Diastereoselective Allylation of Carbonyl Compounds and Imines:
... crotylboropinacolate, respectively. The reactions should be performed in the presence of a Lewis acid. Thus, the SnCl4 catalyzed addition of allylboropinacolate to the chiral aldehyde gave minimal selectivity (46:54) favoring the syn-diastereomer. However, better diastereoselectivity was obtained wh ...
... crotylboropinacolate, respectively. The reactions should be performed in the presence of a Lewis acid. Thus, the SnCl4 catalyzed addition of allylboropinacolate to the chiral aldehyde gave minimal selectivity (46:54) favoring the syn-diastereomer. However, better diastereoselectivity was obtained wh ...
R - Evans - Harvard University
... reaction^.^^*^.^ In conjunction with our interests in amino acid derived natural products, we anticipated that these enolates might be attractive precursors to enantiomerically pure a-amino acids if effective electrophilic aminating agents could be developed. The simple strategy that was envisioned ...
... reaction^.^^*^.^ In conjunction with our interests in amino acid derived natural products, we anticipated that these enolates might be attractive precursors to enantiomerically pure a-amino acids if effective electrophilic aminating agents could be developed. The simple strategy that was envisioned ...
OChem1 Course Pack
... The secondary carbocation that is formed undergoes rearrangement to produce a better tertiary cation that no longer has the strained cyclobutyl ring involved. The tertiary carbocation is then deprotonated to give the most highly substituted alkene which is stabilized by four electron-donating alkyl ...
... The secondary carbocation that is formed undergoes rearrangement to produce a better tertiary cation that no longer has the strained cyclobutyl ring involved. The tertiary carbocation is then deprotonated to give the most highly substituted alkene which is stabilized by four electron-donating alkyl ...
Transformation of Carbon Dioxide
... formation of a methyl carbonato complex through a reaction of tin methoxide with CO2 was reported in 1967.48,49 The formation of DMC upon thermolysis of methyl (carbonato) tin was first reported in 1975, but the yield was very low; only 10% based on tin.50 Another group later reported similar result ...
... formation of a methyl carbonato complex through a reaction of tin methoxide with CO2 was reported in 1967.48,49 The formation of DMC upon thermolysis of methyl (carbonato) tin was first reported in 1975, but the yield was very low; only 10% based on tin.50 Another group later reported similar result ...
Ch. 6 - Department of Chemistry and Biochemistry
... Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the Department of Chemistry at the University of Gu ...
... Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the Department of Chemistry at the University of Gu ...
Palladium(II)-Catalyzed Oxidative Cyclization Strategies Andreas K. Å. Persson
... The use of transition metal catalysts has proven to be one of the most diverse tools for the mild and selective formation of carbon-carbon bonds. In particular palladium-catalyzed cross-coupling reactions have revolutionized the field. The main focus of this thesis has been directed towards preparat ...
... The use of transition metal catalysts has proven to be one of the most diverse tools for the mild and selective formation of carbon-carbon bonds. In particular palladium-catalyzed cross-coupling reactions have revolutionized the field. The main focus of this thesis has been directed towards preparat ...
Chem E2b - Organic Chemistry II What is Organic Chemistry?
... • Halogens are deactivating (inductive effect) and ortho/para directing (resonance) • Nucleophiles can add to arenediazonium salts, strongly activated aryl halides (SNAr), or benzyne intermediates • 5-Membered heteroaromatics participate in EAS at C2position. • 6-Membered heteroaromatics participate ...
... • Halogens are deactivating (inductive effect) and ortho/para directing (resonance) • Nucleophiles can add to arenediazonium salts, strongly activated aryl halides (SNAr), or benzyne intermediates • 5-Membered heteroaromatics participate in EAS at C2position. • 6-Membered heteroaromatics participate ...
Organic Chemistry II Introduction
... Alcohols and Phenols • Important solvents and intermediates • Phenols contain an OH group connected to a carbon in a benzene ...
... Alcohols and Phenols • Important solvents and intermediates • Phenols contain an OH group connected to a carbon in a benzene ...
Efficient and Convenient Procedure for Protection of Hydroxyl
... these methods may suffer from one or more disadvantages such as the use of expensive or less easily available regents, vigorous reaction conditions, unsatisfactory yields, prolonged standing, and tedious manipulations in isolation of the pure products or the use of toxic volatile solvents. Therefore ...
... these methods may suffer from one or more disadvantages such as the use of expensive or less easily available regents, vigorous reaction conditions, unsatisfactory yields, prolonged standing, and tedious manipulations in isolation of the pure products or the use of toxic volatile solvents. Therefore ...
Handout VI
... The attack of hydride ion on the carbonyl group, i.e., the reduction of aldehydes and ketones may be seen as nucleophilic addition but the actual pathway is quite complicated. Lithium aluminium hydride is a very powerful reducing agent and easily reduces both aldehydes and ketones to their correspon ...
... The attack of hydride ion on the carbonyl group, i.e., the reduction of aldehydes and ketones may be seen as nucleophilic addition but the actual pathway is quite complicated. Lithium aluminium hydride is a very powerful reducing agent and easily reduces both aldehydes and ketones to their correspon ...
Scope and Limitations - Organic Reactions Wiki
... face of the alkene (most easily envisaged in cyclic systems) has proven to be very reliable with few exceptions reported. In fact, the high level of anti selectivity that is observed in such dihydroxylations has led to a problem: how to overturn this bias and obtain dihydroxylation on the same face ...
... face of the alkene (most easily envisaged in cyclic systems) has proven to be very reliable with few exceptions reported. In fact, the high level of anti selectivity that is observed in such dihydroxylations has led to a problem: how to overturn this bias and obtain dihydroxylation on the same face ...
NAME Chem 204 Spring, 1990 Final Exam FINAL EXAM: Chaps. 1
... alkyl chloride, there is NO one reaction that will convert a chiral alcohol into its alkyl chloride with "retained" configuration. Thus, it is necessary to use several reaction steps, each of 100% inversion or 100% retention of configuration, in order to convert (X) into (XI). Outlines below a serie ...
... alkyl chloride, there is NO one reaction that will convert a chiral alcohol into its alkyl chloride with "retained" configuration. Thus, it is necessary to use several reaction steps, each of 100% inversion or 100% retention of configuration, in order to convert (X) into (XI). Outlines below a serie ...
Microsoft Word
... using DIBAL-H in THF exploiting the sulfinyl group as an intramolecular chiral auxiliary yielded the -hydroxy sulfoxide 9 (dr >95:<5), which on treatment with freshly recrystallized Nbromosuccinimide (NBS) afforded bromohydrin 19 as a single isomer regio- and stereoselectively. To transform bromohy ...
... using DIBAL-H in THF exploiting the sulfinyl group as an intramolecular chiral auxiliary yielded the -hydroxy sulfoxide 9 (dr >95:<5), which on treatment with freshly recrystallized Nbromosuccinimide (NBS) afforded bromohydrin 19 as a single isomer regio- and stereoselectively. To transform bromohy ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... oxidation of amines via radical cations. In 2014, Garcia and coworkers reported a seminal study on using graphenes as carbocatalysts for hydrogenation of alkenes (Figure 2C).28 The authors demonstrated the activation of H2 on the graphene surface, which possibly contains frustrated Lewis acid−base s ...
... oxidation of amines via radical cations. In 2014, Garcia and coworkers reported a seminal study on using graphenes as carbocatalysts for hydrogenation of alkenes (Figure 2C).28 The authors demonstrated the activation of H2 on the graphene surface, which possibly contains frustrated Lewis acid−base s ...
Title Several Reactions of Isocyanide and Related Compounds
... N-methylani line, required higher reaction temperatures ...
... N-methylani line, required higher reaction temperatures ...
Boron and Metal Catalyzed CC and CH Bond Formation
... of these novel reactions have been developed. These include alternate routes to diphenylmethanes and 1,4-diynes from easily prepared dialkynylboron chlorides. In addition, E and Z alkenyl halides can now be prepared using boron trichloride without the use of butyllithium. The stereochemistry of the ...
... of these novel reactions have been developed. These include alternate routes to diphenylmethanes and 1,4-diynes from easily prepared dialkynylboron chlorides. In addition, E and Z alkenyl halides can now be prepared using boron trichloride without the use of butyllithium. The stereochemistry of the ...
Anionic rearrangement of 2-benzyloxypyridine derivatives and a synthetic approach to aldingenin B
... [1,2]-Anionic rearrangements are important tools for altering the complexity of molecules at hand. In Part I of this dissertation, an anionic rearrangement of 2-benzyloxypyridine is described. Pyridine-directed metallation of the benzylic carbon leads to 1,2-migration of pyridine via a postulated as ...
... [1,2]-Anionic rearrangements are important tools for altering the complexity of molecules at hand. In Part I of this dissertation, an anionic rearrangement of 2-benzyloxypyridine is described. Pyridine-directed metallation of the benzylic carbon leads to 1,2-migration of pyridine via a postulated as ...
12_chemistry_impq_CH10_haloalkanes_and_haloarenes_02
... Q4. Of benzene and phenol, which is more easily nitrated and why? Ans. Nitration is an electrophilic substitution. The –OH group in phenol increases the electron density at ortho and para position as follows Since phenol has higher electron density due to electron releasing nature of -OH group , com ...
... Q4. Of benzene and phenol, which is more easily nitrated and why? Ans. Nitration is an electrophilic substitution. The –OH group in phenol increases the electron density at ortho and para position as follows Since phenol has higher electron density due to electron releasing nature of -OH group , com ...
Chemical Reactivity and Biological Activity of Diketene
... reaction, which results in an amide bond in the NBP-diketene adduct. The lability of the amide bond as opposed to the amine bonds formed by β-propiolactone and β-butyrolactone could be one of the differential factors responsible for the lack of carcinogenicity of diketene. (v) Ab initio calculations ...
... reaction, which results in an amide bond in the NBP-diketene adduct. The lability of the amide bond as opposed to the amine bonds formed by β-propiolactone and β-butyrolactone could be one of the differential factors responsible for the lack of carcinogenicity of diketene. (v) Ab initio calculations ...
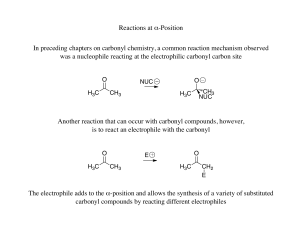
Reactions at α-Position In preceding chapters on carbonyl chemistry
... The enolate formed could still react with either carbonyl to generate two different products, but since an aldehyde is more reactive than a ketone benzaldehyde will react preferentially ...
... The enolate formed could still react with either carbonyl to generate two different products, but since an aldehyde is more reactive than a ketone benzaldehyde will react preferentially ...
Enol esters: Versatile substrates in synthesis of fine and specialty
... 2. Asymmetric hydroformylation of enol esters ................................................................................. 6 2.2. AHF/ Wittig tandem process involving enol esters ................................................................. 7 3. Asymmetric epoxidation of (Z)-enol esters ... ...
... 2. Asymmetric hydroformylation of enol esters ................................................................................. 6 2.2. AHF/ Wittig tandem process involving enol esters ................................................................. 7 3. Asymmetric epoxidation of (Z)-enol esters ... ...
Organic synthesis and methodology related to the malaria drug artemisinin
... Access to artemisinin through isolation, bio-engineering, and chemical synthesis will be described. Our attempts to access the artemisinin family of anti-malarials through the total synthesis of dihydro-epi-deoxyarteannuin B and dihydroartemisinic acid will be discussed fully. Key features of the sy ...
... Access to artemisinin through isolation, bio-engineering, and chemical synthesis will be described. Our attempts to access the artemisinin family of anti-malarials through the total synthesis of dihydro-epi-deoxyarteannuin B and dihydroartemisinic acid will be discussed fully. Key features of the sy ...
Full-Text PDF
... mol % of tetrakis(acetonitrile)copper(I) triflate was able to efficiently catalyze the transformation of phenol 1 to the corresponding phenyl acetate 2 in 3 min and in quantitative yield. the model substrate and acetic anhydride (4 equivalents) as the acetylation agent, we found that 1 the model ...
... mol % of tetrakis(acetonitrile)copper(I) triflate was able to efficiently catalyze the transformation of phenol 1 to the corresponding phenyl acetate 2 in 3 min and in quantitative yield. the model substrate and acetic anhydride (4 equivalents) as the acetylation agent, we found that 1 the model ...
Diels–Alder reaction
.png?width=300)
The Diels–Alder reaction is an organic chemical reaction (specifically, a [4+2] cycloaddition) between a conjugated diene and a substituted alkene, commonly termed the dienophile, to form a substituted cyclohexene system. It was first described by Otto Paul Hermann Diels and Kurt Alder in 1928, for which work they were awarded the Nobel Prize in Chemistry in 1950. The Diels–Alder reaction is particularly useful in synthetic organic chemistry as a reliable method for forming 6-membered systems with good control over regio- and stereochemical properties. The underlying concept has also been applied to other π-systems, such as carbonyls and imines, to furnish the corresponding heterocycles, known as the hetero-Diels–Alder reaction. Diels–Alder reactions can be reversible under certain conditions; the reverse reaction is known as the retro-Diels–Alder reaction.