MrsB-Chemistry
... B. A scientist thought that matter could not be divided into smaller pieces because chemical reactions only combine elements. They don’t cause elements to change into other elements. C. Alpha particles were used like bullets, and small, positively charged particles shot out from the center of the at ...
... B. A scientist thought that matter could not be divided into smaller pieces because chemical reactions only combine elements. They don’t cause elements to change into other elements. C. Alpha particles were used like bullets, and small, positively charged particles shot out from the center of the at ...
ch2_objectives
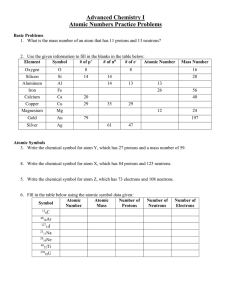
... 5. Distinguish between each of the following pairs of terms: a. neutron and proton b. atomic number and mass number c. atomic weight and mass number 6. Explain how the atomic number and mass number of an atom can be used to determine the number of neutrons. 7. Explain how two isotopes of an element ...
... 5. Distinguish between each of the following pairs of terms: a. neutron and proton b. atomic number and mass number c. atomic weight and mass number 6. Explain how the atomic number and mass number of an atom can be used to determine the number of neutrons. 7. Explain how two isotopes of an element ...
Canyon High School Chemistry
... Chemistry content standards addressed in this unit: 1a. Know how the position of an element in the periodic table relates to its atomic number and atomic mass; 1e. Know the nucleus of the atom is smaller than the atom yet contains most of its mass; 1f. Know transuranium elements are all synthesized ...
... Chemistry content standards addressed in this unit: 1a. Know how the position of an element in the periodic table relates to its atomic number and atomic mass; 1e. Know the nucleus of the atom is smaller than the atom yet contains most of its mass; 1f. Know transuranium elements are all synthesized ...
Test 2 Review Test 2 Review (15-16)_2
... (17) ____________ Which column from above contains VERY non-reactive elements? (18) ____________ How many of these elements are gases at 0 degrees Celsius? (19) ____________ How many of these elements are metalloids? (20) ____________ How many of these elements are NON-metals and solids? (21) ______ ...
... (17) ____________ Which column from above contains VERY non-reactive elements? (18) ____________ How many of these elements are gases at 0 degrees Celsius? (19) ____________ How many of these elements are metalloids? (20) ____________ How many of these elements are NON-metals and solids? (21) ______ ...
Chapter 18 Notes
... Proton- P+ positive charged - in nucleus Number of P+ distinguishes one atom from another Made of 2 up quarks (+2/3 charge) and 1 down ...
... Proton- P+ positive charged - in nucleus Number of P+ distinguishes one atom from another Made of 2 up quarks (+2/3 charge) and 1 down ...
Atomic Structure Power Point
... All matter is composed of atoms. Atoms cannot be created or destroyed. Atoms of elements combine to form compounds. Each element is made up of different kinds of atoms. ...
... All matter is composed of atoms. Atoms cannot be created or destroyed. Atoms of elements combine to form compounds. Each element is made up of different kinds of atoms. ...
Ch. 2 note packet
... Metallurgy - extraction of metals from ore Robert Boyle - (1661 - Skeptical Chymist) - first quantitative experiments; current concept of “element” Georg Stahl – suggested “phlogiston” flowed out of burning material Joseph Priestley - (1733-1804) “discovered” oxygen (not phlogiston) 2.2 - 2.3 Fundam ...
... Metallurgy - extraction of metals from ore Robert Boyle - (1661 - Skeptical Chymist) - first quantitative experiments; current concept of “element” Georg Stahl – suggested “phlogiston” flowed out of burning material Joseph Priestley - (1733-1804) “discovered” oxygen (not phlogiston) 2.2 - 2.3 Fundam ...
atoms
... Element X contains three naturally occurring isotopes with masses of 78.0, 79.0, and 80.0 amu. What data is needed to calculate its average atomic mass? The natural abundance in percent of each isotope (Can you do these calculations?) ...
... Element X contains three naturally occurring isotopes with masses of 78.0, 79.0, and 80.0 amu. What data is needed to calculate its average atomic mass? The natural abundance in percent of each isotope (Can you do these calculations?) ...
Extra Credit Test Review
... 12.One atom has 17 protons, 18 neutrons, and 17 electrons. Another atom has 17 protons, 19 neutrons and 17 electrons. Are these the same element? Yes No Explain: __________________________________________________________________ 13.Today we use Mendeleev’s arrangement, elements are arranged by incre ...
... 12.One atom has 17 protons, 18 neutrons, and 17 electrons. Another atom has 17 protons, 19 neutrons and 17 electrons. Are these the same element? Yes No Explain: __________________________________________________________________ 13.Today we use Mendeleev’s arrangement, elements are arranged by incre ...
atom - Images
... radiation from an element to achieve a more stable state. Uranium was the first radioactive element isolated (by Bequerel), followed by radium and polonium (by Marie Curie and her husband Pierre). There are no stable isotopes for elements after Bismuth (#83). ...
... radiation from an element to achieve a more stable state. Uranium was the first radioactive element isolated (by Bequerel), followed by radium and polonium (by Marie Curie and her husband Pierre). There are no stable isotopes for elements after Bismuth (#83). ...
3-2 Radioactivity and the nucleus
... He concluded that most of the atom is made of ‘empty space’, and that the core of the atom carried a positive charge (he called the core nucleus and the positive charges the protons). ...
... He concluded that most of the atom is made of ‘empty space’, and that the core of the atom carried a positive charge (he called the core nucleus and the positive charges the protons). ...
10.2
... ‘empty space’, and that the core of the atom carried a positive charge (he called the core nucleus and the positive charges the protons). • He also suggested that electrons orbit the nucleus like planets orbit the Sun (Fig.3 p.281). ...
... ‘empty space’, and that the core of the atom carried a positive charge (he called the core nucleus and the positive charges the protons). • He also suggested that electrons orbit the nucleus like planets orbit the Sun (Fig.3 p.281). ...
Chemistry: The Nature of Matter
... Periodic Table of Elements Over 100 elements known, but only about 2 dozen commonly found in living systems ...
... Periodic Table of Elements Over 100 elements known, but only about 2 dozen commonly found in living systems ...
Atom - Sites
... atoms join together chemically. •Combinations of two or more different elements are called compounds. •All compounds are molecules but not all molecules are compounds. (ex. H2O vs. O2) •Molecules can also join together to form larger molecules. •Many, many repeating small molecules joined together f ...
... atoms join together chemically. •Combinations of two or more different elements are called compounds. •All compounds are molecules but not all molecules are compounds. (ex. H2O vs. O2) •Molecules can also join together to form larger molecules. •Many, many repeating small molecules joined together f ...
ChLM Final Review Name: Period: Base Knowledge 1. Classify the
... 21. Why don’t the noble gases react with anything? ...
... 21. Why don’t the noble gases react with anything? ...
Chapter 4: The Structure of the Atom &
... o Gamma radiation is made of GAMMA RAYS (high energy radiation) No mass or charge Symbol is _______________ ...
... o Gamma radiation is made of GAMMA RAYS (high energy radiation) No mass or charge Symbol is _______________ ...
22-Introduction to Radioactivity
... conducting an experiment that models what happens during radioactive decay. Reviewing Some Ideas about Atoms Use your knowledge and information from the textbook to print the best word(s) in each sentence. 1. Atoms are the “basic” particles of chemical ______________________________________. 2. Ther ...
... conducting an experiment that models what happens during radioactive decay. Reviewing Some Ideas about Atoms Use your knowledge and information from the textbook to print the best word(s) in each sentence. 1. Atoms are the “basic” particles of chemical ______________________________________. 2. Ther ...
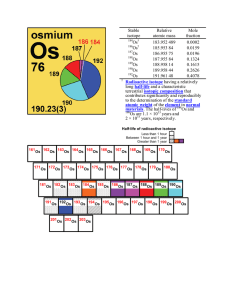
Stable isotope Relative atomic mass Mole fraction Os 183.952 489
... either positive or negative charge (an electron or positron). [return] electron – elementary particle of matter with a negative electric charge and a rest mass of about 9.109 × 10–31 kg. element (chemical element) – a species of atoms; all atoms with the same number of protons in the atomic nucleus. ...
... either positive or negative charge (an electron or positron). [return] electron – elementary particle of matter with a negative electric charge and a rest mass of about 9.109 × 10–31 kg. element (chemical element) – a species of atoms; all atoms with the same number of protons in the atomic nucleus. ...
Understanding the Atom GN
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
SNC1D0 Atomic History
... electrons orbiting around the nucleus. Later experiments showed that the positively charged particles, now called protons, have an equal but opposite charge to the electrons, and have a mass 1836 x greater! The neutron and the existence of isotopes were also discovered ...
... electrons orbiting around the nucleus. Later experiments showed that the positively charged particles, now called protons, have an equal but opposite charge to the electrons, and have a mass 1836 x greater! The neutron and the existence of isotopes were also discovered ...
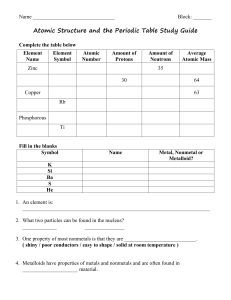
Atomic Numbers Practice Problems
... 3. Write the chemical symbol for atom Y, which has 27 protons and a mass number of 59. ...
... 3. Write the chemical symbol for atom Y, which has 27 protons and a mass number of 59. ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.