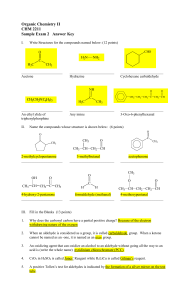
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... Why does the carbonyl carbon have a partial positive charge? Because of the electron withdrawing nature of the oxygen ...
... Why does the carbonyl carbon have a partial positive charge? Because of the electron withdrawing nature of the oxygen ...
ALDEHYDES AND KETONES I. NUCLEOPHILIC ADDITION TO …
... As we have already seen, substituted amines can react with aldehydes and ketones to form a variety of products. ...
... As we have already seen, substituted amines can react with aldehydes and ketones to form a variety of products. ...
carbonyl compounds
... These are vigorous reactions and white fumes are HCl are produced Water, alcohols, ammonia and primary amines are all nucleophiles. They have a lone pair on an O or N The reaction with an alcohol produces an ester. Compared with esterification (using a carboxylic acid), the acylation reaction has th ...
... These are vigorous reactions and white fumes are HCl are produced Water, alcohols, ammonia and primary amines are all nucleophiles. They have a lone pair on an O or N The reaction with an alcohol produces an ester. Compared with esterification (using a carboxylic acid), the acylation reaction has th ...
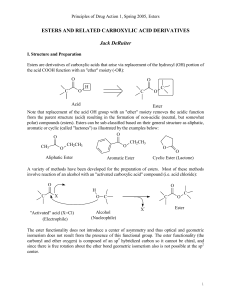
esters and related carboxylic acid derivatives
... There are a number of other functional groups that are derivatives of traditional esters or "esterlike" including amides and amide derivatives (carbamates, ureas, etc.), carbonates, anhydrides and phosphate esters. The amide and amide derivatives are discussed in a separate chapter: O ...
... There are a number of other functional groups that are derivatives of traditional esters or "esterlike" including amides and amide derivatives (carbamates, ureas, etc.), carbonates, anhydrides and phosphate esters. The amide and amide derivatives are discussed in a separate chapter: O ...
View/Open
... electrophilic substitution (Section 16.5). A chlorine substituent, for example, inductively withdraws electrons from an acyl group in the same way that it withdraws electrons from and thus deactivates an aromatic ring. Similarly, amino, methoxyl, and methylthio substituents donate electrons to acyl ...
... electrophilic substitution (Section 16.5). A chlorine substituent, for example, inductively withdraws electrons from an acyl group in the same way that it withdraws electrons from and thus deactivates an aromatic ring. Similarly, amino, methoxyl, and methylthio substituents donate electrons to acyl ...
Organic Chemistry Fifth Edition
... Elimination occurs in the direction that gives the less-substituted double bond. This is called the Hofmann rule. H2C ...
... Elimination occurs in the direction that gives the less-substituted double bond. This is called the Hofmann rule. H2C ...
ALKANES – STRUCTURE, PROPERTIES, AND SYNTHESIS
... describe the most stable conformations in these systems. Also, give or identify the types of strain present in different conformations in these systems, and use this information to explain relative stabilities. Important types of strain in conformational analysis are: o torsional strain (favors stag ...
... describe the most stable conformations in these systems. Also, give or identify the types of strain present in different conformations in these systems, and use this information to explain relative stabilities. Important types of strain in conformational analysis are: o torsional strain (favors stag ...
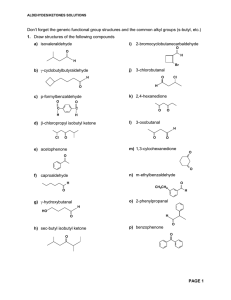
Aldehydes/Ketones Solutions
... 6. Write equations showing how the following transformations can be carried out. No mechanisms required but show all reagents and intermediate products formed. More than one step may be necessary a) O O C H ...
... 6. Write equations showing how the following transformations can be carried out. No mechanisms required but show all reagents and intermediate products formed. More than one step may be necessary a) O O C H ...
Amidations of Rosin with Isocyanates
... for the addition amounts of the catalysts between 10 and 20 mg/g abietic acid. The results demonstrate that the tertiary amines can greatly accelerate the reaction rate of abietic acid with phenyl isocyanate. Amidation of rosin with phenyl isocyanate Table 3 shows the results of amidation of rosin w ...
... for the addition amounts of the catalysts between 10 and 20 mg/g abietic acid. The results demonstrate that the tertiary amines can greatly accelerate the reaction rate of abietic acid with phenyl isocyanate. Amidation of rosin with phenyl isocyanate Table 3 shows the results of amidation of rosin w ...
Aromatic Compounds
... • Addition of a reagent such as HCl to an alkene • The electrophilic hydrogen approaches the p electrons ...
... • Addition of a reagent such as HCl to an alkene • The electrophilic hydrogen approaches the p electrons ...
Preparation and Physical Properties of Chitosan Benzoic Acid
... Chitin, the second abundant natural polymer in Nature, and chitosan, a partially deacetylated form of chitin, have recently received much of attention owing to their applicability in wide range of fields such as pharmaceuticals, cosmetics, agriculture, foods, and material sciences [1–5]. Their film ...
... Chitin, the second abundant natural polymer in Nature, and chitosan, a partially deacetylated form of chitin, have recently received much of attention owing to their applicability in wide range of fields such as pharmaceuticals, cosmetics, agriculture, foods, and material sciences [1–5]. Their film ...
Handout 3
... rearrangement ).(hydride or methyl shift) Oxymercuration reaction is anti addition of H, OH that follows Mark.Rule (However, no rearrangement occurs). Hydroboration Method For the preparation of all types of alcohols 1°, 2°, 3° -Syn addition i.e. both H and OH are added to the same face of double bo ...
... rearrangement ).(hydride or methyl shift) Oxymercuration reaction is anti addition of H, OH that follows Mark.Rule (However, no rearrangement occurs). Hydroboration Method For the preparation of all types of alcohols 1°, 2°, 3° -Syn addition i.e. both H and OH are added to the same face of double bo ...
CH 2 - ResearchGate
... It has been developed by H.E. Simmons and R.D. Smith of the DuPont Company. During this synthesis diiodomethane and zinc-copper couple are stirred together with alkene. The diiodomethane and zinc react together to produce carbene like species called as carbenoid which brings stereospecific addition ...
... It has been developed by H.E. Simmons and R.D. Smith of the DuPont Company. During this synthesis diiodomethane and zinc-copper couple are stirred together with alkene. The diiodomethane and zinc react together to produce carbene like species called as carbenoid which brings stereospecific addition ...
Document
... This increase in the basicity of an amine by alkylation is slightly enhanced by introduction of a second alkyl group. Further alkylation of a 2o amine does further increase basicity, but only if measured in the gas phase. In the usual aqueous media, aminium ions from 1o and 2o amines are very effect ...
... This increase in the basicity of an amine by alkylation is slightly enhanced by introduction of a second alkyl group. Further alkylation of a 2o amine does further increase basicity, but only if measured in the gas phase. In the usual aqueous media, aminium ions from 1o and 2o amines are very effect ...
Forward
... in much the same way that Grignard reagents do, except that they function as hydride donors rather than as carbanion sources. Borohydride transfers a hydrogen with its pair of bonding electrons to the positively polarized carbon of a carbonyl group. The negatively polarized oxygen attacks boron. Ult ...
... in much the same way that Grignard reagents do, except that they function as hydride donors rather than as carbanion sources. Borohydride transfers a hydrogen with its pair of bonding electrons to the positively polarized carbon of a carbonyl group. The negatively polarized oxygen attacks boron. Ult ...
Development of New Synthetic Routes to Organoboronates by Catalytic Allylic Substitution and
... However, these reactive species are known to undergo facile metallotropic rearrangements,109-111 which may lead to scrambling of both regio- and stereoisomers in the case of substituted substrates (Scheme 9). Furthermore, the incompatibility of lithium and Grignard reagents with many functional grou ...
... However, these reactive species are known to undergo facile metallotropic rearrangements,109-111 which may lead to scrambling of both regio- and stereoisomers in the case of substituted substrates (Scheme 9). Furthermore, the incompatibility of lithium and Grignard reagents with many functional grou ...
7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic
... which one group on the molecule (a leaving group) is replaced by another group (a nucleophile). The transformation of haloalkanes (R-X) into alcohols (R-OH) where an OH group replaces the halogen (X) is an example of nucleophilic substitution. Most nucleophilic substitution reactions take place by e ...
... which one group on the molecule (a leaving group) is replaced by another group (a nucleophile). The transformation of haloalkanes (R-X) into alcohols (R-OH) where an OH group replaces the halogen (X) is an example of nucleophilic substitution. Most nucleophilic substitution reactions take place by e ...
File
... 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. Wha ...
... 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. Wha ...
Learning Guide for Chapter 23: Amines
... Once an amine has been protonated, it is an ionic compound. Amine salts are solids, and are usually soluble in water but insoluble in organic solvents. Most biologically active amines are used as their amine salts, which dissolve to make injectable or drinkable water solutions, and are also less pro ...
... Once an amine has been protonated, it is an ionic compound. Amine salts are solids, and are usually soluble in water but insoluble in organic solvents. Most biologically active amines are used as their amine salts, which dissolve to make injectable or drinkable water solutions, and are also less pro ...
Oxidation involving CO System ( O
... cannot be further converted to the acid. The aldehyde is converted back to an alcohol by alcohol (keto) reductases (reversible), however, it goes forward as the aldehyde is converted to carboxylic acid; 3° alcohols and phenolic alcohols cannot be oxidized by this enzyme; No “H” attached to adjacent ...
... cannot be further converted to the acid. The aldehyde is converted back to an alcohol by alcohol (keto) reductases (reversible), however, it goes forward as the aldehyde is converted to carboxylic acid; 3° alcohols and phenolic alcohols cannot be oxidized by this enzyme; No “H” attached to adjacent ...
Cerium(IV) Ammonium Nitrate as a Catalyst in
... more sustainable technologies such as heterogeneous catalysis, the CAN/sodium bromate system has been further modified by immobilizing the cerium catalyst on phosphonate and phosphonic acid-modified silica gel, and indeed the species thus prepared was believed to be the first example of a Ce(IV)-bas ...
... more sustainable technologies such as heterogeneous catalysis, the CAN/sodium bromate system has been further modified by immobilizing the cerium catalyst on phosphonate and phosphonic acid-modified silica gel, and indeed the species thus prepared was believed to be the first example of a Ce(IV)-bas ...
Ch 9 Lecture 2
... 2) No Hydrogen Bonding is possible in R—O—R 3) Boiling Points are much lower than alcohols, more like haloalkanes 4) Water solubility much less than alcohols a) MeOMe and EtOEt have some water solubility b) Larger ethers are insoluble, very much like alkanes 5) Fairly unreactive, nonpolar solvents f ...
... 2) No Hydrogen Bonding is possible in R—O—R 3) Boiling Points are much lower than alcohols, more like haloalkanes 4) Water solubility much less than alcohols a) MeOMe and EtOEt have some water solubility b) Larger ethers are insoluble, very much like alkanes 5) Fairly unreactive, nonpolar solvents f ...
Chapter 20 Amines - FIU Faculty Websites
... The acyl azide is obtained from an acid chloride Rearrangement of the acyl azide occurs with loss of N2, a very stable leaving group In the last step, the isocyanate is hydrolyzed by adding water ...
... The acyl azide is obtained from an acid chloride Rearrangement of the acyl azide occurs with loss of N2, a very stable leaving group In the last step, the isocyanate is hydrolyzed by adding water ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.