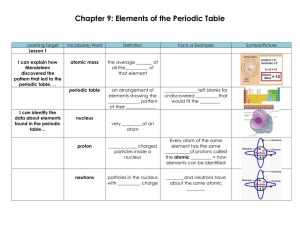
Chapter 9: Elements of the Periodic Table
... I can describe how metals are classified in the periodic table… Metals are classified as ____________metals, _____________________ metals, ______________ metals, metals in mixed groups, __________________, and ___________________. Lesson 3 I can summarize the properties of nonmetals… In general, mos ...
... I can describe how metals are classified in the periodic table… Metals are classified as ____________metals, _____________________ metals, ______________ metals, metals in mixed groups, __________________, and ___________________. Lesson 3 I can summarize the properties of nonmetals… In general, mos ...
File - dr. stephen alfred
... Most Periodic Table models also indicate whether an element is a solid, liquid, or gas. ...
... Most Periodic Table models also indicate whether an element is a solid, liquid, or gas. ...
the modern periodic law
... - in 1863, he arranged the known elements in order of increasing atomic mass - he noticed that similar chemical and physical properties occurred every eight element: ex: atoms 2, 9 and 16 (Li, Na and K) resembled each other chemically - he classified the 49 known elements in seven groups of seven el ...
... - in 1863, he arranged the known elements in order of increasing atomic mass - he noticed that similar chemical and physical properties occurred every eight element: ex: atoms 2, 9 and 16 (Li, Na and K) resembled each other chemically - he classified the 49 known elements in seven groups of seven el ...
Electron Energy Level
... • By the mid-1800s, about 70 elements were known to exist • Dmitri Mendeleev – a Russian chemist and teacher • Arranged elements in order of increasing atomic mass • Thus, the first “Periodic Table” ...
... • By the mid-1800s, about 70 elements were known to exist • Dmitri Mendeleev – a Russian chemist and teacher • Arranged elements in order of increasing atomic mass • Thus, the first “Periodic Table” ...
Chapter 5 Organizing The Elements
... • Describe how properties of elements change across a period in the periodic table • Relate the number of valence electrons to groups in the periodic table and to properties of elements in those groups • Predict the reactivity of some elements based on their locations within a group • Identify some ...
... • Describe how properties of elements change across a period in the periodic table • Relate the number of valence electrons to groups in the periodic table and to properties of elements in those groups • Predict the reactivity of some elements based on their locations within a group • Identify some ...
Periodic Properties of the Elements
... 1. In Exploration 1, select the element from period 2, group 1, on the periodic table. When selected, the group and period number will be highlighted by red circles. 2. Select the element’s period (row) number, represented by a red circle. 3. Record the element’s properties from the list provided in ...
... 1. In Exploration 1, select the element from period 2, group 1, on the periodic table. When selected, the group and period number will be highlighted by red circles. 2. Select the element’s period (row) number, represented by a red circle. 3. Record the element’s properties from the list provided in ...
The placement of an element on the periodic table gives clues about
... The periodic table is so named because it is organized into "periods". A period is defined as an interval required for a cycle to repeat itself. In the periodic table, the periods are the horizontal rows that extend from left to right. These periods consist of as few as two elements and as many as t ...
... The periodic table is so named because it is organized into "periods". A period is defined as an interval required for a cycle to repeat itself. In the periodic table, the periods are the horizontal rows that extend from left to right. These periods consist of as few as two elements and as many as t ...
File
... 6. List five properties that all (or almost all) metals have in common. 7. List five properties that most solid non-metals have in common. 8. As you move down the groups in the periodic table, how does the reactivity change for: a. ...
... 6. List five properties that all (or almost all) metals have in common. 7. List five properties that most solid non-metals have in common. 8. As you move down the groups in the periodic table, how does the reactivity change for: a. ...
Topic 1 – Atomic structure and the periodic table
... o realised from the big jumps in atomic mass that there were still some elements to discoverleft some gaps in his table E.g he left two gaps between zinc and arsenic Based on the known elements around them, Mendeleev predicted the properties of the elements which should go in the gaps In the ...
... o realised from the big jumps in atomic mass that there were still some elements to discoverleft some gaps in his table E.g he left two gaps between zinc and arsenic Based on the known elements around them, Mendeleev predicted the properties of the elements which should go in the gaps In the ...
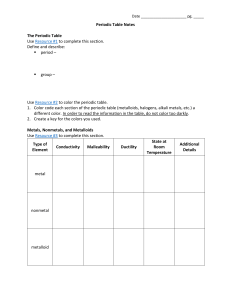
Periodic Table Notes The Periodic Table Use Resource #1
... Periodic Table Notes The Periodic Table Use Resource #1 to complete this section. Define and describe: period – ...
... Periodic Table Notes The Periodic Table Use Resource #1 to complete this section. Define and describe: period – ...
Ch 5 power point
... History of the Periodic Table - Mosley • In 1911, English scientists Mosley and Rutherford recognized that elements fit into patterns better when they were arranged according to increasing number of protons, rather than increasing atomic mass. • Mosley’s work led to the modern definition of atomic ...
... History of the Periodic Table - Mosley • In 1911, English scientists Mosley and Rutherford recognized that elements fit into patterns better when they were arranged according to increasing number of protons, rather than increasing atomic mass. • Mosley’s work led to the modern definition of atomic ...
Unit Six: Atomic structure
... 1. Elements are arranged on the periodic table according to similar properties. 2. The modern periodic table is organized by increasing atomic number. ...
... 1. Elements are arranged on the periodic table according to similar properties. 2. The modern periodic table is organized by increasing atomic number. ...
Ch. 6 - The Periodic Table
... Mendeleev organized elements into groups based on a set of repeating properties and according to increasing ...
... Mendeleev organized elements into groups based on a set of repeating properties and according to increasing ...
Periodic Table Powerpoint
... Noble Gases are colorless gases that are extremely unreactive. One important property of the noble gases is their inactivity. They are inactive because their outermost energy level is full. Because they do not readily combine with other elements to form compounds, the noble gases are called inert. T ...
... Noble Gases are colorless gases that are extremely unreactive. One important property of the noble gases is their inactivity. They are inactive because their outermost energy level is full. Because they do not readily combine with other elements to form compounds, the noble gases are called inert. T ...
Ionization Energy
... On the periodic table, elements are arranged least to greatest by atomic number, which is the number of protons in the nucleus. The periodic table is also arranged in groups which are vertical columns of elements, and periods, which are the horizontal rows of elements. Each element has ionization en ...
... On the periodic table, elements are arranged least to greatest by atomic number, which is the number of protons in the nucleus. The periodic table is also arranged in groups which are vertical columns of elements, and periods, which are the horizontal rows of elements. Each element has ionization en ...
Periodic Table Notes.notebook
... Why? Elements near the top of the period table have few electrons to begin with; every electron is a big deal. They have a stronger desire to acquire more electrons. Elements near the bottom of the chart have so many electrons that loosing or acquiring an electron is not as big a deal. This is du ...
... Why? Elements near the top of the period table have few electrons to begin with; every electron is a big deal. They have a stronger desire to acquire more electrons. Elements near the bottom of the chart have so many electrons that loosing or acquiring an electron is not as big a deal. This is du ...
Chapter 4 - Blair Community Schools
... 63 elements in a table to show similarities & periodic trends by increase atomic mass. Chemical & physical properties w/ symbols of each element on cards. Gaps in table, predicted properties of missing elements (Ga, Sc & Ge) Not all fit by increasing atomic mass (Te & I), needed to switch but did no ...
... 63 elements in a table to show similarities & periodic trends by increase atomic mass. Chemical & physical properties w/ symbols of each element on cards. Gaps in table, predicted properties of missing elements (Ga, Sc & Ge) Not all fit by increasing atomic mass (Te & I), needed to switch but did no ...
File u1 sec2.2 slide show
... Periods: Horizontal rows Numbered 1 to 7 Q: What period is Nickel located? _____ Q: How many elements are in period 1? _____ ...
... Periods: Horizontal rows Numbered 1 to 7 Q: What period is Nickel located? _____ Q: How many elements are in period 1? _____ ...
Science 90: Physical Science – Atoms and Elements Outcome: AE9
... CaCO₃, SO₂, FeO, NO₂, O₃, CH₄, C₃H₈, NH₃, NaHCO₃, KCl, HCl, H₂SO₄, ZnO, and NaCl). h. Construct Bohr model representations of the first 18 elements. i. Trace the historical development of the modern periodic table and compare alternative arrangements that convey information about the classification ...
... CaCO₃, SO₂, FeO, NO₂, O₃, CH₄, C₃H₈, NH₃, NaHCO₃, KCl, HCl, H₂SO₄, ZnO, and NaCl). h. Construct Bohr model representations of the first 18 elements. i. Trace the historical development of the modern periodic table and compare alternative arrangements that convey information about the classification ...
February 10 Clicker Questions
... 1. [Ne]3p7 2. [Ne]3s23p5 3. [Ne]3s23p6 4. [Ne]3s23d5 5. [Ne]3s23p33d2 Note: 1 is impossible (p-orbitals can hold a maximum of 6 electrons), 4 and 5 are not ground state configurations, while 3 has 18 electrons. ...
... 1. [Ne]3p7 2. [Ne]3s23p5 3. [Ne]3s23p6 4. [Ne]3s23d5 5. [Ne]3s23p33d2 Note: 1 is impossible (p-orbitals can hold a maximum of 6 electrons), 4 and 5 are not ground state configurations, while 3 has 18 electrons. ...
File
... The modern periodic table The modern periodic table arranges elements in order of increasing atomic number, not atomic mass. Scientists have been adding elements to the periodic table, as more are discovered or created. The last naturally occurring element to be discovered is Francium (Fr) in 1939. ...
... The modern periodic table The modern periodic table arranges elements in order of increasing atomic number, not atomic mass. Scientists have been adding elements to the periodic table, as more are discovered or created. The last naturally occurring element to be discovered is Francium (Fr) in 1939. ...
The Periodic Table - Science
... The atomic mass is the average mass of an element in atomic mass units ("amu"). Though individual atoms always have a whole number of amus, the atomic mass on the periodic table is shown as a decimal number because it is an average of all the isotopes of an element. ...
... The atomic mass is the average mass of an element in atomic mass units ("amu"). Though individual atoms always have a whole number of amus, the atomic mass on the periodic table is shown as a decimal number because it is an average of all the isotopes of an element. ...
Questions Periodic Table Live
... a new type of orbital, a g orbital, that has to be filled before the third lanthanide/actinide series; there are nine different g orbitals. Thus, element 164 would be below element ...
... a new type of orbital, a g orbital, that has to be filled before the third lanthanide/actinide series; there are nine different g orbitals. Thus, element 164 would be below element ...
Oct 17-Oct 21
... identify elements likely to form ions and use information to construct formulas for compounds ...
... identify elements likely to form ions and use information to construct formulas for compounds ...
The Periodic Table
... The representative elements in the s-block and p-block are more predictable and have less exceptions than the d-block and f-block elements. ...
... The representative elements in the s-block and p-block are more predictable and have less exceptions than the d-block and f-block elements. ...
Dmitri Mendeleev

Dmitri Ivanovich Mendeleev (/ˌmɛndəlˈeɪəf/; Russian: Дми́трий Ива́нович Менделе́ев; IPA: [ˈdmʲitrʲɪj ɪˈvanəvʲɪtɕ mʲɪndʲɪˈlʲejɪf]; 8 February 1834 – 2 February 1907 O.S. 27 January 1834 – 20 January 1907) was a Russian chemist and inventor. He formulated the Periodic Law, created his own version of the periodic table of elements, and used it to correct the properties of some already discovered elements and also to predict the properties of eight elements yet to be discovered.