Part D Questions and Problems
... filled first and second levels. When chlorine reacts, it gains an electron in its highest occupied energy level. An ion with three occupied energy levels is larger than an ion with two occupied energy levels. 4. Across a period from left to right the principal energy level remains the same, but the ...
... filled first and second levels. When chlorine reacts, it gains an electron in its highest occupied energy level. An ion with three occupied energy levels is larger than an ion with two occupied energy levels. 4. Across a period from left to right the principal energy level remains the same, but the ...
Chapter 5 - Geocities
... The Periodic Law Section 1: History of the Periodic Table Periodic law: The physical and chemical properties of the elements are periodic functions of their atomic numbers. Reworded Periodic law: When the elements are arranged in order of increasing atomic number, elements with similar properties ap ...
... The Periodic Law Section 1: History of the Periodic Table Periodic law: The physical and chemical properties of the elements are periodic functions of their atomic numbers. Reworded Periodic law: When the elements are arranged in order of increasing atomic number, elements with similar properties ap ...
Study Guide for Quiz on Tuesday February 26th - seys
... Periodic table- a table of the elements arranged by atomic number that shows a pattern in their properties Group- a vertical column in the periodic table of the elements, elements have similar properties Period- a horizontal row in the periodic table of the elements, elements have varying propertie ...
... Periodic table- a table of the elements arranged by atomic number that shows a pattern in their properties Group- a vertical column in the periodic table of the elements, elements have similar properties Period- a horizontal row in the periodic table of the elements, elements have varying propertie ...
ionic and covalent bonding - Atomic Theory and Periodic Table
... The formula of an ionic compound is actually the _______________ in which the ions are present. This can be easily determined because the overall charge on an ionic compound must neutral be _______________. As this formula gives a ratio rather than actual numbers of ions, it is empirical called an _ ...
... The formula of an ionic compound is actually the _______________ in which the ions are present. This can be easily determined because the overall charge on an ionic compound must neutral be _______________. As this formula gives a ratio rather than actual numbers of ions, it is empirical called an _ ...
periodic table - rosedalegrade9chemistry
... known elements in the early 1800’s. Could the elements be organized based on properties like colour, smell or taste? Not really, because the characteristics or properties were not unique. Early scientists found a property unique to each element, atomic mass. ...
... known elements in the early 1800’s. Could the elements be organized based on properties like colour, smell or taste? Not really, because the characteristics or properties were not unique. Early scientists found a property unique to each element, atomic mass. ...
Periodic Table
... They are also harder than group 1 metals and have higher melting points. How many valence e- do they have? Differences in their reactivity is how they react with water. They are good conductors of electricity. Mg can be as hard as steel when mixed with other metals but is extremely light. How could ...
... They are also harder than group 1 metals and have higher melting points. How many valence e- do they have? Differences in their reactivity is how they react with water. They are good conductors of electricity. Mg can be as hard as steel when mixed with other metals but is extremely light. How could ...
Chapter Assessment
... 8. The trend in the atomic radii as you move down the group 1A elements is partially due to ...
... 8. The trend in the atomic radii as you move down the group 1A elements is partially due to ...
Periodic Table Notes Fill In
... 14. Where are the Transition Metals located? ______________________________________ 15. Where are the Lanthanides located? __________________________________________ 16. Where are the Actinides located? _____________________________________________ 17. Where are the Halogens located? _______________ ...
... 14. Where are the Transition Metals located? ______________________________________ 15. Where are the Lanthanides located? __________________________________________ 16. Where are the Actinides located? _____________________________________________ 17. Where are the Halogens located? _______________ ...
File - Unit #1-0
... 18. What sublevels are filling across the Transition Elements? 19. Elements within a group have a similar number of 20. Elements across a series have the same number of 21. A colored ion generally indicates a 22. As you go down a group, the elements generally become ( more / less) metallic. 23. The ...
... 18. What sublevels are filling across the Transition Elements? 19. Elements within a group have a similar number of 20. Elements across a series have the same number of 21. A colored ion generally indicates a 22. As you go down a group, the elements generally become ( more / less) metallic. 23. The ...
The Periodic Table
... is capitalized and the other letters are lower case. – Usually the symbol is the same as the first letter of the element • If it is not, it may be the first letter of the Latin word for the same element. Example – Iron’s symbol is Fe…short for Ferrium • If the single letter has already been used, th ...
... is capitalized and the other letters are lower case. – Usually the symbol is the same as the first letter of the element • If it is not, it may be the first letter of the Latin word for the same element. Example – Iron’s symbol is Fe…short for Ferrium • If the single letter has already been used, th ...
Elements and the Periodic Table
... fit together they can form diamond, coal, or graphite. How many elements are there? There are currently 118 known elements. Of these, only 94 are thought to naturally exist on Earth. Families of Elements Elements are sometimes grouped together because they have similar properties. Here a few of the ...
... fit together they can form diamond, coal, or graphite. How many elements are there? There are currently 118 known elements. Of these, only 94 are thought to naturally exist on Earth. Families of Elements Elements are sometimes grouped together because they have similar properties. Here a few of the ...
atomic number - Net Start Class
... Noble Gases are colorless gases that are extremely un-reactive. One important property of the noble gases is their inactivity. They are inactive because their outermost energy level is full. Because they do not readily combine with other elements to form compounds, the noble gases are called inert. ...
... Noble Gases are colorless gases that are extremely un-reactive. One important property of the noble gases is their inactivity. They are inactive because their outermost energy level is full. Because they do not readily combine with other elements to form compounds, the noble gases are called inert. ...
Finals Review ans 2012sem 1
... Which of the following is a clue of a chemical change? a gas forms when vinegar and baking soda are mixed ____ 43. Moving from left to right across a row of the periodic table, which of the following values increases by exactly one from element to element? atomic number ____ 44. A substance has a me ...
... Which of the following is a clue of a chemical change? a gas forms when vinegar and baking soda are mixed ____ 43. Moving from left to right across a row of the periodic table, which of the following values increases by exactly one from element to element? atomic number ____ 44. A substance has a me ...
Section 12.3
... silvery in their pure form and are highly reactive. This group includes the elements lithium (Li), sodium (Na), and potassium (K). ...
... silvery in their pure form and are highly reactive. This group includes the elements lithium (Li), sodium (Na), and potassium (K). ...
File
... List your choice here _________________________________________________ Step 2 – Families – The “Elements” in the Periodic table are grouped into Families, which are the vertical columns on the chart. Elements in Families have similar properties. Your table should have a minimum of four families (su ...
... List your choice here _________________________________________________ Step 2 – Families – The “Elements” in the Periodic table are grouped into Families, which are the vertical columns on the chart. Elements in Families have similar properties. Your table should have a minimum of four families (su ...
Chapter 7:
... group 1A, it is not a member of any group because it has properties of both metals and nonmetals. It behaves as a metal when it loses its electron. It behaves as a nonmetal when it gains an electron. The universe contains more than 90% hydrogen by mass. Hydrogen reacts violently with oxygen in the p ...
... group 1A, it is not a member of any group because it has properties of both metals and nonmetals. It behaves as a metal when it loses its electron. It behaves as a nonmetal when it gains an electron. The universe contains more than 90% hydrogen by mass. Hydrogen reacts violently with oxygen in the p ...
The Periodic Table
... Group 18: The Noble Gases (The Inert Gases) Nonreactive Colorless gases Nonmetal Charge is 0: 2 or 8 valence electrons- have a full outer energy level ...
... Group 18: The Noble Gases (The Inert Gases) Nonreactive Colorless gases Nonmetal Charge is 0: 2 or 8 valence electrons- have a full outer energy level ...
22 diatomic molecules
... Diatomic molecules Molecules made up of only two atoms are called diatomic molecules (two atom), e.g. hydrogen chloride, HCl (one carbon atom and one chlorine atom), and carbon monoxide, CO, (one carbon atom and one oxygen atom). Certain elements normally exist as diatomic molecules. Since diatomic ...
... Diatomic molecules Molecules made up of only two atoms are called diatomic molecules (two atom), e.g. hydrogen chloride, HCl (one carbon atom and one chlorine atom), and carbon monoxide, CO, (one carbon atom and one oxygen atom). Certain elements normally exist as diatomic molecules. Since diatomic ...
ATOMIC THEORY OF MATTER
... which involve electron interactions. The electrons act as the “glue” between atoms. – If electrons are shared between two atoms, the bond is a covalent bond. I.e., the bond between two non-metal atoms. – If electrons are transferred to produce ions, the bond is ionic. • Ions are charged particles wh ...
... which involve electron interactions. The electrons act as the “glue” between atoms. – If electrons are shared between two atoms, the bond is a covalent bond. I.e., the bond between two non-metal atoms. – If electrons are transferred to produce ions, the bond is ionic. • Ions are charged particles wh ...
The Periodic Table & Formation of Ions
... they have equal #’s of protons and electrons When an atom gains or loses electrons, this equality is upset… The atom is no longer neutral…and is no longer called an atom ...
... they have equal #’s of protons and electrons When an atom gains or loses electrons, this equality is upset… The atom is no longer neutral…and is no longer called an atom ...
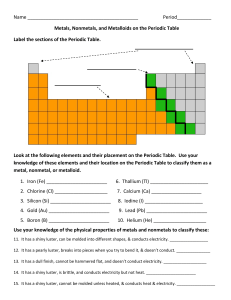
Name Period_____________ Metals, Nonmetals, and Metalloids on
... 13. It has a dull finish, cannot be hammered flat, and doesn’t conduct electricity. ___________________ 14. It has a shiny luster, is brittle, and conducts electricity but not heat. ______________________ 15. It has a shiny luster, cannot be molded unless heated, & conducts heat & electricity. _____ ...
... 13. It has a dull finish, cannot be hammered flat, and doesn’t conduct electricity. ___________________ 14. It has a shiny luster, is brittle, and conducts electricity but not heat. ______________________ 15. It has a shiny luster, cannot be molded unless heated, & conducts heat & electricity. _____ ...
Periodic Table of Elements
... • On the periodic table, the average mass of the atoms in an element is expressed as the atomic mass. • Adding the protons and neutrons will give you the atomic mass. • For example Iron (Fe) has an atomic mass of 56 because there are 26 protons and 30 neutrons and added together that makes 56. ...
... • On the periodic table, the average mass of the atoms in an element is expressed as the atomic mass. • Adding the protons and neutrons will give you the atomic mass. • For example Iron (Fe) has an atomic mass of 56 because there are 26 protons and 30 neutrons and added together that makes 56. ...
Review for Chemistry Unit Test #2 (Chapters 4, 11, and 12) Chapter
... How can compounds be broken down? How can you increase the solubility rate of a sugar cube? Define an alloy. What do we call a pure substance made of 2 or more elements? How do elements join to form compounds? Differentiate between an element, mixture, and a compound. What is a solution? What is a s ...
... How can compounds be broken down? How can you increase the solubility rate of a sugar cube? Define an alloy. What do we call a pure substance made of 2 or more elements? How do elements join to form compounds? Differentiate between an element, mixture, and a compound. What is a solution? What is a s ...
Period 3 element
A period 3 element is one of the chemical elements in the third row (or period) of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behaviour of the elements as their atomic number increases: a new row is begun when the periodic table skips a row and a chemical behaviour begins to repeat, meaning that elements with similar behavior fall into the same vertical columns. The third period contains eight elements: sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine, and argon. The first two, sodium and magnesium, are members of the s-block of the periodic table, while the others are members of the p-block. Note that there is a 3d orbital, but it is not filled until Period 4, such giving the period table its characteristic shape of ""two rows at a time"". All of the period 3 elements occur in nature and have at least one stable isotope.