Another look at chemical reactions HYDROGEN PEROXIDE WATER
... - slightly more massive than the proton ELECTRON - a small particle that carries an overall unit NEGATIVE CHARGE - about 2000 times LESS massive than either protons or neutrons ...
... - slightly more massive than the proton ELECTRON - a small particle that carries an overall unit NEGATIVE CHARGE - about 2000 times LESS massive than either protons or neutrons ...
Practice exam Part 3 Name 1) A Ca 2+ ion differs from a Ca0 atom in
... 2) Which particles are referred to as nucleons (subatomic particles located in the nucleus)? a) protons and neutrons c) neutrons, only b) protons and electrons d) neutrons and electrons 3) What is the mass number of an atom that contains 19 protons, 19 electrons, and 20 neutrons? a) 39 b) 19 c) ...
... 2) Which particles are referred to as nucleons (subatomic particles located in the nucleus)? a) protons and neutrons c) neutrons, only b) protons and electrons d) neutrons and electrons 3) What is the mass number of an atom that contains 19 protons, 19 electrons, and 20 neutrons? a) 39 b) 19 c) ...
Document
... • The energies of K" x-rays can be calculated by replacing Z by Z–1 ! identification of some chemical elements for the first time. • Stimulated emission, the laser. Monday, April 5, 2010 ...
... • The energies of K" x-rays can be calculated by replacing Z by Z–1 ! identification of some chemical elements for the first time. • Stimulated emission, the laser. Monday, April 5, 2010 ...
I. Atoms II. Chemical Symbols III. Structure
... Atoms are the basic building blocks that make up you and I as well as everything around us, both living and non-living. A substance made up of one type of atom only is called an element. There are currently 118 different elements of which 98 occur naturally, the others have been created by humans. T ...
... Atoms are the basic building blocks that make up you and I as well as everything around us, both living and non-living. A substance made up of one type of atom only is called an element. There are currently 118 different elements of which 98 occur naturally, the others have been created by humans. T ...
Structure of the atom
... Calcium-40: mass number = 40; atomic number = 20; number of protons = 20; number of neutrons = 20; number of electrons = 20 (Inquiry point 1) Carbon-14: mass number = 14; atomic number = 6; number of protons = 6; number of neutrons = 8; number of electrons = 6 (Inquiry point 2) Nitrogen-14: mass num ...
... Calcium-40: mass number = 40; atomic number = 20; number of protons = 20; number of neutrons = 20; number of electrons = 20 (Inquiry point 1) Carbon-14: mass number = 14; atomic number = 6; number of protons = 6; number of neutrons = 8; number of electrons = 6 (Inquiry point 2) Nitrogen-14: mass num ...
Parts of the Atom - Dalton Local Schools
... 14. What is true about the number of electrons and protons in an element? a. There is always twice the number of electrons than protons in the nucleus. b. The numbers of protons and electrons are always changing. c. The number of electrons in an atom always equals the number of protons in the nucle ...
... 14. What is true about the number of electrons and protons in an element? a. There is always twice the number of electrons than protons in the nucleus. b. The numbers of protons and electrons are always changing. c. The number of electrons in an atom always equals the number of protons in the nucle ...
5.7 Nuclear Radiation
... – The process continues until unstable isotopes of one element are changed, or transformed, into stable isotopes of a different element. – These stable isotopes are not radioactive. – Nuclear radiation is emitted during radioactive decay. – There are three main types of nuclear radiation: alpha rad ...
... – The process continues until unstable isotopes of one element are changed, or transformed, into stable isotopes of a different element. – These stable isotopes are not radioactive. – Nuclear radiation is emitted during radioactive decay. – There are three main types of nuclear radiation: alpha rad ...
Chapter 18 - An Introduction to Chemistry: Nuclear
... the same atomic number, but they have a different mass number, which is the sum of the numbers of protons and neutrons in the nucleus. In the context of nuclear science, protons and neutrons are called nucleons, because they reside in the nucleus. The atom’s mass number is often called the nucleon n ...
... the same atomic number, but they have a different mass number, which is the sum of the numbers of protons and neutrons in the nucleus. In the context of nuclear science, protons and neutrons are called nucleons, because they reside in the nucleus. The atom’s mass number is often called the nucleon n ...
atomic number = ZE = Element symbol
... Atoms are composed of three main subatomic particles: protons, neutrons and electrons. Protons and neutrons are found in the nucleus of an atom. The total number of protons and neutrons determines an atom’s mass. The number of protons defines the element. Some nuclei are unstable, so they decompose ...
... Atoms are composed of three main subatomic particles: protons, neutrons and electrons. Protons and neutrons are found in the nucleus of an atom. The total number of protons and neutrons determines an atom’s mass. The number of protons defines the element. Some nuclei are unstable, so they decompose ...
Chapter 3
... Isotopes: atoms of the same element (X) with different numbers of neutrons in their nuclei ...
... Isotopes: atoms of the same element (X) with different numbers of neutrons in their nuclei ...
Atomic Terms/People
... Dalton later published his atomic theory. This was used to determine chemical formulas. Dalton made the first table of atomic masses o Later proven inaccurate. Dalton changed his theory based on the Atomic Number: It now states that all atoms of an element contain the same # of protons but they ca ...
... Dalton later published his atomic theory. This was used to determine chemical formulas. Dalton made the first table of atomic masses o Later proven inaccurate. Dalton changed his theory based on the Atomic Number: It now states that all atoms of an element contain the same # of protons but they ca ...
A = Atomic Number
... To find the number of neutrons in an atom, subtract the atomic number from the mass number. # NEUTRONS = MASS NUMBER - ATOMIC NUMBER ...
... To find the number of neutrons in an atom, subtract the atomic number from the mass number. # NEUTRONS = MASS NUMBER - ATOMIC NUMBER ...
Chapter 4 Notes - DunlapChemistry
... In different energy levels; each row on the periodic table is a new energy level Do not contribute to atomic mass A proton’s mass is 1800 times greater than that of an electron Gained, lost or shared in bonding Neutrons Neutral charge (no charge) In the nucleus Contribute to atomic mass Never gained ...
... In different energy levels; each row on the periodic table is a new energy level Do not contribute to atomic mass A proton’s mass is 1800 times greater than that of an electron Gained, lost or shared in bonding Neutrons Neutral charge (no charge) In the nucleus Contribute to atomic mass Never gained ...
Unit 1 – Atomic Structure
... The Quantum Mechanical Model A. Probability and the Electron 1. The position and direction of motion of the electron cannot be simultaneously determined Translated: “The more certain I am about where it is, the less certain I can be about where it is going. The more certain I am about where it is go ...
... The Quantum Mechanical Model A. Probability and the Electron 1. The position and direction of motion of the electron cannot be simultaneously determined Translated: “The more certain I am about where it is, the less certain I can be about where it is going. The more certain I am about where it is go ...
Atomic Structure/Atomic Theory
... Has a mass of 1 atomic mass unit (u) The number of protons determines the type of element o Neutron Neutral (no charge) particle in the nucleus Has a mass of 1 atomic mass unit (u) o Electron Negative charged particle found in the electron cloud Electrons circle the nucleus Electrons c ...
... Has a mass of 1 atomic mass unit (u) The number of protons determines the type of element o Neutron Neutral (no charge) particle in the nucleus Has a mass of 1 atomic mass unit (u) o Electron Negative charged particle found in the electron cloud Electrons circle the nucleus Electrons c ...
Worksheet - Rudds Classroom
... 18. A uranium atom can have an atomic mass of 235 or 238. Each atom is, therefore, a. a different isotope c. a different element b. negatively charged d. stable 19. Atoms that emit particles and energy from their nuclei are called a. contaminated b. stable c. heavy d. radioactive. 20. The electromag ...
... 18. A uranium atom can have an atomic mass of 235 or 238. Each atom is, therefore, a. a different isotope c. a different element b. negatively charged d. stable 19. Atoms that emit particles and energy from their nuclei are called a. contaminated b. stable c. heavy d. radioactive. 20. The electromag ...
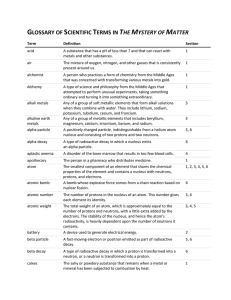
GLOSSARY OF SCIENTIFIC TERMS IN THE MYSTERY OF MATTER
... charge. Electromagnetism is the force that causes the interaction between electrically charged particles; the areas in which this ...
... charge. Electromagnetism is the force that causes the interaction between electrically charged particles; the areas in which this ...
Topic_7_atomic_and_nuclear_energy_IB_problems
... Explain why the temperature and pressure of the gases in the Sun’s core must both be very high for it to produce its radiant energy. High temperature: ............................................................................................ ...
... Explain why the temperature and pressure of the gases in the Sun’s core must both be very high for it to produce its radiant energy. High temperature: ............................................................................................ ...
Atomic Structure
... •Even though isotopes have different amounts of neutrons they are still chemically alike since they have the same number of protons and electrons. •To find the most common isotope round the atomic mass to nearest whole number. -Ex: Carbon-12 is the most common isotope of carbon Which isotope is the ...
... •Even though isotopes have different amounts of neutrons they are still chemically alike since they have the same number of protons and electrons. •To find the most common isotope round the atomic mass to nearest whole number. -Ex: Carbon-12 is the most common isotope of carbon Which isotope is the ...
The parts of Dalton`s theory Matter is composed of small, chemically
... ELEMENTS are kinds of matter that contain only a single kind of atom. All the atoms of an element have identical chemical properties. COMPOUNDS are kinds of matter that are composed of atoms of two or more ELEMENTS which are combined in simple, whole number ratios. Most importantly, CHEMICAL REACTIO ...
... ELEMENTS are kinds of matter that contain only a single kind of atom. All the atoms of an element have identical chemical properties. COMPOUNDS are kinds of matter that are composed of atoms of two or more ELEMENTS which are combined in simple, whole number ratios. Most importantly, CHEMICAL REACTIO ...
Atomic Structure Atoms. Summary Atomic Number.
... The mass of a neutron and a proton are the same. An electron is very much smaller, about 1/2000th the size of a proton, although it has an equal and opposite electrical charge. The electrons, although tiny, take up most of the space of an atom. This means that most of the space of an atom contains h ...
... The mass of a neutron and a proton are the same. An electron is very much smaller, about 1/2000th the size of a proton, although it has an equal and opposite electrical charge. The electrons, although tiny, take up most of the space of an atom. This means that most of the space of an atom contains h ...
Cornell Notes 6
... •Protons give atoms their IDENTITY. In other words, if you have an atom with one proton, it HAS to be a Hydrogen atom. Two protons means it HAS to be a Helium atom and so on. •This means all atoms of the same element have the same number of protons •Electrons give atoms their PROPERTIES (except for ...
... •Protons give atoms their IDENTITY. In other words, if you have an atom with one proton, it HAS to be a Hydrogen atom. Two protons means it HAS to be a Helium atom and so on. •This means all atoms of the same element have the same number of protons •Electrons give atoms their PROPERTIES (except for ...
Canyon High School Chemistry
... Chemistry content standards addressed in this unit: 1a. Know how the position of an element in the periodic table relates to its atomic number and atomic mass; 1e. Know the nucleus of the atom is smaller than the atom yet contains most of its mass; 1f. Know transuranium elements are all synthesized ...
... Chemistry content standards addressed in this unit: 1a. Know how the position of an element in the periodic table relates to its atomic number and atomic mass; 1e. Know the nucleus of the atom is smaller than the atom yet contains most of its mass; 1f. Know transuranium elements are all synthesized ...