6.5 Main Group
... … if you combine the last three reactions with this one CaCO3 + heat → CaO + CO2(g) … you can build castles, bridges, houses, etc. - why ? Amphoteric Oxide: Can react with both acids and bases, acting either as an acidic or basic oxide, depending on conditions: ...
... … if you combine the last three reactions with this one CaCO3 + heat → CaO + CO2(g) … you can build castles, bridges, houses, etc. - why ? Amphoteric Oxide: Can react with both acids and bases, acting either as an acidic or basic oxide, depending on conditions: ...
Solid State Synthesis
... • This method is easy and effective when it works. It is not suitable when Reactants of comparable water solubility cannot be found. The precipitation rates of the reactants are markedly different. These limitations make this route unpractical for many combinations of ions. Furthermore, accurate sto ...
... • This method is easy and effective when it works. It is not suitable when Reactants of comparable water solubility cannot be found. The precipitation rates of the reactants are markedly different. These limitations make this route unpractical for many combinations of ions. Furthermore, accurate sto ...
Sections 6.4 - 6.5
... … if you combine the last three reactions with this one CaCO3 + heat → CaO + CO2(g) … you can build castles, bridges, houses, etc. - why ? Amphoteric Oxide: Can react with both acids and bases, acting either as an acidic or basic oxide, depending on conditions: ...
... … if you combine the last three reactions with this one CaCO3 + heat → CaO + CO2(g) … you can build castles, bridges, houses, etc. - why ? Amphoteric Oxide: Can react with both acids and bases, acting either as an acidic or basic oxide, depending on conditions: ...
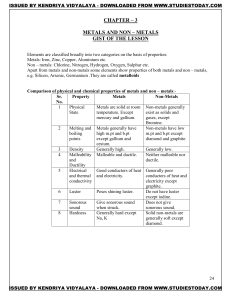
METALS AND NON – METALS Concepts
... Metals react with water to Non-metals do not react with water form metal oxides or metal with water, steam to hydroxide and H2 gas is evolve hydrogen gas. ...
... Metals react with water to Non-metals do not react with water form metal oxides or metal with water, steam to hydroxide and H2 gas is evolve hydrogen gas. ...
CERAMICS MATERIALS - Wits Structural Chemistry
... (b) Electronic and Magnetic properties The 3d-metal oxides such as MnO, FeO, CoO and NiO are semiconductors and TiO and VO are metallic conductors. 3d-metal oxides - MnO, Fe1-xO, CoO and NiO have low conductivity that increase with temperature or have such large band gaps that become insulators. Th ...
... (b) Electronic and Magnetic properties The 3d-metal oxides such as MnO, FeO, CoO and NiO are semiconductors and TiO and VO are metallic conductors. 3d-metal oxides - MnO, Fe1-xO, CoO and NiO have low conductivity that increase with temperature or have such large band gaps that become insulators. Th ...
Ei otsikkoa
... neutral aqueous solution weakly acidic aq. sol. acidic acidic reaction with water produces HCl acidic and the element's oxoacid acidic acidic ...
... neutral aqueous solution weakly acidic aq. sol. acidic acidic reaction with water produces HCl acidic and the element's oxoacid acidic acidic ...
Polyatomic Ions (Memorize for Wednesday, January 31
... CHEMICAL NOMENCLATURE Oxidation Numbers (similar to valence electrons) Group 1 = +1 Group 2 = +2 Groups 13-18 = group # - 10 for most elements d-block elements must be looked up (groups 3-12 use roman numerals) oxidation numbers for ALL compounds MUST equal zero Metal-Non-metal (binary ionic compoun ...
... CHEMICAL NOMENCLATURE Oxidation Numbers (similar to valence electrons) Group 1 = +1 Group 2 = +2 Groups 13-18 = group # - 10 for most elements d-block elements must be looked up (groups 3-12 use roman numerals) oxidation numbers for ALL compounds MUST equal zero Metal-Non-metal (binary ionic compoun ...
Unit 10
... Extracting metal – getting metal from ores. What are the metal compounds from mineral ores? Are they soluble in water? Insoluble metal oxides, carbonates and sulphides – found in ores Which metals are found free in nature? Unreactive metals such as gold and platinum found free (as elements) in natur ...
... Extracting metal – getting metal from ores. What are the metal compounds from mineral ores? Are they soluble in water? Insoluble metal oxides, carbonates and sulphides – found in ores Which metals are found free in nature? Unreactive metals such as gold and platinum found free (as elements) in natur ...
Inweld Nibral, Aluminum Bronze 46
... metal is used for MIG and TIG welding of cast and wrought nickel-aluminum-bronze parts where there is a requirement for high resistance to corrosion, erosion and cavitation in salt or brackish water. The most popular application of Inweld Nibral is for repair and maintenance of ship propellers. It i ...
... metal is used for MIG and TIG welding of cast and wrought nickel-aluminum-bronze parts where there is a requirement for high resistance to corrosion, erosion and cavitation in salt or brackish water. The most popular application of Inweld Nibral is for repair and maintenance of ship propellers. It i ...
Lecture 2-Extraction of Elements
... Properties of Ellingham diagram • All metal oxide curves slop upwards • If materials melt / vaporize, the slope changes • When the curve crosses ∆Go = 0, decomposition of oxide begins (Ag, Au, Hg) • Electropositive metal curves are at the bottom of the diagram • Any metal will reduce the oxide of o ...
... Properties of Ellingham diagram • All metal oxide curves slop upwards • If materials melt / vaporize, the slope changes • When the curve crosses ∆Go = 0, decomposition of oxide begins (Ag, Au, Hg) • Electropositive metal curves are at the bottom of the diagram • Any metal will reduce the oxide of o ...
Metals and non-metals III IMPORTANT POINTS Non-metals
... form ions by gaining electrons and are said to be electronegative nare oxidising agents as they react by gaining electrons N + n e == N react with air or oxygen to form acidic oxides, which can dissolve in water to form acids do not react with dilute acids, water or steam form covalent compounds wit ...
... form ions by gaining electrons and are said to be electronegative nare oxidising agents as they react by gaining electrons N + n e == N react with air or oxygen to form acidic oxides, which can dissolve in water to form acids do not react with dilute acids, water or steam form covalent compounds wit ...
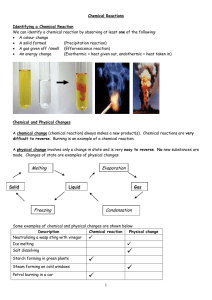
Chemical Reactions
... Chemists often want to speed up chemical reactions. To do this they decrease the particle size, increase the temperature, increase the concentration or use a catalyst. ...
... Chemists often want to speed up chemical reactions. To do this they decrease the particle size, increase the temperature, increase the concentration or use a catalyst. ...
9F Reactivity - Parrs Wood High School
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
Naming Ionic Compounds
... ** this is just like you learned for molecular compounds except you are not worried about the numbers of an element examples: NaCl – sodium chloride CaCl2 – calcium chloride Mg3N2 – magnesium nitride PbO – lead oxide ...
... ** this is just like you learned for molecular compounds except you are not worried about the numbers of an element examples: NaCl – sodium chloride CaCl2 – calcium chloride Mg3N2 – magnesium nitride PbO – lead oxide ...
Balancing Single Replacement Reactions - Kossmann
... Name: __________________________ Class: ____________________ Date: _____________ ...
... Name: __________________________ Class: ____________________ Date: _____________ ...
Metals & Metallurgy
... Because the bands are so close, it is easy to promote electrons to a higher energy level. With M.O.'s, half of the orbitals are bonding and half are antibonding. Thus, with halffilled d subshells, the bonding M.O.'s are filled. After that, the electrons begin to fill antibonding orbitals which weake ...
... Because the bands are so close, it is easy to promote electrons to a higher energy level. With M.O.'s, half of the orbitals are bonding and half are antibonding. Thus, with halffilled d subshells, the bonding M.O.'s are filled. After that, the electrons begin to fill antibonding orbitals which weake ...
C1a - Mr Corfe
... Q2. when potassium reacts with water the colour of the flame is? Q3. When baking powder is heated, it breaks down to form new substances. This is? Q4. A coin was reacted to form a solution. Sodium hydroxide solution was added to this solution. A blue precipitate showed the coin contained? Q5. Gas fr ...
... Q2. when potassium reacts with water the colour of the flame is? Q3. When baking powder is heated, it breaks down to form new substances. This is? Q4. A coin was reacted to form a solution. Sodium hydroxide solution was added to this solution. A blue precipitate showed the coin contained? Q5. Gas fr ...
Shape-Memory Alloy
... These materials are classified according to the predominant metal in the alloy, which may be cobalt, nickel, or iron. Other alloying elements include the refractory metals (Nb, Mo,W, Ta), chromium, and titanium. ...
... These materials are classified according to the predominant metal in the alloy, which may be cobalt, nickel, or iron. Other alloying elements include the refractory metals (Nb, Mo,W, Ta), chromium, and titanium. ...
Unit A Remediation Review
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
Contamination profile of Printed Circuit Board Assemblies in relation
... contaminated with substantial amounts of flux residues [1] [2] including the activator components, mainly weak organic acids (WOA). Typical WOAs found in no-clean flux systems are di-carboxylic acids such as adipic, glutaric, succinic and malic acids, which results in ionic residues on the PCBA surf ...
... contaminated with substantial amounts of flux residues [1] [2] including the activator components, mainly weak organic acids (WOA). Typical WOAs found in no-clean flux systems are di-carboxylic acids such as adipic, glutaric, succinic and malic acids, which results in ionic residues on the PCBA surf ...
Corrosion and Electrolysis
... the process will itself become reduced. This rapidly oxidized or rusts the one metal but prevents the less active metal from oxidizing (rusting) ...
... the process will itself become reduced. This rapidly oxidized or rusts the one metal but prevents the less active metal from oxidizing (rusting) ...
Switchable nanocatalysts: using ferroelectric oxides to control surface catalysis
... Department of Materials Science and Engineering University of Pennsylvania ...
... Department of Materials Science and Engineering University of Pennsylvania ...
Flux (metallurgy)

In metallurgy, a flux (derived from Latin fluxus meaning “flow”) is a chemical cleaning agent, flowing agent, or purifying agent. Fluxes may have more than one function at a time. They are used in both extractive metallurgy and metal joining.Some of the earliest known fluxes were carbonate of soda, potash, charcoal, coke, borax, lime, lead sulfide and certain minerals containing phosphorus. Iron ore was also used as a flux in the smelting of copper. These agents served various functions, the simplest being a reducing agent which prevented oxides from forming on the surface of the molten metal, while others absorbed impurities into the slag which could be scraped off the molten metal.As cleaning agents, fluxes facilitate soldering, brazing, and welding by removing oxidation from the metals to be joined. Common fluxes are: ammonium chloride or rosin for soldering tin; hydrochloric acid and zinc chloride for soldering galvanized iron (and other zinc surfaces); and borax for brazing or braze-welding ferrous metals. In the process of smelting, inorganic chlorides, fluorides (see fluorite), limestone and other materials are designated as ""fluxes"" when added to the contents of a smelting furnace or a cupola for the purpose of purging the metal of chemical impurities such as phosphorus, and of rendering slag more liquid at the smelting temperature. The slag is a liquid mixture of ash, flux, and other impurities. This reduction of slag viscosity with temperature, increasing the flow of slag in smelting, is the original origin of the word flux in metallurgy. Fluxes are also used in foundries for removing impurities from molten nonferrous metals such as aluminum, or for adding desirable trace elements such as titanium.In high-temperature metal joining processes (welding, brazing and soldering), the primary purpose of flux is to prevent oxidation of the base and filler materials. Tin-lead solder (e.g.) attaches very well to copper, but poorly to the various oxides of copper, which form quickly at soldering temperatures. Flux is a substance which is nearly inert at room temperature, but which becomes strongly reducing at elevated temperatures, preventing the formation of metal oxides. Additionally, flux allows solder to flow easily on the working piece rather than forming beads as it would otherwise.The role of a flux in joining processes is typically dual: dissolving of the oxides on the metal surface, which facilitates wetting by molten metal, and acting as an oxygen barrier by coating the hot surface, preventing its oxidation. In some applications molten flux also serves as a heat transfer medium, facilitating heating of the joint by the soldering tool or molten solder.Fluxes for soft soldering are typically of organic nature, though inorganic fluxes, usually based on halogenides and/or acids, are also used in non-electronics applications. Fluxes for brazing operate at significantly higher temperatures and are therefore mostly inorganic; the organic compounds tend to be of supplementary nature.