Lithium Bromide Original Commentary
... Lithium bromide catalyzes the one-pot condensation of aldehydes, β-keto esters, and ureas to form dihydropyrimidinones (Biginelli reaction, eq 14).34 LiBr is also a suitable Lewis acid for promotion of the one-pot Bischler-Möhlau indole synthesis (eq 15).35 LiBr interacts with and can influence the ...
... Lithium bromide catalyzes the one-pot condensation of aldehydes, β-keto esters, and ureas to form dihydropyrimidinones (Biginelli reaction, eq 14).34 LiBr is also a suitable Lewis acid for promotion of the one-pot Bischler-Möhlau indole synthesis (eq 15).35 LiBr interacts with and can influence the ...
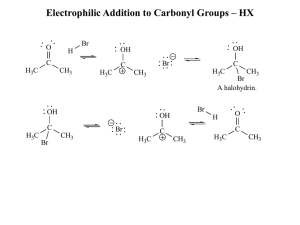
Electrophilic Addition to Carbonyl Groups – HX
... Nucleophilic Addition to Carbonyl Groups: Enolate and Aldol Reactions • This reaction is known as the aldol reaction. It takes an aldehyde and converts it into an alcohol (while extending the carbon chain). • If an aldol reaction is worked up under acidic conditions, an E2 reaction will follow, giv ...
... Nucleophilic Addition to Carbonyl Groups: Enolate and Aldol Reactions • This reaction is known as the aldol reaction. It takes an aldehyde and converts it into an alcohol (while extending the carbon chain). • If an aldol reaction is worked up under acidic conditions, an E2 reaction will follow, giv ...
Chapter 17 Amines
... Secondary amines: both aryl amines and alkyl amines react with nitrous acid to yield N-nitrosoamines(亚硝胺) N O NHCH3 + NaNO2 + 2 HCl ...
... Secondary amines: both aryl amines and alkyl amines react with nitrous acid to yield N-nitrosoamines(亚硝胺) N O NHCH3 + NaNO2 + 2 HCl ...
Organic Chemistry/Fourth Edition: e-Text
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
PowerPoint 演示文稿
... • Nucleophilicity roughly parallels basicity when comparing nucleophiles that have the same attacking atom. RO ...
... • Nucleophilicity roughly parallels basicity when comparing nucleophiles that have the same attacking atom. RO ...
Organic Chemistry II Introduction
... ArOH is more acidic than ROH Soluble in dilute NaOH Anion is resonance stabilized EWG make phenols more acidic than phenol EDG make phenols less acidic than phenol O ...
... ArOH is more acidic than ROH Soluble in dilute NaOH Anion is resonance stabilized EWG make phenols more acidic than phenol EDG make phenols less acidic than phenol O ...
Organic Chemistry
... Only van der Waals force: London force. Boiling point increases with length of chain. Combust to give mainly CO2 and H2O Nomenclature suffix “‐ane” ...
... Only van der Waals force: London force. Boiling point increases with length of chain. Combust to give mainly CO2 and H2O Nomenclature suffix “‐ane” ...
Chemistry 360 - Athabasca University
... Why must the condenser be ‘clean and dry’ prior to use? a) Clean and dry glassware automatically guarantees a higher yield b) Water is a by-product of the reaction and having ‘wet glassware’ will slow the reaction down c) Chemists are just neat ...
... Why must the condenser be ‘clean and dry’ prior to use? a) Clean and dry glassware automatically guarantees a higher yield b) Water is a by-product of the reaction and having ‘wet glassware’ will slow the reaction down c) Chemists are just neat ...
Organic Chemistry Fifth Edition
... Solvation of a chloride ion by ion-dipole attractive forces with water. The negatively charged chloride ion interacts with the positively polarized hydrogens of water. ...
... Solvation of a chloride ion by ion-dipole attractive forces with water. The negatively charged chloride ion interacts with the positively polarized hydrogens of water. ...
Document
... While dehydrations have favorable entropy changes, the position of equilibrium favors reactants. Operation of the Principle of Le Châtelier in Alcohol Dehydrations • removing a product from a reaction mixture as it is formed drives the equilibrium to the right, forming more product. • Thus, the alke ...
... While dehydrations have favorable entropy changes, the position of equilibrium favors reactants. Operation of the Principle of Le Châtelier in Alcohol Dehydrations • removing a product from a reaction mixture as it is formed drives the equilibrium to the right, forming more product. • Thus, the alke ...
Chapter 1 Structure and Bonding
... Hyperconjugation makes substituted alkenes more stable by stabilizing p-orbitals cis alkenes are less stable than trans alkenes because of steric crowding cis cycloalkenes are more stable than trans for the small rings ...
... Hyperconjugation makes substituted alkenes more stable by stabilizing p-orbitals cis alkenes are less stable than trans alkenes because of steric crowding cis cycloalkenes are more stable than trans for the small rings ...
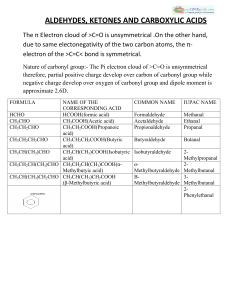
ALDEHYDES, KETONES AND CARBOXYLIC ACIDS
... due to same electonegativity of the two carbon atoms, the π‐ electron of the >C=C< bond is symmetrical. Nature of carbonyl group:- The Pi electron cloud of >C=O is unsymmetrical therefore, partial positive charge develop over carbon of carbonyl group while negative charge develop over oxygen of car ...
... due to same electonegativity of the two carbon atoms, the π‐ electron of the >C=C< bond is symmetrical. Nature of carbonyl group:- The Pi electron cloud of >C=O is unsymmetrical therefore, partial positive charge develop over carbon of carbonyl group while negative charge develop over oxygen of car ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... reaction showed C/O atomic ratio of 1.95, which increased to C/O atomic ratio of 3.46 after the reaction, indicating that some oxygen functionalities have been removed from the GO surface during the reaction (Figure SI-1). (6) Detailed XPS analysis of the GO material before the reaction showed C/O a ...
... reaction showed C/O atomic ratio of 1.95, which increased to C/O atomic ratio of 3.46 after the reaction, indicating that some oxygen functionalities have been removed from the GO surface during the reaction (Figure SI-1). (6) Detailed XPS analysis of the GO material before the reaction showed C/O a ...
Ethers - Home - KSU Faculty Member websites
... Cleavage of ethers by hot concentrated acids Ethers are quite stable compounds. Ethers react only under strongly acidic condition.When ethers are heated in concentrated acid ...
... Cleavage of ethers by hot concentrated acids Ethers are quite stable compounds. Ethers react only under strongly acidic condition.When ethers are heated in concentrated acid ...
Ethers, Epoxides and Sulfides
... ¾ Sulfides are relatively strong nucleophiles. - sulfur is large and more polarizable, its valence electrons are less tightly held. ¾ Sulfides attack unhindered alkyl halides to give ...
... ¾ Sulfides are relatively strong nucleophiles. - sulfur is large and more polarizable, its valence electrons are less tightly held. ¾ Sulfides attack unhindered alkyl halides to give ...
adjacent to - dl1.ponato.com
... polar mechanisms taking place under basic conditions, polar mechanisms taking place under acidic conditions, pericyclic reactions, free-radical reactions, and transition-metal-mediated and -catalyzed reactions, giving typical examples and general mechanistic patterns for each class of reaction along ...
... polar mechanisms taking place under basic conditions, polar mechanisms taking place under acidic conditions, pericyclic reactions, free-radical reactions, and transition-metal-mediated and -catalyzed reactions, giving typical examples and general mechanistic patterns for each class of reaction along ...
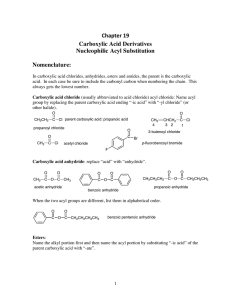
Chapter 19 Carboxylic Acid Derivatives Nucleophilic Acyl
... Since a nucleophile can attack either carbonyl, symmetrical anhydrides are usually used so as to give one product. (1) Preparation of Esters (a) We can use neutral alcohols. With the neutral alcohols we usually use acid catalysis to activate the carbonyl carbon of the anhydride to nucleophilic attac ...
... Since a nucleophile can attack either carbonyl, symmetrical anhydrides are usually used so as to give one product. (1) Preparation of Esters (a) We can use neutral alcohols. With the neutral alcohols we usually use acid catalysis to activate the carbonyl carbon of the anhydride to nucleophilic attac ...
Nucleophilic Acyl Substitution
... and aromatic hydrocarbons. Like alcohols and ethers, carbonyl compounds with fewer than four carbons are soluble in water. Esters, N,N-disubstituted amides, and nitriles are often used as solvents because they are polar, but do not have reactive hydroxyl or amino groups. We have seen that dimethylfo ...
... and aromatic hydrocarbons. Like alcohols and ethers, carbonyl compounds with fewer than four carbons are soluble in water. Esters, N,N-disubstituted amides, and nitriles are often used as solvents because they are polar, but do not have reactive hydroxyl or amino groups. We have seen that dimethylfo ...
OChem1 Course Pack
... The secondary carbocation that is formed undergoes rearrangement to produce a better tertiary cation that no longer has the strained cyclobutyl ring involved. The tertiary carbocation is then deprotonated to give the most highly substituted alkene which is stabilized by four electron-donating alkyl ...
... The secondary carbocation that is formed undergoes rearrangement to produce a better tertiary cation that no longer has the strained cyclobutyl ring involved. The tertiary carbocation is then deprotonated to give the most highly substituted alkene which is stabilized by four electron-donating alkyl ...
10 Haloalkanes and Haloarenes
... Order of reactivity of hydrogen halides is: HI > HBr > HCl > HF In case of symmetrical alkenes, alkyl group or number of hydrogen atoms present on either side of the doubly bonded carbon atoms is same, therefore during addition of HX, only one type of product is ...
... Order of reactivity of hydrogen halides is: HI > HBr > HCl > HF In case of symmetrical alkenes, alkyl group or number of hydrogen atoms present on either side of the doubly bonded carbon atoms is same, therefore during addition of HX, only one type of product is ...
Alkenes
... reaction yields the more highly substituted alkene as the major product. The more stable alkene product predominates. ...
... reaction yields the more highly substituted alkene as the major product. The more stable alkene product predominates. ...
06_10_13.html
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
SYNTHESIS OF NEW DICLOFENAC DERIVATIVES BY COUPLING WITH CHALCONE
... are based in general on the formation of carbon-carbon bond and here it is the Enone moiety (i.e., the α,β-unsaturated ketone). Among other strategies, the Claisen-Schmidt condensation appeared to be the most appealing one, where it involves the condensation of an aromatic ketone with an aromatic al ...
... are based in general on the formation of carbon-carbon bond and here it is the Enone moiety (i.e., the α,β-unsaturated ketone). Among other strategies, the Claisen-Schmidt condensation appeared to be the most appealing one, where it involves the condensation of an aromatic ketone with an aromatic al ...
Wolff rearrangement

The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo [2+2] cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical retention of the migrating group. However, the Wolff rearrangement has limitations due to the highly reactive nature of α-diazocarbonyl compounds, which can undergo a variety of competing reactions.The Wolff rearrangement can be induced via thermolysis, photolysis, or transition metal catalysis. In this last case, the reaction is sensitive to the transition metal; silver (I) oxide or other Ag(I) catalysts work well and are generally used. The Wolff rearrangement has been used in many total syntheses; the most common use is trapping the ketene intermediate with nucleophiles to form carboxylic acid derivatives. The Arndt-Eistert homologation is a specific example of this use, wherein a carboxylic acid may be elongated by a methylene unit. Another common use is in ring-contraction methods; if the α-diazo ketone is cyclic, the Wolff rearrangement results in a ring-contracted product. The Wolff rearrangement works well in generating ring-strained systems, where other reactions may fail.