Name: Per: _____ Date: ______ Unit 5 Redemption Packet: The
... questions. If you missed any notes, you will find them at www.starkchemistry.weebly.com. 1. Mendeleev arranged the elements in his periodic table by increasing _______________ ________________. 2. Why did Mendeleev leave spaces in his periodic table? _________________________________________________ ...
... questions. If you missed any notes, you will find them at www.starkchemistry.weebly.com. 1. Mendeleev arranged the elements in his periodic table by increasing _______________ ________________. 2. Why did Mendeleev leave spaces in his periodic table? _________________________________________________ ...
HISTORY OF THE PERIODIC TABLE
... IX 1930 Glenn Seaborg – “plucked out” the heaviest Elements (Actinide series & Lanthanide series) X ROY ALEXANDER – designed a three-dimensional Periodic chart (1994) retains the separate Lanthanide and Actinide series. ...
... IX 1930 Glenn Seaborg – “plucked out” the heaviest Elements (Actinide series & Lanthanide series) X ROY ALEXANDER – designed a three-dimensional Periodic chart (1994) retains the separate Lanthanide and Actinide series. ...
Periodic Table Cloze - Science
... Fill in the blanks with words from the box. atom gold nonmetals ...
... Fill in the blanks with words from the box. atom gold nonmetals ...
2.5-The Periodic Table
... take its place. He predicted the properties of the “unknown” elements, and within the next sixteen years those gaps were filled in with newly discovered elements that matched precisely with Mendeleev’s predictions! ...
... take its place. He predicted the properties of the “unknown” elements, and within the next sixteen years those gaps were filled in with newly discovered elements that matched precisely with Mendeleev’s predictions! ...
The Atom and how it is organized - Cashmere
... central nucleus with orbiting electrons. ◦ A nucleus is made up of positively charged PROTONS and neutral NEUTRONS. ◦ ELECTRONS are negatively charged and orbit around the nucleus. ...
... central nucleus with orbiting electrons. ◦ A nucleus is made up of positively charged PROTONS and neutral NEUTRONS. ◦ ELECTRONS are negatively charged and orbit around the nucleus. ...
PERIODIC TABLE - WordPress.com
... 6. Name three metalloids (semi-metals) from the Periodic Table. 7. Which block (s, p, d, f) does iron belong to in the Periodic Table? 8. Which group in the Periodic Table does an element with electronic configuration 1s 2 2s2 2p5 belong to? 9. Which blocks (s, p, d, f) in the Periodic Table contain ...
... 6. Name three metalloids (semi-metals) from the Periodic Table. 7. Which block (s, p, d, f) does iron belong to in the Periodic Table? 8. Which group in the Periodic Table does an element with electronic configuration 1s 2 2s2 2p5 belong to? 9. Which blocks (s, p, d, f) in the Periodic Table contain ...
1. Define: a. Period b. group or family
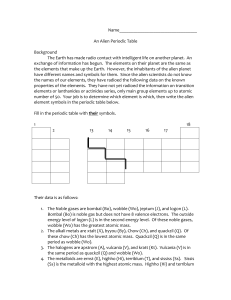
... Look at the six elements below and fill in their symbols. ...
... Look at the six elements below and fill in their symbols. ...
Name
... exchange of information has begun. The elements on their planet are the same as the elements that make up the Earth. However, the inhabitants of the alien planet have different names and symbols for them. Since the alien scientists do not know the names of our elements, they have radioed the followi ...
... exchange of information has begun. The elements on their planet are the same as the elements that make up the Earth. However, the inhabitants of the alien planet have different names and symbols for them. Since the alien scientists do not know the names of our elements, they have radioed the followi ...
atomic number
... element has. For instance, hydrogen has 1 proton, so it’s atomic number is 1. The atomic number is unique to that element. No two elements have the same atomic number. ...
... element has. For instance, hydrogen has 1 proton, so it’s atomic number is 1. The atomic number is unique to that element. No two elements have the same atomic number. ...
Name: Date: Period: ______ Graphing Periodic Trends Purpose:To
... now no longer considered to be a planet. Of these three elements, only Dd is naturallyoccurring and it is also an alpha decay product of Cc. They were all discovered at the University of California at Berkeley. Isoptopes of Bb are beta decay products of Dd. 12. Ee, Ff, and Gg are in the same group. ...
... now no longer considered to be a planet. Of these three elements, only Dd is naturallyoccurring and it is also an alpha decay product of Cc. They were all discovered at the University of California at Berkeley. Isoptopes of Bb are beta decay products of Dd. 12. Ee, Ff, and Gg are in the same group. ...
Chemistry 104: Introduction to the Chemistry of Materials
... penalized 10% per day. No papers will be accepted after 12:45 pm on Monday, September 29. Grammar and Spelling 2 points Although the paper will be only a few paragraphs long, the information should be reported in complete sentences. Please use double spacing. Content 6 points 1. Identify the element ...
... penalized 10% per day. No papers will be accepted after 12:45 pm on Monday, September 29. Grammar and Spelling 2 points Although the paper will be only a few paragraphs long, the information should be reported in complete sentences. Please use double spacing. Content 6 points 1. Identify the element ...
Periodic table
... An element’s chemical properties are related to the number of electrons in their outside shell ...
... An element’s chemical properties are related to the number of electrons in their outside shell ...
- Priddy ISD
... alkali metal - group 1 elements, except for hydrogen, they are reactive and usually exist as compounds with other elements alkaline earth metal - group 2 elements in the modern periodic table; highly reactive aufbau principle - states that each electron occupies the lowest energy orbital available e ...
... alkali metal - group 1 elements, except for hydrogen, they are reactive and usually exist as compounds with other elements alkaline earth metal - group 2 elements in the modern periodic table; highly reactive aufbau principle - states that each electron occupies the lowest energy orbital available e ...
Basics of the Periodic Table
... *Directions: Use the Periodic Table in your textbook to answer the following questions. Read carefully! ...
... *Directions: Use the Periodic Table in your textbook to answer the following questions. Read carefully! ...
Ch. 14 Test Review
... Ch. 14 Test Review CHEMICAL PERIODICITY COMPLETION decreases increases (3) groups (2) periods transition metals ionization energy atomic # noble gases representative electronegativity The periodic table organizes the elements into vertical ____________ and horizontal ____________ in order of increas ...
... Ch. 14 Test Review CHEMICAL PERIODICITY COMPLETION decreases increases (3) groups (2) periods transition metals ionization energy atomic # noble gases representative electronegativity The periodic table organizes the elements into vertical ____________ and horizontal ____________ in order of increas ...
CI_Chap_1_Test_A_Study_Guide
... Transition metals like Gold are toward the middle of the periodic table. The most common element in the universe is hydrogen. Atoms of an element always have a certain number of protons. Isotopes of an element differ in the number of neutrons. When an atom loses one or more electrons in becomes an i ...
... Transition metals like Gold are toward the middle of the periodic table. The most common element in the universe is hydrogen. Atoms of an element always have a certain number of protons. Isotopes of an element differ in the number of neutrons. When an atom loses one or more electrons in becomes an i ...
The Chinese High School
... Elements have been placed systematically into the Periodic Table according to their properties. Scientists have classified elements into 2 main groups, the metals and non-metals. Each vertical column is called a _____________________. Each group’s number is reflected at the top of the periodic table ...
... Elements have been placed systematically into the Periodic Table according to their properties. Scientists have classified elements into 2 main groups, the metals and non-metals. Each vertical column is called a _____________________. Each group’s number is reflected at the top of the periodic table ...
Atomic and Molecular Structure – Standard 1 Review
... 1d.4 Be able to predict the combination of elements in a compound using your knowledge of their valence electrons. ...
... 1d.4 Be able to predict the combination of elements in a compound using your knowledge of their valence electrons. ...