File
... F. Decomposition reaction G. Physical change H. Radiation decay J. Synthesis reaction C.12.B-R 13. In which group are the particles arranged in order of decreasing mass? A. alpha, beta, neutron B. alpha, neutron, beta C. neutron, beta, alpha D. neutron, alpha, beta C.12.A-S 14. As you move from left ...
... F. Decomposition reaction G. Physical change H. Radiation decay J. Synthesis reaction C.12.B-R 13. In which group are the particles arranged in order of decreasing mass? A. alpha, beta, neutron B. alpha, neutron, beta C. neutron, beta, alpha D. neutron, alpha, beta C.12.A-S 14. As you move from left ...
Lesson 13 - Highline Public Schools
... weighted average of the masses of the isotopes in a sample of the element. The most common isotope of an element, frequently has a mass that is close to the average atomic mass given in the periodic table. ...
... weighted average of the masses of the isotopes in a sample of the element. The most common isotope of an element, frequently has a mass that is close to the average atomic mass given in the periodic table. ...
RAD 354 Chapt 3 Structure of Matter
... • The atom is the smallest part of an element that has all the properties of the element – 112 elements have been identified 92 are natural 20 were artificially produced ...
... • The atom is the smallest part of an element that has all the properties of the element – 112 elements have been identified 92 are natural 20 were artificially produced ...
Minerals * Chemistry Review
... • The number of protons plus neutrons gives the atom its atomic mass • All atoms of a given element have the same number of protons ...
... • The number of protons plus neutrons gives the atom its atomic mass • All atoms of a given element have the same number of protons ...
Chapter 1 Learning Objective Summary
... changes to the composition of the nucleus. This means that alchemy is possible (though not economical!), because transmutation of one element into another can be accomplished via radioactive decay or bombardment with another particle. Many isotopes are unstable, and undergo spontaneous radioactive d ...
... changes to the composition of the nucleus. This means that alchemy is possible (though not economical!), because transmutation of one element into another can be accomplished via radioactive decay or bombardment with another particle. Many isotopes are unstable, and undergo spontaneous radioactive d ...
PP - myndrs.com
... the nucleus of an atom in orbits or shells. • Each orbit is a certain distance from the nucleus and contains a definite number of electrons. • The orbits are filled in a routine way: – First orbit: 2 electrons – Second orbit: 8 electrons – Third orbit: 8 electrons ...
... the nucleus of an atom in orbits or shells. • Each orbit is a certain distance from the nucleus and contains a definite number of electrons. • The orbits are filled in a routine way: – First orbit: 2 electrons – Second orbit: 8 electrons – Third orbit: 8 electrons ...
Structure of an Atom structure_of_atom
... the nucleus of an atom in orbits or shells. • Each orbit is a certain distance from the nucleus and contains a definite number of electrons. • The orbits are filled in a routine way: – First orbit: 2 electrons – Second orbit: 8 electrons – Third orbit: 8 electrons ...
... the nucleus of an atom in orbits or shells. • Each orbit is a certain distance from the nucleus and contains a definite number of electrons. • The orbits are filled in a routine way: – First orbit: 2 electrons – Second orbit: 8 electrons – Third orbit: 8 electrons ...
BC1 Atoms Unit Standards
... of the protons, neutrons, and electrons in an atom of an element. 2a For a given element, determine the number of protons 2b When given a number of protons, identify the element name and symbol 2c Identify the number of neutrons in an atom from atomic number and mass number 2d Identify the number of ...
... of the protons, neutrons, and electrons in an atom of an element. 2a For a given element, determine the number of protons 2b When given a number of protons, identify the element name and symbol 2c Identify the number of neutrons in an atom from atomic number and mass number 2d Identify the number of ...
Chemistry lecture notes
... Isotopes have the same atomic number (same number of protons), but a different atomic mass number (a different number of neutrons). Isotopes behave the same chemically, because they are the same element. The only difference is that one is heavier than the other, because of the additional ...
... Isotopes have the same atomic number (same number of protons), but a different atomic mass number (a different number of neutrons). Isotopes behave the same chemically, because they are the same element. The only difference is that one is heavier than the other, because of the additional ...
and the atomic
... around the nucleus • based on information about how the energy of an atom changes when it absorbs and ...
... around the nucleus • based on information about how the energy of an atom changes when it absorbs and ...
Beryllium isotopes in geochronology Cosmogenic Be and Be
... rocks at three sites on the Level 2 terrace in upper Holtwood Gorge, Pennsylvania, approximately 50 km upstream of Chesapeake Bay [38]. ...
... rocks at three sites on the Level 2 terrace in upper Holtwood Gorge, Pennsylvania, approximately 50 km upstream of Chesapeake Bay [38]. ...
Name Test Review Chapters 4 and 25 Honors Chemistry 1. Fill in
... 22. If the half-life for the radioactive decay of zirconium-84 is 26 minutes and I start with a 175 gram sample, how much will be left over after 104 minutes? 23. Mercury -197 is used for kidney scans and has a half-life of 3 days. If the amount of mercury-197 needed for a study is 1.0 gram and the ...
... 22. If the half-life for the radioactive decay of zirconium-84 is 26 minutes and I start with a 175 gram sample, how much will be left over after 104 minutes? 23. Mercury -197 is used for kidney scans and has a half-life of 3 days. If the amount of mercury-197 needed for a study is 1.0 gram and the ...
Name Test Review Chemistry Unit 2: The Atom 1. Fill in the blank
... 23. Mercury -197 is used for kidney scans and has a half-life of 3 days. If the amount of mercury-197 needed for a study is 1.0 gram and the time allowed for shipment is 15 days, how much mercury-197 will need to be ordered? 24. How much strontium-90 will remain after 4 half-lives if the initial am ...
... 23. Mercury -197 is used for kidney scans and has a half-life of 3 days. If the amount of mercury-197 needed for a study is 1.0 gram and the time allowed for shipment is 15 days, how much mercury-197 will need to be ordered? 24. How much strontium-90 will remain after 4 half-lives if the initial am ...
Classifying Atoms
... appears on pages 698–699 of the Appendix. Of the more than 100 known elements listed there, 92 occur naturally on Earth in significant amounts. The rest are synthetic elements produced by scientists. In each row of the periodic table, elements are listed from left to right in order of increasing num ...
... appears on pages 698–699 of the Appendix. Of the more than 100 known elements listed there, 92 occur naturally on Earth in significant amounts. The rest are synthetic elements produced by scientists. In each row of the periodic table, elements are listed from left to right in order of increasing num ...
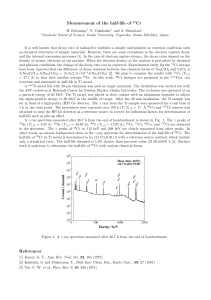
Measurement of the half-life of
... It is well known that decay rate of radioactive nuclides is usually independent on external conditions such as chemical structures of sample materials. However, there are some exceptions in the electron capture decay and the internal conversion processes [1]. In the case of electron capture decays, ...
... It is well known that decay rate of radioactive nuclides is usually independent on external conditions such as chemical structures of sample materials. However, there are some exceptions in the electron capture decay and the internal conversion processes [1]. In the case of electron capture decays, ...
1st Term Review
... 14. Based on the gold foil experiment, what did Rutherford conclude about the atom? 15. An atom of chromium-60 contains how many protons, neutron and electrons? 16. What is the difference between a compound and an element? 17. What is the electron configuration of a neutral calcium atom? 18. Atomic ...
... 14. Based on the gold foil experiment, what did Rutherford conclude about the atom? 15. An atom of chromium-60 contains how many protons, neutron and electrons? 16. What is the difference between a compound and an element? 17. What is the electron configuration of a neutral calcium atom? 18. Atomic ...
Lecture
... the binding energy is(not) great enough to hold the nucleus together. Unstable atoms will lose neutrons or protons as they attempt to become stable. They are called radioactive atoms. What is a radioactive decay? o spontaneous breakdown of an atomic nucleus resulting in the release of nuclear radiat ...
... the binding energy is(not) great enough to hold the nucleus together. Unstable atoms will lose neutrons or protons as they attempt to become stable. They are called radioactive atoms. What is a radioactive decay? o spontaneous breakdown of an atomic nucleus resulting in the release of nuclear radiat ...
Atomic Structure AKS Correlation Use the modern atomic theory to
... Atoms mostly about matter Matter cannot be empty space, dense and its’ makecreated or center up of atoms destroyed Atoms of same element look the same Chem reactions occur when atoms are rearranged Compounds formed by 2 or more atoms Describe the basic structure of the atom as protons, neutrons and ...
... Atoms mostly about matter Matter cannot be empty space, dense and its’ makecreated or center up of atoms destroyed Atoms of same element look the same Chem reactions occur when atoms are rearranged Compounds formed by 2 or more atoms Describe the basic structure of the atom as protons, neutrons and ...
Introduction to the Periodic Table
... The number of protons and neutrons in the nucleus of an atom. ...
... The number of protons and neutrons in the nucleus of an atom. ...
Isotopes, Ions Worksheet
... Different atoms of the same element have the SAME half-life. b) Do different isotopes have different half-lifes (t ½ )? YES Different isotopes have a different neutron number which results in different half-life 21. List THREE Nuclear Applications 1. _______________________ 2. ______________________ ...
... Different atoms of the same element have the SAME half-life. b) Do different isotopes have different half-lifes (t ½ )? YES Different isotopes have a different neutron number which results in different half-life 21. List THREE Nuclear Applications 1. _______________________ 2. ______________________ ...
Vocabulary for Periodic Table
... 11) Group: a vertical column in the periodic table of the elements that have similar properties; also called a family. 12) Period: a horizontal row in the periodic table of elements that have varying properties. 13) Reactive: indicates how likely an element is to undergo a chemical change. 14) Metal ...
... 11) Group: a vertical column in the periodic table of the elements that have similar properties; also called a family. 12) Period: a horizontal row in the periodic table of elements that have varying properties. 13) Reactive: indicates how likely an element is to undergo a chemical change. 14) Metal ...
History of the Atom and Periodic Table
... made of tiny, indivisible particles called “atoms”. Aristotle disagreed and claimed matter was made of earth, wind, water, and fire. ...
... made of tiny, indivisible particles called “atoms”. Aristotle disagreed and claimed matter was made of earth, wind, water, and fire. ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.