Document
... properties similar to ________________________________________________________. List a few elements that seem familiar to you: __________________________________ Name a few elements that you’ve never heard of before: _______________________________________________________________________ The horizon ...
... properties similar to ________________________________________________________. List a few elements that seem familiar to you: __________________________________ Name a few elements that you’ve never heard of before: _______________________________________________________________________ The horizon ...
Periodic Table Funsheet (KEY) 1. What family has the most active
... 14. Group 17 elements are called HALOGENS. 15. Group 18 elements are called NOBLE GASES. 16. As you go from left to right across the periodic table, the elements go from (METALS / nonmetals) to (metals / NONMETALS). 17. The most active element in Group 17 is FLUORINE. 18. What sublevels are filling ...
... 14. Group 17 elements are called HALOGENS. 15. Group 18 elements are called NOBLE GASES. 16. As you go from left to right across the periodic table, the elements go from (METALS / nonmetals) to (metals / NONMETALS). 17. The most active element in Group 17 is FLUORINE. 18. What sublevels are filling ...
Periodic Table Vocabulary Alkali metals
... 9. Representative elements-The representative elements are all the elements in groups one, two, thirteen, fourteen, fifteen, sixteen, seventeen and eighteen of the periodic table. Sentence- One group of the representative elements is the Halogens. ...
... 9. Representative elements-The representative elements are all the elements in groups one, two, thirteen, fourteen, fifteen, sixteen, seventeen and eighteen of the periodic table. Sentence- One group of the representative elements is the Halogens. ...
Periodic Table Organization Comprehension Questions
... Directions: Read each question carefully. Be sure to answer each question fully and with detail. If you think you can add more to your response, do so. ...
... Directions: Read each question carefully. Be sure to answer each question fully and with detail. If you think you can add more to your response, do so. ...
The Periodic Table of the Elements
... elements were organized by increasing atomic mass, certain similarities in their chemical properties occurred at regular intervals. Such repeating patterns are referred to as PERIODIC. • Mendeleev created a table in which elements with similar properties were grouped together—the first Periodic Tabl ...
... elements were organized by increasing atomic mass, certain similarities in their chemical properties occurred at regular intervals. Such repeating patterns are referred to as PERIODIC. • Mendeleev created a table in which elements with similar properties were grouped together—the first Periodic Tabl ...
Section 12.3
... Explain how the periodic table is organized. Use the periodic table to become familiar with groups of elements and their properties. Identify metals, semimetals, and nonmetals on the periodic table. ...
... Explain how the periodic table is organized. Use the periodic table to become familiar with groups of elements and their properties. Identify metals, semimetals, and nonmetals on the periodic table. ...
Reading the Periodic Table - Science
... A way of organizing & classifying elements according to their PROPERTIES • Arranged in rows and columns ...
... A way of organizing & classifying elements according to their PROPERTIES • Arranged in rows and columns ...
Periodic Trends Student
... • Scientists involved with making the periodic table: – Mendeleev – Came up with the first Periodic Table • Arranged elements by increasing ATOMIC MASSES (Not quite right) ...
... • Scientists involved with making the periodic table: – Mendeleev – Came up with the first Periodic Table • Arranged elements by increasing ATOMIC MASSES (Not quite right) ...
The Periodic table
... electrons”. They are shiny, ductile and malleable. Metals are located to the left of the stair-step line found on the periodic table. ...
... electrons”. They are shiny, ductile and malleable. Metals are located to the left of the stair-step line found on the periodic table. ...
Students` Work* Introduction to Chemistry
... Table is the Red Stair Case which separates the metals from nonmetals. Metals on the left and gasses on the right. In each element box there are three parts the Atomic Symbol which is an abbreviation of the element. The Average Atomic Mass which is the weight of the element and if you subtract the A ...
... Table is the Red Stair Case which separates the metals from nonmetals. Metals on the left and gasses on the right. In each element box there are three parts the Atomic Symbol which is an abbreviation of the element. The Average Atomic Mass which is the weight of the element and if you subtract the A ...
Name: Date: _____ Pd: _____ Chemistry, PERIODIC TABLE Spring
... 13. Explain, in terms of atomic structure, why Group 18 elements on the Periodic Table rarely form compounds. Base your answers to questions 14 and 15 on the information below and on your knowledge of chemistry. Silver-plated utensils were popular before stainless steel became widely used to make ea ...
... 13. Explain, in terms of atomic structure, why Group 18 elements on the Periodic Table rarely form compounds. Base your answers to questions 14 and 15 on the information below and on your knowledge of chemistry. Silver-plated utensils were popular before stainless steel became widely used to make ea ...
Chemistry Chapter 5 The Periodic Law
... In 1869, Dmitri Mendeleev published his first “periodic table”. – He started by placing the known elements in order using their atomic masses. – He recognized that certain properties repeated themselves “periodically”. – He then rearranged the elements so that elements with similar properties appear ...
... In 1869, Dmitri Mendeleev published his first “periodic table”. – He started by placing the known elements in order using their atomic masses. – He recognized that certain properties repeated themselves “periodically”. – He then rearranged the elements so that elements with similar properties appear ...
PERIODIC TRENDS PRACTICE QUIZ
... a. Lower right-hand corner of the periodic table. b. Upper right-hand corner of the periodic table. c. Lower left-hand corner of the periodic table. d. Upper left-hand corner of the periodic table. 8. Of the following elements, which one would have the smallest ionization energy? a. Neon b. Lithium ...
... a. Lower right-hand corner of the periodic table. b. Upper right-hand corner of the periodic table. c. Lower left-hand corner of the periodic table. d. Upper left-hand corner of the periodic table. 8. Of the following elements, which one would have the smallest ionization energy? a. Neon b. Lithium ...
PERIODIC TRENDS PRACTICE QUIZ
... a. Lower right-hand corner of the periodic table. b. Upper right-hand corner of the periodic table. c. Lower left-hand corner of the periodic table. d. Upper left-hand corner of the periodic table. 8. Of the following elements, which one would have the smallest ionization energy? a. Neon b. Lithium ...
... a. Lower right-hand corner of the periodic table. b. Upper right-hand corner of the periodic table. c. Lower left-hand corner of the periodic table. d. Upper left-hand corner of the periodic table. 8. Of the following elements, which one would have the smallest ionization energy? a. Neon b. Lithium ...
powerpoint
... take chemistry.It was designed by a Russian chemist in 1869 by the name of Dimitri Mendeleev. The periodic table arranges all the elements in columns and rows in order of increasing atomic number. The columns are known as periods and the rows are known as groups. The design of the periodic table has ...
... take chemistry.It was designed by a Russian chemist in 1869 by the name of Dimitri Mendeleev. The periodic table arranges all the elements in columns and rows in order of increasing atomic number. The columns are known as periods and the rows are known as groups. The design of the periodic table has ...
Document
... Elements are organized in the periodic table by increasing atomic number. In the late 1800’s, Dmitri Mendeleev devised the first periodic table based on atomic mass. In 1913, Henry G. J. Moseley arranged the elements by atomic number rather than atomic mass. ...
... Elements are organized in the periodic table by increasing atomic number. In the late 1800’s, Dmitri Mendeleev devised the first periodic table based on atomic mass. In 1913, Henry G. J. Moseley arranged the elements by atomic number rather than atomic mass. ...
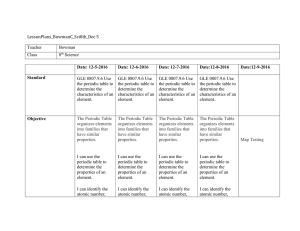
LessonPlans_BowmanC_Sci8th_Dec 5 Teacher Bowman Class 8th
... (Then First Letter of Mendeleev Video Last Name) IV. Count the Compare Days of Table (How many the Week, Groups, How many Calendars, Cards Periods) II. Periodic Table with A Video Series about Mendeleev: Mendeleev Video III. Review Atomic Symbols, Atomic Mass, Atomic Number IV. We Do: Periodic Tabl ...
... (Then First Letter of Mendeleev Video Last Name) IV. Count the Compare Days of Table (How many the Week, Groups, How many Calendars, Cards Periods) II. Periodic Table with A Video Series about Mendeleev: Mendeleev Video III. Review Atomic Symbols, Atomic Mass, Atomic Number IV. We Do: Periodic Tabl ...
periodictrendsss - rlsciencecurriculum
... When elements are listed in order by their atomic numbers and grouped according to similar properties, they form seven horizontal rows called periods. Each vertical column in the table contains elements with similar properties. These are called groups or families. The alkali metal family for example ...
... When elements are listed in order by their atomic numbers and grouped according to similar properties, they form seven horizontal rows called periods. Each vertical column in the table contains elements with similar properties. These are called groups or families. The alkali metal family for example ...
File
... elements that had not yet been discovered would fit right in AND HE WAS RIGHT! The new elements fit his pattern They later realized that it was more useful to group the elements based on atomic number instead of mass and this led to what we now call the Periodic Table ...
... elements that had not yet been discovered would fit right in AND HE WAS RIGHT! The new elements fit his pattern They later realized that it was more useful to group the elements based on atomic number instead of mass and this led to what we now call the Periodic Table ...
Chemistry test Review
... 7. If you were given samples of two different elements, but they were not labeled, how could you determine what they are? • Density • Color • Melting point • Boiling point • texture ...
... 7. If you were given samples of two different elements, but they were not labeled, how could you determine what they are? • Density • Color • Melting point • Boiling point • texture ...
File
... • Periodic Law: when elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. ...
... • Periodic Law: when elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. ...
Chapter 2 The Periodic Table
... c. Note: He, Ne, Ar, and Kr were undiscovered d. Introduced the idea of periodicity ...
... c. Note: He, Ne, Ar, and Kr were undiscovered d. Introduced the idea of periodicity ...
Dmitri Mendeleev

Dmitri Ivanovich Mendeleev (/ˌmɛndəlˈeɪəf/; Russian: Дми́трий Ива́нович Менделе́ев; IPA: [ˈdmʲitrʲɪj ɪˈvanəvʲɪtɕ mʲɪndʲɪˈlʲejɪf]; 8 February 1834 – 2 February 1907 O.S. 27 January 1834 – 20 January 1907) was a Russian chemist and inventor. He formulated the Periodic Law, created his own version of the periodic table of elements, and used it to correct the properties of some already discovered elements and also to predict the properties of eight elements yet to be discovered.