosion at pH = 5.5 which corrodes rapidly at pH = 8.5. Lecturer-II /VI Corrosion control-selection and designing of materials, Sacrificial and Imparessed current cathodic protection. The corrosion process depends on the metal and the environment. So control measures are aimed at the metal and environment. (i.e.) (i) Metal based controlled measures and (ii) Environment based controlled measures. Metal based controlled measures 1. Proper choice (or) Selection of metal 2. Proper design 3. Cathodic protection Proper choice (or) Selection of metal • Before fabrication of equipments, proper selection of metal or alloy have to be studied according to the environmental conditions. • Corrosion can be avoided by replacing the metal parts by plastics, rubbers, ceramics etc. as far as possible. Metals Not affected by Stainless steel HNO3 Steel Con.H2SO4 Ti Hot acids and alkalies Proper design Design play an important role in corrosion control. Important design principle are • Avoid direct contact of dissimilar metals by using adhesives. • When two dissimilar metals are in contact, the anodic part should have large area than the cathodic part. • Care must be taken in designing an equipment so that the liquid can be drained off completely. • Crevice corrosion and caustic embrittlement can be prevented by avoiding hair cracks, gaps and protruding parts (rivets etc). • Single crystalline solids avoid inter granular corrosion. • Stress corrosion can be avoided by heat treatment technique (annealing). • Heterogeneity is reduced by using pure metals, which decreases corrosion rate. (c) Cathodic protection In cathodic protection, the corroding metal is forced to act as cathode. • Sacrificial anodic protection • Impressed current cathodic protection Advantages (i) Sacrificial anodic protection No need of external power supply. Installation and maintenance cost is low. Limitations Not suited for large objects due to limited driving potential. Uncoated parts cannot be protected. (ii) Impressed current cathodic protection In this method an impressed current is applied in the opposite direction of the corrosion current to nullify it, which converts corroding metal from anode to cathode. Advantages • Larger objects can be protected due to larger driving voltage. • Uncoated parts can be protected. Limitations • Larger installation cost. • Higher maintenance cost. Lecturer-II /VII Paints-Constitution and functions PROTECTIVE COATINGS: Protective coatings are the physical barrier between the metal surface and the environment which prevents corrosion. They are also used for decorative purpose and to impart some special properties such as hardness, electrical properties, oxidation resistance, and thermal insulation. Paints: It is a mechanical dispersion of one or more finely divided pigments in a vehicle of medium. The medium consists of non volatile, film forming materials like drying oils or resins in suitable volatile solvents (thinners).When paint is applied to a metal surface, the thinner evaporates and the vehicle undergoes slow oxidation forming a pigment film Requisites (characteristics) of a good paint • It should spread easily on the metal surface • It should adhere well to the surface • It should not crack on drying • It should give stable and decent colour • It should be corrosion and water resistant Constituents of a paint and their functions a)Pigments : Pigments are solid and colour producing substances in the paint Sl.No. Colour 1. 2. White pigments Black pigments 3. 4. 5. Red pigments Blue pigments Green pigments Compounds used White lead (2PbCO3.Pb(OH)2 Lithophone (75% BaSO4 ; 25% ZnS), TiO2 Lamp black, carbon black Venetian red (Fe2O3 and CaSO4) Indian red (Fe2O3) Prussian blue Fe4[Fe(CN)6] Chromium oxide Functions • It provides colour and opacity to the film • It gives strength to the film B ) Vehicle or drying oil • It is a non volatile portion of the medium. It is a film forming constituent of the paint. Examples : Linseed oil, dehydrated castor oil. Functions • They form protective film by the oxidation and polymerization of the vegetable oil. • They hold pigment particles together on the metal surface. • They impart water repellency, toughness and durability to the film. c) Thinners ( solvents ) It is a volatile portion of the medium which easily evaporates after the application of the paints. Examples : Acetone, Diethylether, Chloroform, etc. Functions It reduces the viscosity of the paint It acts as a dispersing medium for the oil and pigments It increases the elasticity of the film It increases penetrating power of the vehicle d) Extenders (Fillers): They are low refractive indices materials which increases the durability of paint. Examples : Talc, gypsum, china clay. Functions • It reduces the cost of the paint. • It retards the settling of the pigment and cracking. e)Driers: They are oxygen carrier catalyst, which accelerate the process of drying. Examples : Metallic soaps, linoleates, resinates of Co, Mn and Pb. Function • They accelerate the drying of the oil film through oxidation, polymerisation and condensation. f) Plasticisers : These are chemicals added to paint to increase the plasticity and prevent cracking of the film. Examples : Triphenyl phosphate, tricresylphosphate, etc. g) Anti skinning agents: These are chemicals that prevents gelling and skinning of the paint film. Example : Polyhydroxy phenol Pigment volume concentration ( P.V.C ) It is a property of a paint which indicates the durability, adhesion and consistency. Higher the value, lower will be these qualitie PVC = Volume of pigment in the Paint Volume of (pigment + vehicle) in the paint Lecturer-II /VIII Electoplating – introduction and method of electroplating copper Electroplating (Electrodeposition ): The process of deposition of coating metal on the base metal by passing a direct current through an electrolytic solution which contains the soluble salt of the coating metal. Electroplating on metals • To increase the resistance to corrosion of the plated metal. • To improve the hardness and physical appearance. • To increase the decorative and commercial values of the metal. • To increase resistance to chemical attack. • To improve the surface properties. On non-metals • To increase strength. • To decorate surfaces of non metals like wood, plastic, glass. • For obtaining surface conductivity. Electrodeposition: (Also knows as electroplating) Electroplating is using a small sheet of metal in an electrocytic cell to coat another object. It is used to protect objects from damage against rusting and corrosion of metals. Lecturer-II /IX Electroless plating-introduction and method of plating nickel. Principle Electroless plating is a technique of depositing a noble metal (noble metal salt solution) on a catalytically active surface of the base metal by using a suitable reducing agent without electric current. Metal ions + reducing agent Metal + Oxidised products 1. Electroless nickel plating Step 1 : Pretreatment and activation of the surface: The surface to be plated is degreased first by using an organic solvent or alkali followed by acid treatment. Step 2 : Plating bath Sl.No. Nature of the content Compound Quantity (g/L) 1. Coating solution NiCl2 20 2. Sodium hypophosphite 20 3. Reducing agent Complexing agent cum exhaltaant 4. Sodium succinate 15 Buffer Sodium acetate 10 5. pH 4.5 - 6. Temperature 93oC - Step 3 : Plating Procedure The pretreated object is placed in the plating bath for the required time. The reduction reaction occurs and nickel gets coated over the object A) H2PO2- + H2O H2PO3- + 2 H+ + 2eB) Ni2++ 2e- Ni Over all reaction Ni2+ + H2PO2- + H2O Ni + H2PO3- + 2 H+ Applications • It is used in domestic and automotive fields (eg., jewellary, tops of perfume bottle) Advantages of electro less plating over electroplating • No electricity is required • It can be easily plated on insulators • Electroless nickel coated polymers are used as decorative material. • Complicated parts can be plated uniformly. • Copper and nickel coated plastic cabinets are used in digital and electronic instruments. • This coating possess unique mechanical, chemical and magnetic features