Chemistry 4.1 Atomic structure and the periodic table NEED TO
... Before the discovery of protons, neutrons and electrons scientists attempted to classify the elements by arranging them in order of their atomic weights. The early periodic tables were incomplete and some elements were placed in inappropriate groups if the strict order of atomic weights was followed ...
... Before the discovery of protons, neutrons and electrons scientists attempted to classify the elements by arranging them in order of their atomic weights. The early periodic tables were incomplete and some elements were placed in inappropriate groups if the strict order of atomic weights was followed ...
Periodic Trends
... • Dimitri Mendeleev was the first scientist to publish an organized periodic table of the known elements. • He was perpetually in trouble with the Russian government and the Russian Orthodox Church, but he was brilliant never-the-less. ...
... • Dimitri Mendeleev was the first scientist to publish an organized periodic table of the known elements. • He was perpetually in trouble with the Russian government and the Russian Orthodox Church, but he was brilliant never-the-less. ...
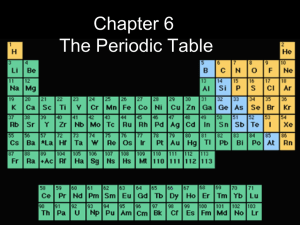
Study Guide Chapter 6
... (f) semiconductor: An element which is neither a good conductor or a good insulator, but rather lies somewhere between the two. ...
... (f) semiconductor: An element which is neither a good conductor or a good insulator, but rather lies somewhere between the two. ...
the periodic table
... CHEMISTRY TEST REVIEW Use this as a study tool to review for your CCA, October 18 th. ...
... CHEMISTRY TEST REVIEW Use this as a study tool to review for your CCA, October 18 th. ...
The Periodic Table
... Mendeleev, the father of the PT, organized the elements by ________________. The modern PT is organized by _____________. Three general categories or elements include ____________, ______________, and ___________. The Periodic Law states that when elements are organized by ________ _________ _______ ...
... Mendeleev, the father of the PT, organized the elements by ________________. The modern PT is organized by _____________. Three general categories or elements include ____________, ______________, and ___________. The Periodic Law states that when elements are organized by ________ _________ _______ ...
What is matter? - Waterford Public Schools
... • One can calculate the average atomic weight of an element if the abundance of each isotope for that element is known Average Atomic Mass % natural abundance ...
... • One can calculate the average atomic weight of an element if the abundance of each isotope for that element is known Average Atomic Mass % natural abundance ...
Periodic Table WebQuest
... 9. Is the quantity of protons always equal to the quantity of neutrons in an atom? Explain. 10. List 3 uses of isotopes. 11. Describe how scientists calculate the average atomic mass of an element. Use http://www.chem4kids.com/files/elem_intro.html, http://www.chem4kids.com/files/elem_transmetal.ht ...
... 9. Is the quantity of protons always equal to the quantity of neutrons in an atom? Explain. 10. List 3 uses of isotopes. 11. Describe how scientists calculate the average atomic mass of an element. Use http://www.chem4kids.com/files/elem_intro.html, http://www.chem4kids.com/files/elem_transmetal.ht ...
Chemistry 4.2
... able to position elements more properly on the Periodic Table • Changed the way the PT appeared ...
... able to position elements more properly on the Periodic Table • Changed the way the PT appeared ...
Ch_6_Notes_Periodic_Table
... The atomic radius decreases from left to right across a period in the periodic table. Lithium has the largest atomic radius in the second period. As the number of electrons added to the same energy level increases, atomic size generally decreases. List the symbols for sodium, sulfur, and cesium in o ...
... The atomic radius decreases from left to right across a period in the periodic table. Lithium has the largest atomic radius in the second period. As the number of electrons added to the same energy level increases, atomic size generally decreases. List the symbols for sodium, sulfur, and cesium in o ...
Chemistry 1 Chapter 4, The Periodic Table
... •he also predicted the properties of these missing elements based on the column they were in ...
... •he also predicted the properties of these missing elements based on the column they were in ...
Unit One Periodicity of Elements and their Properties
... 3- Discovered that the nucleus of the atom contains positively charged protons. ...
... 3- Discovered that the nucleus of the atom contains positively charged protons. ...
Periodic Table - MunterChemistry
... Dmitri Mendeleev • Russian chemist • Created a table by arranging elements according to atomic masses • Noticed that chemical properties of the elements followed a repeating pattern ...
... Dmitri Mendeleev • Russian chemist • Created a table by arranging elements according to atomic masses • Noticed that chemical properties of the elements followed a repeating pattern ...
(FOR STUDENTS 2015)
... Atomic Radii - distance from the nucleus to outermost electron. Ionization Energy - energy required to remove an electron (kJ/mol) Electron Affinity – energy change when neutral atom gains electron ...
... Atomic Radii - distance from the nucleus to outermost electron. Ionization Energy - energy required to remove an electron (kJ/mol) Electron Affinity – energy change when neutral atom gains electron ...
Unit 3.2 worksheet 4 atomic model of matter
... Tips and tricks! Hope I help :)) Video Rating: / 5. Click Here - Movie Star Planet. Hi i am writing u to ask what is the state requirments while growing for person medical needs. what will make it completely legal where if visited by the law i wanna. Getting Started. USATestprep is very user-friendl ...
... Tips and tricks! Hope I help :)) Video Rating: / 5. Click Here - Movie Star Planet. Hi i am writing u to ask what is the state requirments while growing for person medical needs. what will make it completely legal where if visited by the law i wanna. Getting Started. USATestprep is very user-friendl ...
week 13 - My CCSD
... THE STUDENT WILL RELATE THE PERIODIC LAW TO THE POSITIONS OF ELEMENTS ON THE PERIODIC TABLE. [P.12.A.2] 5.13 THE STUDENT WILL PREDICT THE VALENCE ORBITALS FILLED IN THE EIGHT MAIN GROUPS (REPRESENTATIVE ELEMENTS). [P.12.A.9] OBJECTIVE: The student will be able to: ...
... THE STUDENT WILL RELATE THE PERIODIC LAW TO THE POSITIONS OF ELEMENTS ON THE PERIODIC TABLE. [P.12.A.2] 5.13 THE STUDENT WILL PREDICT THE VALENCE ORBITALS FILLED IN THE EIGHT MAIN GROUPS (REPRESENTATIVE ELEMENTS). [P.12.A.9] OBJECTIVE: The student will be able to: ...
Honors Chemistry- Chapter 5 Homework Packet The Periodic Law
... 2) What is the relationship between the electron configuration of an element and the period in which that element appears on the periodic table? ...
... 2) What is the relationship between the electron configuration of an element and the period in which that element appears on the periodic table? ...
The Periodic Table
... Atoms of the same element always have the same number of protons. This identifies them as the element that they are. But all atoms of an element don’t have to have the same number of neutrons. For example, all boron atoms have 5 protons. However, four-fifths of them have 6 neutrons and one-fifth of ...
... Atoms of the same element always have the same number of protons. This identifies them as the element that they are. But all atoms of an element don’t have to have the same number of neutrons. For example, all boron atoms have 5 protons. However, four-fifths of them have 6 neutrons and one-fifth of ...
Periodic Table
... have at least 2 elements in that group. You must also give your element a symbol similar in style to the symbols for the elements in the lab. In other words your symbol must have a capital letter and a small letter, and elements in the same group must start with the same capital letter. Identifying ...
... have at least 2 elements in that group. You must also give your element a symbol similar in style to the symbols for the elements in the lab. In other words your symbol must have a capital letter and a small letter, and elements in the same group must start with the same capital letter. Identifying ...
Distinguishing Between Atoms
... 2. If the mass of 293Nv is 293.15 amu (red)and that of 295Nv is 295.15 amu (blue), what is the atomic mass of Nv? ...
... 2. If the mass of 293Nv is 293.15 amu (red)and that of 295Nv is 295.15 amu (blue), what is the atomic mass of Nv? ...
Chapter Test B
... In the space provided, list the charge of the ion most likely to be formed from the element and the name of the noble gas with an electron configuration achieved by that formation. (The atomic numbers of the noble gases are: He, 2; Ne, 10; Ar, 18; Kr, 36; Xe, 54; and Rn, 86.) ...
... In the space provided, list the charge of the ion most likely to be formed from the element and the name of the noble gas with an electron configuration achieved by that formation. (The atomic numbers of the noble gases are: He, 2; Ne, 10; Ar, 18; Kr, 36; Xe, 54; and Rn, 86.) ...
The Periodic Table of Elements - PAMS-Doyle
... atomic weight he could put them into 7 groups, or columns. • He found the pattern, all the elements in a column have the same valences, the number of electrons in the outer shell ...
... atomic weight he could put them into 7 groups, or columns. • He found the pattern, all the elements in a column have the same valences, the number of electrons in the outer shell ...
lab19
... 1. Properties such as atomic radius and energy required to remove the easiest electron are periodic functions of their atomic numbers. 2. As the atomic number increases, electrons increase, which causes there to be more valence electrons, and results in a larger atom (larger atomic radius value). 3. ...
... 1. Properties such as atomic radius and energy required to remove the easiest electron are periodic functions of their atomic numbers. 2. As the atomic number increases, electrons increase, which causes there to be more valence electrons, and results in a larger atom (larger atomic radius value). 3. ...
Sub Unit Plan 1 Chem Periodic Table
... II.3 Elements can be classified by their properties and located on the Periodic Table as metals, nonmetals, metalloids (B, Si, Ge, As, Sb, Te), and noble gases. (3.1v) II.4 Elements can be differentiated by their physical properties. Physical properties of substances, such as density, conductivity, ...
... II.3 Elements can be classified by their properties and located on the Periodic Table as metals, nonmetals, metalloids (B, Si, Ge, As, Sb, Te), and noble gases. (3.1v) II.4 Elements can be differentiated by their physical properties. Physical properties of substances, such as density, conductivity, ...
Dmitri Mendeleev

Dmitri Ivanovich Mendeleev (/ˌmɛndəlˈeɪəf/; Russian: Дми́трий Ива́нович Менделе́ев; IPA: [ˈdmʲitrʲɪj ɪˈvanəvʲɪtɕ mʲɪndʲɪˈlʲejɪf]; 8 February 1834 – 2 February 1907 O.S. 27 January 1834 – 20 January 1907) was a Russian chemist and inventor. He formulated the Periodic Law, created his own version of the periodic table of elements, and used it to correct the properties of some already discovered elements and also to predict the properties of eight elements yet to be discovered.