Week 9 (wk9) - Riverside Local Schools
... outter s and d electron number is equal to the… 2. The d-block elements are metals with typical metallic properties and are often referred to as… 3. Transition elements are good… 4. Of the d-block elements, palladium, platinum, and gold are among the least… p-BLOCK ELEMENTS: GROUPS 13-18 (pg. 136-13 ...
... outter s and d electron number is equal to the… 2. The d-block elements are metals with typical metallic properties and are often referred to as… 3. Transition elements are good… 4. Of the d-block elements, palladium, platinum, and gold are among the least… p-BLOCK ELEMENTS: GROUPS 13-18 (pg. 136-13 ...
Sept. 28th powerpoint
... • Measure the bulk densities and calculate atomic densities of the three transition elements in the same raw of the periodic table. • Relate measured density to atomic size, a periodic trend. ...
... • Measure the bulk densities and calculate atomic densities of the three transition elements in the same raw of the periodic table. • Relate measured density to atomic size, a periodic trend. ...
Chemical Periodicity - Fort Thomas Independent Schools
... chemical properties of the missing elements. Eventually these elements were discovered and were found to have properties similar to those predicted. There were many exceptions in his table, however. ...
... chemical properties of the missing elements. Eventually these elements were discovered and were found to have properties similar to those predicted. There were many exceptions in his table, however. ...
Chemical Periodicity - Fort Thomas Independent Schools
... chemical properties of the missing elements. Eventually these elements were discovered and were found to have properties similar to those predicted. There were many exceptions in his table, however. ...
... chemical properties of the missing elements. Eventually these elements were discovered and were found to have properties similar to those predicted. There were many exceptions in his table, however. ...
The periodic table shows all the elements and their
... The periodic table is structured as an 18 X 7 grid, positioned above a smaller double row of elements. The periodic table only lists chemical elements, and includes each isotope of each element within one cell. In the typical periodic table, each element is listed by its element symbol and atomic nu ...
... The periodic table is structured as an 18 X 7 grid, positioned above a smaller double row of elements. The periodic table only lists chemical elements, and includes each isotope of each element within one cell. In the typical periodic table, each element is listed by its element symbol and atomic nu ...
d. Group 1
... explain why aluminum (Al) is a better choice than magnesium (Mg) or Iron (Fe) for certain real world uses such as boats and house siding. ...
... explain why aluminum (Al) is a better choice than magnesium (Mg) or Iron (Fe) for certain real world uses such as boats and house siding. ...
REVIEW Through Course Task
... ________ side of the periodic table while the LEAST METALLIC ELEMENTS would be found on the LEFT ____________ side of the periodic table. RIGHT 10. While metals come in many colors, most at the far left side of the periodic table are very ____________ in color and tend to become more _________ or da ...
... ________ side of the periodic table while the LEAST METALLIC ELEMENTS would be found on the LEFT ____________ side of the periodic table. RIGHT 10. While metals come in many colors, most at the far left side of the periodic table are very ____________ in color and tend to become more _________ or da ...
Activity 16 Elements and the Periodic Table
... Do you see another of the patterns that Mendeleev and other scientists recognized? ___________ Neutral atoms contain equal numbers of protons (+) and electrons (-). Because of this, you know that neutral atoms of lithium, beryllium, boron and carbon also have 3, 4, 5, and 6 electrons respectively. W ...
... Do you see another of the patterns that Mendeleev and other scientists recognized? ___________ Neutral atoms contain equal numbers of protons (+) and electrons (-). Because of this, you know that neutral atoms of lithium, beryllium, boron and carbon also have 3, 4, 5, and 6 electrons respectively. W ...
THE PERIODIC TABLE
... • Describe the historical development of the periodic table. • Describe the organization of the modern periodic table according to the periodic law. A. Patterns in Element Properties 1. The elements vary widely in their properties, but in an orderly way. 2. English chemist, John Newlands was the fir ...
... • Describe the historical development of the periodic table. • Describe the organization of the modern periodic table according to the periodic law. A. Patterns in Element Properties 1. The elements vary widely in their properties, but in an orderly way. 2. English chemist, John Newlands was the fir ...
Chapter 5 - The Periodic Law
... 1. Vertical columns in atomic weight order a. Mendeleev made some exceptions to place elements in rows with similar properties (1) Tellurium and iodine's places were switched 2. Horizontal rows have similar chemical properties B. Missing Elements 1. Gaps existed in Mendeleev’s table a. Mendeleev pre ...
... 1. Vertical columns in atomic weight order a. Mendeleev made some exceptions to place elements in rows with similar properties (1) Tellurium and iodine's places were switched 2. Horizontal rows have similar chemical properties B. Missing Elements 1. Gaps existed in Mendeleev’s table a. Mendeleev pre ...
The Periodic Table and Periodic Law
... Using atomic number instead of atomic mass as the organizing principle was first proposed by the British chemist Henry Moseley in 1913, and it solved anomalies like this one. Iodine has a higher atomic number than tellurium - so, even though he didn't know why, Mendeleev was right to place it after ...
... Using atomic number instead of atomic mass as the organizing principle was first proposed by the British chemist Henry Moseley in 1913, and it solved anomalies like this one. Iodine has a higher atomic number than tellurium - so, even though he didn't know why, Mendeleev was right to place it after ...
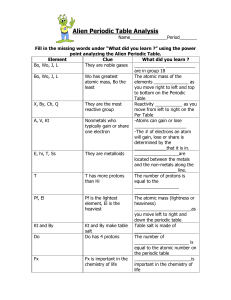
Alien Periodic Table Analysis
... They are the most Reactivity ___________ as you reactive group move from left to right on the Per Table A, V, Kt Nonmetals who -Atoms can gain or lose typically gain or share _____________________ one electron -The # of electrons an atom will gain, lose or share is determined by the _____________tha ...
... They are the most Reactivity ___________ as you reactive group move from left to right on the Per Table A, V, Kt Nonmetals who -Atoms can gain or lose typically gain or share _____________________ one electron -The # of electrons an atom will gain, lose or share is determined by the _____________tha ...
Elements and the Periodic Table
... Scientists call these allotropes. One example of this is carbon. Depending on how carbon atoms fit together they can form diamond, coal, or graphite. How many elements are there? There are currently 118 known elements. Of these, only 94 are thought to naturally exist on Earth. Families of Elements E ...
... Scientists call these allotropes. One example of this is carbon. Depending on how carbon atoms fit together they can form diamond, coal, or graphite. How many elements are there? There are currently 118 known elements. Of these, only 94 are thought to naturally exist on Earth. Families of Elements E ...
PPT Test Review
... elements in the periodic table, the number of valence electrons____________ from left to right. ...
... elements in the periodic table, the number of valence electrons____________ from left to right. ...
Labeling a Blank Periodic Table
... any member of the nonmetallic elements in Group VII A in the periodic table any member of a group of gaseous elements in Group VIII A in the periodic table ...
... any member of the nonmetallic elements in Group VII A in the periodic table any member of a group of gaseous elements in Group VIII A in the periodic table ...
Day 3
... poster about. The poster will include: 1) The atomic symbol. 2) Information about the number of protons, neutrons, and electrons in each atom. 3) A model of the atom. 4) Historical information (such as who discovered it, if applicable, the derivation of the name, etc.) Extend -- Time Estimate: negli ...
... poster about. The poster will include: 1) The atomic symbol. 2) Information about the number of protons, neutrons, and electrons in each atom. 3) A model of the atom. 4) Historical information (such as who discovered it, if applicable, the derivation of the name, etc.) Extend -- Time Estimate: negli ...
Focus On Physical Science
... • The number of protons in an atom of an element is the element’s atomic number. • The periodic table shows elements that increase in atomic number horizontally. • Elements in the same column have similar properties. ...
... • The number of protons in an atom of an element is the element’s atomic number. • The periodic table shows elements that increase in atomic number horizontally. • Elements in the same column have similar properties. ...
SCH3U Periodic Table Worksheet 1. Where are the most active
... 1. Where are the most active metals located? Group 1. Also, the bottom periods of the periodic table. 2. Where are the most active non-metals located? Top right of the periodic table. Fluorine being the highest. Group 17 very reactive. 3. As you go from left to right across a period, the atomic radi ...
... 1. Where are the most active metals located? Group 1. Also, the bottom periods of the periodic table. 2. Where are the most active non-metals located? Top right of the periodic table. Fluorine being the highest. Group 17 very reactive. 3. As you go from left to right across a period, the atomic radi ...
Periodic Table Worksheet 1. Where are the most active metals
... 1. Where are the most active metals located? ____________Group 1____________ 2. Where are the most active non-metals located? _______________Group 17 _______________ 3. As you go from left to right across a period, the atomic radius (increases/decreases). Why? Greater ENC, stronger force of attracti ...
... 1. Where are the most active metals located? ____________Group 1____________ 2. Where are the most active non-metals located? _______________Group 17 _______________ 3. As you go from left to right across a period, the atomic radius (increases/decreases). Why? Greater ENC, stronger force of attracti ...
The Periodic Table/Trends Chapter 5
... The Metalloids or Semimetals have some characteristics of both ...
... The Metalloids or Semimetals have some characteristics of both ...
hc1(5)notes
... elements were arranged in order of increasing atomic weight, certain similarities in their chemical properties appeared at regular intervals; not the first to do this though (John Newlands). • Repeating patterns are referred to as periodic. • Mendeleev created a table in which elements with similar ...
... elements were arranged in order of increasing atomic weight, certain similarities in their chemical properties appeared at regular intervals; not the first to do this though (John Newlands). • Repeating patterns are referred to as periodic. • Mendeleev created a table in which elements with similar ...
Dmitri Mendeleev

Dmitri Ivanovich Mendeleev (/ˌmɛndəlˈeɪəf/; Russian: Дми́трий Ива́нович Менделе́ев; IPA: [ˈdmʲitrʲɪj ɪˈvanəvʲɪtɕ mʲɪndʲɪˈlʲejɪf]; 8 February 1834 – 2 February 1907 O.S. 27 January 1834 – 20 January 1907) was a Russian chemist and inventor. He formulated the Periodic Law, created his own version of the periodic table of elements, and used it to correct the properties of some already discovered elements and also to predict the properties of eight elements yet to be discovered.