Catalytic Nucleophilic Fluorination of Secondary and Tertiary
... Hydrogen bonding to fluoride anions by protic solvents limits the formation of alkene by-products owing to reduced basicity[9] but significantly reduces fluoride nucleophilicity. This dichotomy represents a unique challenge for catalysis, and the catalytic nucleophilic fluorination of comparatively ...
... Hydrogen bonding to fluoride anions by protic solvents limits the formation of alkene by-products owing to reduced basicity[9] but significantly reduces fluoride nucleophilicity. This dichotomy represents a unique challenge for catalysis, and the catalytic nucleophilic fluorination of comparatively ...
Grignard-syn-12-ques
... (OH, NH2, NHR, SH, C=CH, CO2H) Cannot use H2O, CH3OH, CH3CH2OH, etc. as solvents Cannot prepare from substances such as HOCH2CH2Br, etc. ...
... (OH, NH2, NHR, SH, C=CH, CO2H) Cannot use H2O, CH3OH, CH3CH2OH, etc. as solvents Cannot prepare from substances such as HOCH2CH2Br, etc. ...
9: Formation of Alkenes and Alkynes. Elimination Reactions
... This E1 mechanism is analogous to the two-step SN1 substitution mechanism (Chapter 7). The "1" in E1 indicates that the rate determining step of the reaction is unimolecular. This rate determining step is the ionization step (the first step) that involves only the haloalkane substrate (RX). The E1 r ...
... This E1 mechanism is analogous to the two-step SN1 substitution mechanism (Chapter 7). The "1" in E1 indicates that the rate determining step of the reaction is unimolecular. This rate determining step is the ionization step (the first step) that involves only the haloalkane substrate (RX). The E1 r ...
Ch 7 - Practice problem (Answers)
... C) E1 for tertiary halides, E2 for primary and secondary halides. D) E2 for tertiary halides, E1 for primary and secondary halides. Ans: B 25. Which of the following sets of conditions most favors the E1 mechanism? A) when the alkyl halide is tertiary and the base is a weak base B) when the alkyl ha ...
... C) E1 for tertiary halides, E2 for primary and secondary halides. D) E2 for tertiary halides, E1 for primary and secondary halides. Ans: B 25. Which of the following sets of conditions most favors the E1 mechanism? A) when the alkyl halide is tertiary and the base is a weak base B) when the alkyl ha ...
Development of New Synthetic Routes to Organoboronates by Catalytic Allylic Substitution and
... This thesis describes the development of new catalytic methods for the synthesis and application of organometallic reagents, mainly focusing on allylboronic acid derivatives. Thus, palladium pincer-complex catalysis has been applied for extending the scope of palladiumcatalyzed borylation reactions ...
... This thesis describes the development of new catalytic methods for the synthesis and application of organometallic reagents, mainly focusing on allylboronic acid derivatives. Thus, palladium pincer-complex catalysis has been applied for extending the scope of palladiumcatalyzed borylation reactions ...
Synthesis of Natural Products and Related Compounds using Enyne
... our group (Scheme 6).[22] Reactions of five- to sevenmembered cycloalkenes 14 having the substituent at the 3-position of the cycloalkene with 1c under ethylene gas afforded the cyclic compounds 15 in good yields. This reaction could proceed via the highly strained ruthenacyclobutane 16. In each cas ...
... our group (Scheme 6).[22] Reactions of five- to sevenmembered cycloalkenes 14 having the substituent at the 3-position of the cycloalkene with 1c under ethylene gas afforded the cyclic compounds 15 in good yields. This reaction could proceed via the highly strained ruthenacyclobutane 16. In each cas ...
Cerium(IV) Ammonium Nitrate as a Catalyst in
... Some representative examples of these oxidations are listed in Table 2, together with the data for the equivalent reactions carried out in the absence of CAN. The mechanism of this oxidation can be rationalized as shown in Scheme 3, where the starting alcohol is oxidized to the corresponding carbony ...
... Some representative examples of these oxidations are listed in Table 2, together with the data for the equivalent reactions carried out in the absence of CAN. The mechanism of this oxidation can be rationalized as shown in Scheme 3, where the starting alcohol is oxidized to the corresponding carbony ...
Zinc Alkyls in Organic Synthesis
... of a third substituent.[5] Tri-substituted cyclopropanes were therefore synthesized. The authors suggested that the nature of substituents on the starting alkene determines the relative position of the third substituent. The workers also noted that the reaction proceeds by electrophilic addition, pr ...
... of a third substituent.[5] Tri-substituted cyclopropanes were therefore synthesized. The authors suggested that the nature of substituents on the starting alkene determines the relative position of the third substituent. The workers also noted that the reaction proceeds by electrophilic addition, pr ...
Organic Chemistry Fifth Edition
... Do not memorize this chart. But be sure you can figure it out from the general reaction: attack of nucleophile in basic media on less hindered carbon ...
... Do not memorize this chart. But be sure you can figure it out from the general reaction: attack of nucleophile in basic media on less hindered carbon ...
Selective Incorporation of Difluoromethylene
... physical, and biological properties. Accordingly, the incorporation of fluorine atom(s) or fluorine-containing functionalities into organic molecules has received considerable attention. For decades, extensive reports and reviews mainly focused on the fluorination and perfluoroalkylation (especially ...
... physical, and biological properties. Accordingly, the incorporation of fluorine atom(s) or fluorine-containing functionalities into organic molecules has received considerable attention. For decades, extensive reports and reviews mainly focused on the fluorination and perfluoroalkylation (especially ...
Orbitals
... Nucleophilic Addition Reactions of Aldehydes and Ketones Two products can arise from the tetrahedral alkoxide ion intermediate • Protonation by water or acid gives an alcohol • The carbonyl oxygen atom can be protonated and then eliminated as HO- or H2O to give a product with a C=Nu double bond ...
... Nucleophilic Addition Reactions of Aldehydes and Ketones Two products can arise from the tetrahedral alkoxide ion intermediate • Protonation by water or acid gives an alcohol • The carbonyl oxygen atom can be protonated and then eliminated as HO- or H2O to give a product with a C=Nu double bond ...
Exam #3
... A) Using mechanistic reasoning, EXPLAIN WHY menthyl chloride gives only 2-menthene as its E2 elimination product. B) Explain why 3-menthene (rather than 2- menthene) is the major product in the E2 elimination of neomenthyl chloride. H H3C ...
... A) Using mechanistic reasoning, EXPLAIN WHY menthyl chloride gives only 2-menthene as its E2 elimination product. B) Explain why 3-menthene (rather than 2- menthene) is the major product in the E2 elimination of neomenthyl chloride. H H3C ...
Reactions You Should Know When You Begin Organic II
... Addition of symmetrical agents may be anti or syn depending on mechanism or catalyst. Addition of asymmetrical agents follows Markovnikov's Rule except for addition of HBr in the presence of peroxides which adds anti-Markovnikov (only works with HBr). ...
... Addition of symmetrical agents may be anti or syn depending on mechanism or catalyst. Addition of asymmetrical agents follows Markovnikov's Rule except for addition of HBr in the presence of peroxides which adds anti-Markovnikov (only works with HBr). ...
Chem 240 - Napa Valley College
... good he added twice as much benzaldehyde as Grignard reagent and got a lot of white crystalline product. When he analyzed his product he found that he had not made diphenyl methanol, but diphenyl methanal (also called benzophenone) instead. When he asked his research director about it he was told th ...
... good he added twice as much benzaldehyde as Grignard reagent and got a lot of white crystalline product. When he analyzed his product he found that he had not made diphenyl methanol, but diphenyl methanal (also called benzophenone) instead. When he asked his research director about it he was told th ...
Organometallics II
... formula C11H22O, are formed in the reaction of methyl lithium with 3-(R)-tertbutylcyclohexanone. These two alcohols are ...
... formula C11H22O, are formed in the reaction of methyl lithium with 3-(R)-tertbutylcyclohexanone. These two alcohols are ...
Biomimetic Organic Synthesis. 2 Volume Set Brochure
... Biomimetic Organic Synthesis. 2 Volume Set Description: ...
... Biomimetic Organic Synthesis. 2 Volume Set Description: ...
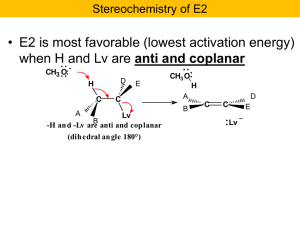
13-Elimination Reactions
... This section describes the stereochemistry of the E1 and E2 reaction mechanisms and compares them with the stereochemistry of the SN1 and SN2 reaction mechanisms, which are covered in Chapter 12. The E1 reaction mechanism is a two-step process that, as with the SN1 mechanism, usually loses all the s ...
... This section describes the stereochemistry of the E1 and E2 reaction mechanisms and compares them with the stereochemistry of the SN1 and SN2 reaction mechanisms, which are covered in Chapter 12. The E1 reaction mechanism is a two-step process that, as with the SN1 mechanism, usually loses all the s ...
Metal-catalysed approaches to amide bond formation
... bonds. Nucleophilic attack of the amine onto the ester then proceeds via a six-membered (path A) or four-membered (path B) transition state. ...
... bonds. Nucleophilic attack of the amine onto the ester then proceeds via a six-membered (path A) or four-membered (path B) transition state. ...
chm238f02.exam2
... (b) Given pKas: CH3OH = 45; CH3OH = 16; H2 = 38; H2O = 16; PhOH = 10: Predict the direction and Keq of the reaction above. ...
... (b) Given pKas: CH3OH = 45; CH3OH = 16; H2 = 38; H2O = 16; PhOH = 10: Predict the direction and Keq of the reaction above. ...
9: Formation of Alkenes and Alkynes. Elimination Reactions
... correlation between alkene substitution and alkene stability. Since chemists now know the direct correlation between alkene stability and substitution, Zaitzev's rule is equivalent to saying that an elimination reaction preferentially gives the most stable alkene product(s). While it would be better ...
... correlation between alkene substitution and alkene stability. Since chemists now know the direct correlation between alkene stability and substitution, Zaitzev's rule is equivalent to saying that an elimination reaction preferentially gives the most stable alkene product(s). While it would be better ...
CHM238-01 EXAM 2 October 14, 2002 103
... that occurs under acidic conditions. Consider the situation shown below. (a) Provide arrows and necessary lone pairs to complete the reaction flow for the unexpected alcohol substitution reaction: H H3C ...
... that occurs under acidic conditions. Consider the situation shown below. (a) Provide arrows and necessary lone pairs to complete the reaction flow for the unexpected alcohol substitution reaction: H H3C ...
Diels–Alder reaction
.png?width=300)
The Diels–Alder reaction is an organic chemical reaction (specifically, a [4+2] cycloaddition) between a conjugated diene and a substituted alkene, commonly termed the dienophile, to form a substituted cyclohexene system. It was first described by Otto Paul Hermann Diels and Kurt Alder in 1928, for which work they were awarded the Nobel Prize in Chemistry in 1950. The Diels–Alder reaction is particularly useful in synthetic organic chemistry as a reliable method for forming 6-membered systems with good control over regio- and stereochemical properties. The underlying concept has also been applied to other π-systems, such as carbonyls and imines, to furnish the corresponding heterocycles, known as the hetero-Diels–Alder reaction. Diels–Alder reactions can be reversible under certain conditions; the reverse reaction is known as the retro-Diels–Alder reaction.