Unit 7: Atomic Theory
... charge is concentrated. This core he called the nucleus. He believed that the rest of the atom was mainly empty space with negative electrons spread throughout. He called this part of the atom the electron cloud. •Rutherford explains his results using the above model for his atom: ...
... charge is concentrated. This core he called the nucleus. He believed that the rest of the atom was mainly empty space with negative electrons spread throughout. He called this part of the atom the electron cloud. •Rutherford explains his results using the above model for his atom: ...
Exemplar exam question – Chapter 2
... The first answer is probably worthy of only 1 mark as it does not make clear that isotopes are different atoms of the same element. The second answer would probably score 0. Although the idea of the same element and different number of neutrons is mentioned, the student has not mentioned different a ...
... The first answer is probably worthy of only 1 mark as it does not make clear that isotopes are different atoms of the same element. The second answer would probably score 0. Although the idea of the same element and different number of neutrons is mentioned, the student has not mentioned different a ...
(or radioactive isotopes).
... • Gamma rays are used to kill bacteria, mould and insects in food. They are also used to kill bacteria on hospital equipment, dressings and bandages. • This is useful particularly on packaged food or on plastic items which would be damaged by heat sterilisation. • There are arguments for using cobal ...
... • Gamma rays are used to kill bacteria, mould and insects in food. They are also used to kill bacteria on hospital equipment, dressings and bandages. • This is useful particularly on packaged food or on plastic items which would be damaged by heat sterilisation. • There are arguments for using cobal ...
Santee Education Complex Chemistry Mini Assessment 11
... b. 0n1 + 13Al27 → 11Na24 + 2He4 c. 13Al27 + 2He4 → 15P30 +0n1 d. 7N14 + 2He4 →1H1 + 8O17 14) A process in which a very heavy nucleus splits into more stable nuclei of intermediate mass is called: a. nuclear fission. b. a chain reaction. c. nuclear fusion. d. radiocarbon dating. 15) An electron emitt ...
... b. 0n1 + 13Al27 → 11Na24 + 2He4 c. 13Al27 + 2He4 → 15P30 +0n1 d. 7N14 + 2He4 →1H1 + 8O17 14) A process in which a very heavy nucleus splits into more stable nuclei of intermediate mass is called: a. nuclear fission. b. a chain reaction. c. nuclear fusion. d. radiocarbon dating. 15) An electron emitt ...
Isotopes Models
... sometimes called Protium. A hydrogen atom that has one proton and one neutron in its nucleus is called Deuterium. Deuterium is not radioactive. Water made from deuterium is called heavy water because the extra neutron makes it heavier. It is used in nuclear reactors. The third isotope of hydrogen is ...
... sometimes called Protium. A hydrogen atom that has one proton and one neutron in its nucleus is called Deuterium. Deuterium is not radioactive. Water made from deuterium is called heavy water because the extra neutron makes it heavier. It is used in nuclear reactors. The third isotope of hydrogen is ...
Study Guide - Chapter 11
... 1. Atomic mass unit – the SI unit of mass that describes the mass of an atom A. Proton has a mass of 1 amu B. Neutron has a mass of 1 amu C. It takes more than 1800 electrons to equal the mass of 1 proton B. Neutrons – particles of the nucleus that have no charge Electrons – negatively charge partic ...
... 1. Atomic mass unit – the SI unit of mass that describes the mass of an atom A. Proton has a mass of 1 amu B. Neutron has a mass of 1 amu C. It takes more than 1800 electrons to equal the mass of 1 proton B. Neutrons – particles of the nucleus that have no charge Electrons – negatively charge partic ...
Script of atoms video
... # of protons gives identity of the element (atomic number) So what is helium’s atomic number? (fig. 2.3) Protons + Neutrons gives atomic mass So what is the atomic mass of helium? (fig. 2.3) For uncharged (electrically neutral) atoms: # of protons = # of electrons Re-cap Protons and neutrons are in ...
... # of protons gives identity of the element (atomic number) So what is helium’s atomic number? (fig. 2.3) Protons + Neutrons gives atomic mass So what is the atomic mass of helium? (fig. 2.3) For uncharged (electrically neutral) atoms: # of protons = # of electrons Re-cap Protons and neutrons are in ...
Basics of Nuclear Physics and Fission
... elements in the middle of the periodic table of elements, with intermediate mass numbers have a higher binding energy per nucleon (that is a lower atomic weight per nucleon). Therefore when a heavy nucleus is fissioned, the resultant products of the nuclear reaction have a slightly smaller combined ...
... elements in the middle of the periodic table of elements, with intermediate mass numbers have a higher binding energy per nucleon (that is a lower atomic weight per nucleon). Therefore when a heavy nucleus is fissioned, the resultant products of the nuclear reaction have a slightly smaller combined ...
Nuclear Chemistry
... "radioactivity" to describe the spontaneous emissions that they studied. This word is still used today to describe this special characteristic of some elements.(radioisotopes). • While comparing the activity of pure uranium to a uranium ore sample, they found that the ore was significantly more radi ...
... "radioactivity" to describe the spontaneous emissions that they studied. This word is still used today to describe this special characteristic of some elements.(radioisotopes). • While comparing the activity of pure uranium to a uranium ore sample, they found that the ore was significantly more radi ...
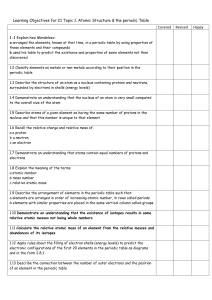
C2- Topic 1: Atomic structure and the periodic table. Assessable
... properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
... properties of these elements and their compounds - used his table to predict the existence and properties of some elements not then discovered ...
Electron Arrangement
... Atomic Number • Moseley, in an experiment called the X-ray tube experiment, determined that each element has a different and unique number of protons. • The number of protons is referred to as the element’s atomic number. • The atomic number can be used to identify the element. • Atomic Number = # ...
... Atomic Number • Moseley, in an experiment called the X-ray tube experiment, determined that each element has a different and unique number of protons. • The number of protons is referred to as the element’s atomic number. • The atomic number can be used to identify the element. • Atomic Number = # ...
C2 Topic 1 Can Do Sheet
... b used his table to predict the existence and properties of some elements not then discovered 1.2 Classify elements as metals or non-metals according to their position in the periodic table 1.3 Describe the structure of an atom as a nucleus containing protons and neutrons, surrounded by electrons in ...
... b used his table to predict the existence and properties of some elements not then discovered 1.2 Classify elements as metals or non-metals according to their position in the periodic table 1.3 Describe the structure of an atom as a nucleus containing protons and neutrons, surrounded by electrons in ...
Chapter 18 Notes
... *so the Atomic Number is 6 and therefore it has 6 protons, & 6 neutrons. -Isotope of carbon- Carbon-14 *Atomic # is still 6, so you have 6 protons & 8 Neutrons. *They use how much each isotope occurs and it’s mass to determine the Average atomic mass. Ex- IF you have an average atomic mass of 35.95 ...
... *so the Atomic Number is 6 and therefore it has 6 protons, & 6 neutrons. -Isotope of carbon- Carbon-14 *Atomic # is still 6, so you have 6 protons & 8 Neutrons. *They use how much each isotope occurs and it’s mass to determine the Average atomic mass. Ex- IF you have an average atomic mass of 35.95 ...
Alpha Beta Fission Fusion
... chemistry to include nuclear changes when he discovered that uranium emitted radiation. Soon after Becquerel's discovery, Marie Sklodowska Curie began studying radioactivity and completed much of the pioneering work on nuclear changes. Curie found that radiation was proportional to the amount of rad ...
... chemistry to include nuclear changes when he discovered that uranium emitted radiation. Soon after Becquerel's discovery, Marie Sklodowska Curie began studying radioactivity and completed much of the pioneering work on nuclear changes. Curie found that radiation was proportional to the amount of rad ...
CH 4 - USD 395
... *key to the power of the sun ~responsible for radioactive decay ~ +/- ---> + and 3. ...
... *key to the power of the sun ~responsible for radioactive decay ~ +/- ---> + and 3. ...
nuclear test 2006
... 90 Th nucleus undergoes radioactive decay by the emission of an alpha particle the new nucleus formed is A ...
... 90 Th nucleus undergoes radioactive decay by the emission of an alpha particle the new nucleus formed is A ...
Parts of an Atom
... An atom consists of a nucleus surrounded by one or more electrons The nucleus is the center region of the atom where the protons and neutrons are located ...
... An atom consists of a nucleus surrounded by one or more electrons The nucleus is the center region of the atom where the protons and neutrons are located ...
subatomic particles
... - Atomic mass unit (amu): 1 proton has a mass of 1 amu; determined by taking the mass of a Carbon-12 atom and dividing by the total number of particles. - Atomic number: the number of protons in an atom. - Mass number: the sum of the protons and neutrons in an atom. - Isotopes: atoms of the same ele ...
... - Atomic mass unit (amu): 1 proton has a mass of 1 amu; determined by taking the mass of a Carbon-12 atom and dividing by the total number of particles. - Atomic number: the number of protons in an atom. - Mass number: the sum of the protons and neutrons in an atom. - Isotopes: atoms of the same ele ...
Drawing Atomic Structure
... The average atomic mass is the ______________________ mass of an element’s various isotopes (takes into consideration which isotopes are more ______________ than others). The average atomic mass can be found on the Periodic Table. Label this on the diagram on the first page. ...
... The average atomic mass is the ______________________ mass of an element’s various isotopes (takes into consideration which isotopes are more ______________ than others). The average atomic mass can be found on the Periodic Table. Label this on the diagram on the first page. ...
Nuclear physics α −
... number of neutrons. In several cases the nucleus is sable structure. What kind of interaction exists inside the nucleus? This interaction is not gravitational (the gravitational interaction is very week), not electrical interaction (the neutrons are neutral, and what is more the protons repulse each ...
... number of neutrons. In several cases the nucleus is sable structure. What kind of interaction exists inside the nucleus? This interaction is not gravitational (the gravitational interaction is very week), not electrical interaction (the neutrons are neutral, and what is more the protons repulse each ...
Article 2: Key Concepts and Vocabulary
... in many different forms, and the quantity of energy does not change as it is transformed from one form to another. In a magnetic fusion reactor, there are many forms of energy. Electric currents flowing through coils of conducting wires create magnetic energy in the form of magnetic fields that conf ...
... in many different forms, and the quantity of energy does not change as it is transformed from one form to another. In a magnetic fusion reactor, there are many forms of energy. Electric currents flowing through coils of conducting wires create magnetic energy in the form of magnetic fields that conf ...
Unit 2, Day 25
... is used because the mass of each subatomic particle is too small to measure in grams The mass of each atom is equal to the number of protons plus the number of neutrons. (Electrons are ignored, because their mass is so small that they don’t affect the mass enough) ...
... is used because the mass of each subatomic particle is too small to measure in grams The mass of each atom is equal to the number of protons plus the number of neutrons. (Electrons are ignored, because their mass is so small that they don’t affect the mass enough) ...
Periodic Table Jeopardy
... A substance that cannot be separated or broken down into simpler substances by chemical means. All atoms in this substance have the same atomic #. ...
... A substance that cannot be separated or broken down into simpler substances by chemical means. All atoms in this substance have the same atomic #. ...