8th Grade Science Notes Chapter 2
... Protons & Neutrons have the same relative mass Niels Bohr - proposed an atomic model that placed electrons in circular orbits called energy levels. Electron Cloud - the modern atomic model. Electrons move in an area represented as a cloud around the nucleus. Quarks - smaller particles that make up p ...
... Protons & Neutrons have the same relative mass Niels Bohr - proposed an atomic model that placed electrons in circular orbits called energy levels. Electron Cloud - the modern atomic model. Electrons move in an area represented as a cloud around the nucleus. Quarks - smaller particles that make up p ...
Isotopes
... left of the chemical symbol, For iron (Fe) we have, for example: 54Fe, 56Fe, 57Fe, and 58Fe. Since the iron has the atomic number zFe = 26, we have 54 - 26 = 28 neutrons in 54Fe, and 30, 31, and 32 neutrons, respectively, in the other three isotopes given. Isotopes come in two basic variants: 1. Rad ...
... left of the chemical symbol, For iron (Fe) we have, for example: 54Fe, 56Fe, 57Fe, and 58Fe. Since the iron has the atomic number zFe = 26, we have 54 - 26 = 28 neutrons in 54Fe, and 30, 31, and 32 neutrons, respectively, in the other three isotopes given. Isotopes come in two basic variants: 1. Rad ...
Homework 1B1 - 3 - Uddingston Grammar School
... (a) Complete the diagram to show how the electrons are arranged. You may wish to use the data booklet to help you. ...
... (a) Complete the diagram to show how the electrons are arranged. You may wish to use the data booklet to help you. ...
Dating the Earth Power Point
... • Alpha decay - Alpha decay is caused when there are too many protons in a nucleus. In this case the element will emit radiation in the form of positively charged particles called alpha particles. • Beta decay - Beta decay is caused when there are too many neutrons in a nucleus. In this case the ele ...
... • Alpha decay - Alpha decay is caused when there are too many protons in a nucleus. In this case the element will emit radiation in the form of positively charged particles called alpha particles. • Beta decay - Beta decay is caused when there are too many neutrons in a nucleus. In this case the ele ...
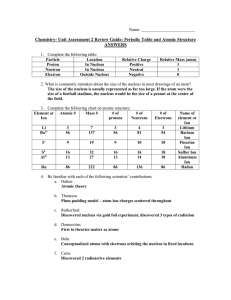
Honors Review Unit 2 answers
... Used an “oil drop” experiment to discover the charge and mass of an electron. ___Millikan__ Proposed the “Plum Pudding” model of the atom. _____Thomson________ Used the word “atomos” to describe matter. _____Democritos________ Called the “Father of Modern Chemistry”. ___Lavoisier_________ Discovered ...
... Used an “oil drop” experiment to discover the charge and mass of an electron. ___Millikan__ Proposed the “Plum Pudding” model of the atom. _____Thomson________ Used the word “atomos” to describe matter. _____Democritos________ Called the “Father of Modern Chemistry”. ___Lavoisier_________ Discovered ...
Keypoints of Basic Atomic Structure
... Atomic Number Atomic Radius Electrons Element Isotope Neutrons Periodic Table Protons Subatomic Particles Concepts 1. Be able to describe how protons, neutrons and electrons are arranged in an atom. 2. Be able to list the charges on the subatomic particles that make up and atom, and giv ...
... Atomic Number Atomic Radius Electrons Element Isotope Neutrons Periodic Table Protons Subatomic Particles Concepts 1. Be able to describe how protons, neutrons and electrons are arranged in an atom. 2. Be able to list the charges on the subatomic particles that make up and atom, and giv ...
atomic structure - IGCSE STUDY BANK
... There are small physical differences between the isotopes eg the heavier isotope has a greater density or boiling point. However, because they have the same number of protons they have the same electronic structure and identical chemically. Examples are illustrated below. Do NOT assume the word isot ...
... There are small physical differences between the isotopes eg the heavier isotope has a greater density or boiling point. However, because they have the same number of protons they have the same electronic structure and identical chemically. Examples are illustrated below. Do NOT assume the word isot ...
The_Atoms_Family
... • When the number of neutrons in nuclei vary, you have an isotope • For example, chlorine always has 17 protons, but the number of neutrons could be either 18 or 20. This in turn, changes the atomic mass • The nuclei of some isotopes are unstable and release radiation • During the radioactive decay ...
... • When the number of neutrons in nuclei vary, you have an isotope • For example, chlorine always has 17 protons, but the number of neutrons could be either 18 or 20. This in turn, changes the atomic mass • The nuclei of some isotopes are unstable and release radiation • During the radioactive decay ...
Physical Science Chapter 6 Study Guide Atomic Theory of Matter
... Parts of an atom o Nucleus—dense central core composed of protons and neutrons o Electrons surround the nucleus o Protons and neutrons are made up of small particles called quarks o Protons have positive charge and electrons have a negative charge ...
... Parts of an atom o Nucleus—dense central core composed of protons and neutrons o Electrons surround the nucleus o Protons and neutrons are made up of small particles called quarks o Protons have positive charge and electrons have a negative charge ...
Structure of the Atom
... • The mass number is the total number of protons and neutrons in the nucleus. • Atoms can have different numbers of neutrons. • Atoms that have different numbers of neutrons are called isotopes. ...
... • The mass number is the total number of protons and neutrons in the nucleus. • Atoms can have different numbers of neutrons. • Atoms that have different numbers of neutrons are called isotopes. ...
Chapter 18
... • Dimitri Mendeleevarranged all the elements known in order of increasing atomic masses and discovered a pattern • Today’s Periodic Table— elements are arranged by increasing atomic number and by changes in physical and chemical properties ...
... • Dimitri Mendeleevarranged all the elements known in order of increasing atomic masses and discovered a pattern • Today’s Periodic Table— elements are arranged by increasing atomic number and by changes in physical and chemical properties ...
Atomic Structure
... as a whole. • If an atom were the size of a football stadium, the nucleus would be about the size of a marble. ...
... as a whole. • If an atom were the size of a football stadium, the nucleus would be about the size of a marble. ...
Chem Unit 2 Review Guide ANSWERS
... Conservation of Mass apply to each type of reaction? Chemical reactions only involve the atoms’ valence electrons. In a nuclear reaction, the nucleus is actually altered. The Law of Conservation of Mass holds true during chemical reactions, but is not during a nuclear reaction, as mass is converted ...
... Conservation of Mass apply to each type of reaction? Chemical reactions only involve the atoms’ valence electrons. In a nuclear reaction, the nucleus is actually altered. The Law of Conservation of Mass holds true during chemical reactions, but is not during a nuclear reaction, as mass is converted ...
Periodic Table Quiz
... 3. An unknown sample is malleable and ductile. Where on the periodic table would the sample most likely be found? a. To the right of the zig-zag line. b. Bordering right on the zig-zag line. c. To the left of the zig-zag line. d. There are no elements that are both malleable and ductile found on the ...
... 3. An unknown sample is malleable and ductile. Where on the periodic table would the sample most likely be found? a. To the right of the zig-zag line. b. Bordering right on the zig-zag line. c. To the left of the zig-zag line. d. There are no elements that are both malleable and ductile found on the ...
Atomic Structure
... ISOTOPES Two atoms with the same # of protons (atomic #) but different #’s of neutrons (atomic masses) are ISOTOPES of the same element ...
... ISOTOPES Two atoms with the same # of protons (atomic #) but different #’s of neutrons (atomic masses) are ISOTOPES of the same element ...
Periodic Table
... protons) will appear at the __________ • The mass number (number of protons plus neutrons will appear at the __________ • The element symbol will appear to the __________ • The different number of neutrons has NO bearing on chemical reactivity ...
... protons) will appear at the __________ • The mass number (number of protons plus neutrons will appear at the __________ • The element symbol will appear to the __________ • The different number of neutrons has NO bearing on chemical reactivity ...
Extension 18.2: Isotopes
... The number of protons does determine which element it is, but the mass number essentially determines its mass (the nucleus’s mass is approximately equal to the mass number times the proton’s mass): M nucleus ≈ A x mproton. So different forms of an element may have different mass. ...
... The number of protons does determine which element it is, but the mass number essentially determines its mass (the nucleus’s mass is approximately equal to the mass number times the proton’s mass): M nucleus ≈ A x mproton. So different forms of an element may have different mass. ...
Chemistry Notes
... Gain valence electrons when they bond Properties of metalloids Elements that touch stair-step line between metals and nonmetals Have characteristics of metals and nonmetals Family/Group – columns of elements Same physical and chemical properties Same number of valence electrons Period – ro ...
... Gain valence electrons when they bond Properties of metalloids Elements that touch stair-step line between metals and nonmetals Have characteristics of metals and nonmetals Family/Group – columns of elements Same physical and chemical properties Same number of valence electrons Period – ro ...
Periodic Table Vocab page 7
... Atomic number: the number of protons in the nucleus of an atom. This number identifies the atom. Mass number: the number of protons and neutrons in the nucleus of an atom. Mass number = protons + neutrons Valence #: The number of outermost electrons in an atom. Periodic Table: An arrangement of elem ...
... Atomic number: the number of protons in the nucleus of an atom. This number identifies the atom. Mass number: the number of protons and neutrons in the nucleus of an atom. Mass number = protons + neutrons Valence #: The number of outermost electrons in an atom. Periodic Table: An arrangement of elem ...
Chapter 14 Inside the Atom Notes
... nucleus of an atom. 1. Isotopes of an atom have the same number of protons but different numbers of neutrons (pg. 415). 2. Mass number is the number of neutrons plus the number of protons. 3. Average atomic mass – the average mass of the mixture of an element’s isotopes ...
... nucleus of an atom. 1. Isotopes of an atom have the same number of protons but different numbers of neutrons (pg. 415). 2. Mass number is the number of neutrons plus the number of protons. 3. Average atomic mass – the average mass of the mixture of an element’s isotopes ...
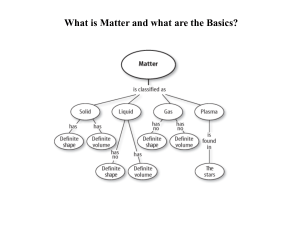
Matter and the Periodic Table
... system of rows and columns on the basis of increasing mass and similar chemical and physical properties. Since the organization exhibited a periodic repetition of similar properties, it became known as the Periodic Table of the Elements. It has become one of modern chemistry's ...
... system of rows and columns on the basis of increasing mass and similar chemical and physical properties. Since the organization exhibited a periodic repetition of similar properties, it became known as the Periodic Table of the Elements. It has become one of modern chemistry's ...
Chapter 2
... 5. Distinguish between each of the following pairs of terms: a. neutron and proton b. atomic number and mass number c. atomic weight and mass number 6. Explain how the atomic number and mass number of an atom can be used to determine the number of neutrons. 7. Explain how two isotopes of an element ...
... 5. Distinguish between each of the following pairs of terms: a. neutron and proton b. atomic number and mass number c. atomic weight and mass number 6. Explain how the atomic number and mass number of an atom can be used to determine the number of neutrons. 7. Explain how two isotopes of an element ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.