Review of Basic Chemistry
... C: The Periodic Table A chemical group/family is a vertical column of elements that have similar physical and chemical properties. On the periodic table there are 18 vertical groups. Column 1: alkali metals Column 2: alkaline earth metals Column 3-11: transition metals Column 17: halogens Column 18 ...
... C: The Periodic Table A chemical group/family is a vertical column of elements that have similar physical and chemical properties. On the periodic table there are 18 vertical groups. Column 1: alkali metals Column 2: alkaline earth metals Column 3-11: transition metals Column 17: halogens Column 18 ...
u4_tqs - Teach-n-Learn-Chem
... 12. Why is the symbol for sodium printed in black? sodium is a solid at room temperature 13. List the chemical symbols of all of the… …alkaline earth metals. …noble gases. Be, Mg, Ca, Sr, Ba, Ra He, Ne, Ar, Kr, Xe, Rn ...
... 12. Why is the symbol for sodium printed in black? sodium is a solid at room temperature 13. List the chemical symbols of all of the… …alkaline earth metals. …noble gases. Be, Mg, Ca, Sr, Ba, Ra He, Ne, Ar, Kr, Xe, Rn ...
Chapter 6
... -gases or brittle solids, dull surfaces, used as insulators -have 5 or more electrons in outer energy level ...
... -gases or brittle solids, dull surfaces, used as insulators -have 5 or more electrons in outer energy level ...
2- Periodic Trends
... • unique properties different from group 1 and 2 metals (make coloured compounds, speed up chemical reactions…) ...
... • unique properties different from group 1 and 2 metals (make coloured compounds, speed up chemical reactions…) ...
Chapter 6 Study Guide
... a. period 3, Group IIIA _____________________________________________ b. period 1, Group VIIIA ____________________________________________ c. period 4, Group IIA ______________________________________________ d. period 6, Group VA ______________________________________________ 7. How many Valence e ...
... a. period 3, Group IIIA _____________________________________________ b. period 1, Group VIIIA ____________________________________________ c. period 4, Group IIA ______________________________________________ d. period 6, Group VA ______________________________________________ 7. How many Valence e ...
Chapter 6 Review“The Periodic Table”
... 7. Dalton said that atoms were indivisible and atoms of the same element were identical. Have any changes been made to this? Yes – isotopes have different number of neutrons 8. The mass number of an element is equal to _____. Number protons + ...
... 7. Dalton said that atoms were indivisible and atoms of the same element were identical. Have any changes been made to this? Yes – isotopes have different number of neutrons 8. The mass number of an element is equal to _____. Number protons + ...
Chapter 21 Chemistry of the Main
... Nonmetallic character dominates in this group Nonmetallic O exists as diatomic molecules Nonmetallic S exists as various covalently bonded polyatomic forms Metalloids Se and Te are more metallic than S, but bear some resemblance to S Po is even more metallic, but its behavior is not well known ...
... Nonmetallic character dominates in this group Nonmetallic O exists as diatomic molecules Nonmetallic S exists as various covalently bonded polyatomic forms Metalloids Se and Te are more metallic than S, but bear some resemblance to S Po is even more metallic, but its behavior is not well known ...
ExamView - chemistry chapter 6 test.tst
... ____ 15. What is the element with the highest electronegativity value? a. helium c. calcium b. cesium d. fluorine ____ 16. Of the elements Fe, Hg, U, and Te, which is a representative element? a. Te c. Fe b. U d. Hg ____ 17. Which statement about noble gases is correct? a. They form compounds with v ...
... ____ 15. What is the element with the highest electronegativity value? a. helium c. calcium b. cesium d. fluorine ____ 16. Of the elements Fe, Hg, U, and Te, which is a representative element? a. Te c. Fe b. U d. Hg ____ 17. Which statement about noble gases is correct? a. They form compounds with v ...
Chapter 22 Chemistry of The NonMetals
... Thermal stability (measured by ∆G°f) decreases as we go down a group and increases across a period. Most stable is HF. Metal hydrides, such as CaH2, react with water to give H2 and metal hydroxide. ...
... Thermal stability (measured by ∆G°f) decreases as we go down a group and increases across a period. Most stable is HF. Metal hydrides, such as CaH2, react with water to give H2 and metal hydroxide. ...
Dimitri Mendeleev- The father of the modern periodic table. Russian
... tablefrom actinium to lawrencium (atomic numbers 89–103). All are radioactive heavy metals; and only the first four (actinium, thorium, protactinium, and uranium) occur in nature. The other 11 (the transuranium elements) are unstable and are produced only artificially. ...
... tablefrom actinium to lawrencium (atomic numbers 89–103). All are radioactive heavy metals; and only the first four (actinium, thorium, protactinium, and uranium) occur in nature. The other 11 (the transuranium elements) are unstable and are produced only artificially. ...
Section 2.5 Molecules and Ions
... b) All atoms of a given element are identical, having the same size, mass, and chemical properties. The atoms of one element are different from atoms of all other elements. (Lego blocks can be used as an example). c) Compounds are composed of atoms of more than one element. In any compound, the rat ...
... b) All atoms of a given element are identical, having the same size, mass, and chemical properties. The atoms of one element are different from atoms of all other elements. (Lego blocks can be used as an example). c) Compounds are composed of atoms of more than one element. In any compound, the rat ...
File 9.08.16 the periodic table
... The modern periodic table was created by the Russian chemist, ____________ ____________. ______________ are represented on the Periodic Table with _____________ _______________. The elements are arranged from left to right based on their ___________ _______________. The atomic number comes from the ...
... The modern periodic table was created by the Russian chemist, ____________ ____________. ______________ are represented on the Periodic Table with _____________ _______________. The elements are arranged from left to right based on their ___________ _______________. The atomic number comes from the ...
sodium
... Atoms of this family have 6 valence electrons. Most elements in this family share electrons when forming compounds. Oxygen is the most abundant element in the earth’s crust. It is extremely active and combines with almost all elements. ...
... Atoms of this family have 6 valence electrons. Most elements in this family share electrons when forming compounds. Oxygen is the most abundant element in the earth’s crust. It is extremely active and combines with almost all elements. ...
AP chemistry Test Review
... c) Most alpha particles passed through the gold foil without being deflected since the nuclei of the gold atoms represent such a small portion of the atomic mass. d) Because of the electrons small masses they did not deflect the alpha particles. e) The high charge of the gold nuclei helps to account ...
... c) Most alpha particles passed through the gold foil without being deflected since the nuclei of the gold atoms represent such a small portion of the atomic mass. d) Because of the electrons small masses they did not deflect the alpha particles. e) The high charge of the gold nuclei helps to account ...
for the quiz on 6 mar
... 19.98 In order to have a stronger acid, you want to make the O-H bond weaker. (There is a figure on p.646 in Ch. 15 which may help with this…) If you increase the number of oxygens in the molecule, you are drawing more electron density away from the Cl by bonding it to more (greater number of) elect ...
... 19.98 In order to have a stronger acid, you want to make the O-H bond weaker. (There is a figure on p.646 in Ch. 15 which may help with this…) If you increase the number of oxygens in the molecule, you are drawing more electron density away from the Cl by bonding it to more (greater number of) elect ...
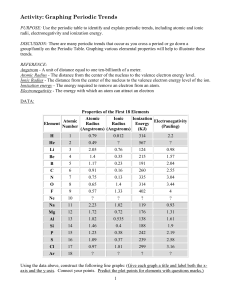
LSHS Graphing Periodic Trends Lab
... Ionization energy - The energy required to remove an electron from an atom. Electronegativity - The energy with which an atom can attract an electron ...
... Ionization energy - The energy required to remove an electron from an atom. Electronegativity - The energy with which an atom can attract an electron ...
Periodic Table Trends
... added to the outermost shell. For example, sodium has one electron in shell 3, followed by magnesium which has two electrons in shell 3. ...
... added to the outermost shell. For example, sodium has one electron in shell 3, followed by magnesium which has two electrons in shell 3. ...
X Unit 11 Test Study Guide (The Periodic Table)
... a sense of the type of questions you will see!! Sample Review Problems: (answer on a separate piece of paper if needed) 1. Fill in the following information for the element astatine: a. Symbol: At b. Group #: 17 c. Group name: Halogens d. Period: 6 e. Metal, nonmetal, or metalloid: nonmetal 2. What ...
... a sense of the type of questions you will see!! Sample Review Problems: (answer on a separate piece of paper if needed) 1. Fill in the following information for the element astatine: a. Symbol: At b. Group #: 17 c. Group name: Halogens d. Period: 6 e. Metal, nonmetal, or metalloid: nonmetal 2. What ...
The History of the Modern Periodic Table
... • was so confident in his table that he used it to predict the physical properties of three elements that were ...
... • was so confident in his table that he used it to predict the physical properties of three elements that were ...
FREE Sample Here
... charges) suffixes. The favored system in use is the Stock system, which designates different cations with Roman numerals. Roman numeral I indicates one positive charge; II means two positive charges, and so on. Table 2.3 can be challenging for students. A tip for learning the table is to indicate th ...
... charges) suffixes. The favored system in use is the Stock system, which designates different cations with Roman numerals. Roman numeral I indicates one positive charge; II means two positive charges, and so on. Table 2.3 can be challenging for students. A tip for learning the table is to indicate th ...
Chapter 2 - Test Bank
... charges) suffixes. The favored system in use is the Stock system, which designates different cations with Roman numerals. Roman numeral I indicates one positive charge; II means two positive charges, and so on. Table 2.3 can be challenging for students. A tip for learning the table is to indicate th ...
... charges) suffixes. The favored system in use is the Stock system, which designates different cations with Roman numerals. Roman numeral I indicates one positive charge; II means two positive charges, and so on. Table 2.3 can be challenging for students. A tip for learning the table is to indicate th ...
Periodic Trends Worksheet
... a. Which element has the highest first ionization energy? ____________________________ b. Which element has the lowest electronegativity? _________________________________ c. Which element has the least metallic character? _________________________________ d. Which element is the largest atom? _____ ...
... a. Which element has the highest first ionization energy? ____________________________ b. Which element has the lowest electronegativity? _________________________________ c. Which element has the least metallic character? _________________________________ d. Which element is the largest atom? _____ ...
Periodic Trends Review Sheet
... 9. Is it easier to form a positive ion with an element that has a high ionization energy or an element that has a low ionization energy? Why? 10. Na+ and Mg2+ ions each have ten electrons surrounding their nuclei. Which ion would you expect to have the larger radius? Explain your answer. 11. a. Expl ...
... 9. Is it easier to form a positive ion with an element that has a high ionization energy or an element that has a low ionization energy? Why? 10. Na+ and Mg2+ ions each have ten electrons surrounding their nuclei. Which ion would you expect to have the larger radius? Explain your answer. 11. a. Expl ...
Period 3 element
A period 3 element is one of the chemical elements in the third row (or period) of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behaviour of the elements as their atomic number increases: a new row is begun when the periodic table skips a row and a chemical behaviour begins to repeat, meaning that elements with similar behavior fall into the same vertical columns. The third period contains eight elements: sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine, and argon. The first two, sodium and magnesium, are members of the s-block of the periodic table, while the others are members of the p-block. Note that there is a 3d orbital, but it is not filled until Period 4, such giving the period table its characteristic shape of ""two rows at a time"". All of the period 3 elements occur in nature and have at least one stable isotope.