NAME…………… - Kcse Online
... (i) Write your name and index number in the spaces provided above. (ii) Sign and write the date of examination in the spaces provided above. (iii) Answer ALL the questions in the spaces provided. (iv) Mathematical tables and silent electronic calculators may be used. (v) All working must be clearly ...
... (i) Write your name and index number in the spaces provided above. (ii) Sign and write the date of examination in the spaces provided above. (iii) Answer ALL the questions in the spaces provided. (iv) Mathematical tables and silent electronic calculators may be used. (v) All working must be clearly ...
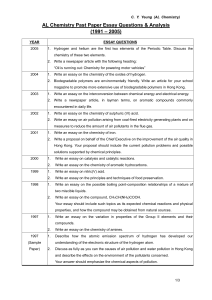
Chemistry
... a) Oxidation and reduction (16 L) General concepts, Electrochemical series and its applications, redox stability in water – Frost( nitrogen) , Latimer ( chlorine in acidic and basic medium, disproportionation of H2O2 into O2 and H2O under acid conditions)) and Pourbaix ( iron species in natural wate ...
... a) Oxidation and reduction (16 L) General concepts, Electrochemical series and its applications, redox stability in water – Frost( nitrogen) , Latimer ( chlorine in acidic and basic medium, disproportionation of H2O2 into O2 and H2O under acid conditions)) and Pourbaix ( iron species in natural wate ...
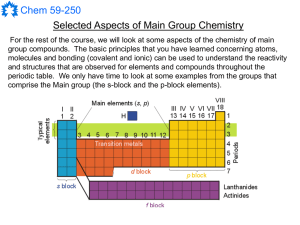
Main Group Notes 1
... E.g.: Na(s) (dissolved in NH3 (l)) Na+(am) + e-(am) At low concentration this is a blue solution that contains solvated electrons! If the reaction warms up or is catalyzed, the free electron reacts with the solvent to reduce some of the protons in the solvent to produce hydrogen gas: ...
... E.g.: Na(s) (dissolved in NH3 (l)) Na+(am) + e-(am) At low concentration this is a blue solution that contains solvated electrons! If the reaction warms up or is catalyzed, the free electron reacts with the solvent to reduce some of the protons in the solvent to produce hydrogen gas: ...
Chemistry A - Montgomery County Public Schools
... Atomic Structure describe the characteristics of protons, neutrons and electrons in terms of location, charge and mass. illustrate the structure of the atom by using the Bohr model, including the charge, relative mass and location of the sub-atomic particles. use atomic mass, atomic number, an ...
... Atomic Structure describe the characteristics of protons, neutrons and electrons in terms of location, charge and mass. illustrate the structure of the atom by using the Bohr model, including the charge, relative mass and location of the sub-atomic particles. use atomic mass, atomic number, an ...
Summer Assignment for AP Chemistry: I hope you are all ready for a
... Writing chemical equations is also an essential skill. In Chem I, you used the rxns packet to help you predict products of reactions. In AP Chem, you have to memorize all of that information and more! Zoikes! Don’t worry; you’ll learn it a little at a time. But, you should at least be able to do it ...
... Writing chemical equations is also an essential skill. In Chem I, you used the rxns packet to help you predict products of reactions. In AP Chem, you have to memorize all of that information and more! Zoikes! Don’t worry; you’ll learn it a little at a time. But, you should at least be able to do it ...
Chemical reactions
... Reaction products = substances formed in a chemical reaction. They are written in the right term of the equation Because in a chemical reaction, the nature of atoms of the substances is not changed, the chemical equations are equalized so that the number of atoms of a certain element from the left t ...
... Reaction products = substances formed in a chemical reaction. They are written in the right term of the equation Because in a chemical reaction, the nature of atoms of the substances is not changed, the chemical equations are equalized so that the number of atoms of a certain element from the left t ...
Chemistry- CST Review
... nitrogen, oxygen, and another carbon commonly form bonds with carbon. Standard 11- Nuclear Processes 1. What elements have radioactive isotopes? Elements with atomic number 84 and above are radioisotopes. There are more like carbon which has a radioisotope of carbon-14. 2. What is the difference bet ...
... nitrogen, oxygen, and another carbon commonly form bonds with carbon. Standard 11- Nuclear Processes 1. What elements have radioactive isotopes? Elements with atomic number 84 and above are radioisotopes. There are more like carbon which has a radioisotope of carbon-14. 2. What is the difference bet ...
Chemistry - School District of Springfield Township
... o Experiments have revealed that the mass of an atom is concentrated in a tiny positively charged nucleus (consisting of protons and neutrons) with a negative cloud of electrons moving around the nucleus. o The number of protons in the nucleus (and electrons in the cloud) is called the atomic number ...
... o Experiments have revealed that the mass of an atom is concentrated in a tiny positively charged nucleus (consisting of protons and neutrons) with a negative cloud of electrons moving around the nucleus. o The number of protons in the nucleus (and electrons in the cloud) is called the atomic number ...
St. Xavier`s College – Autonomous Mumbai Syllabus for 3 Semester
... LEARNING OBJECTIVES 1. To introduce students to the basic concepts involved in chemical bonding. 2. To help students to determine the shape of a molecule by applying VSEPR theory. 3. To encourage students to analyze and integrate concepts relevant to inorganic chemistry required to understand compou ...
... LEARNING OBJECTIVES 1. To introduce students to the basic concepts involved in chemical bonding. 2. To help students to determine the shape of a molecule by applying VSEPR theory. 3. To encourage students to analyze and integrate concepts relevant to inorganic chemistry required to understand compou ...
semester i - Pt. Ravishankar Shukla University
... Determination of the rate constant for the decomposition of hydrogen peroxide by Fe and Cu ions. Determination of the primary salt effect on the kinetics of ionic reactions and testing of the Bronsted relationship (iodide ion is oxidized by persulphate ion). ...
... Determination of the rate constant for the decomposition of hydrogen peroxide by Fe and Cu ions. Determination of the primary salt effect on the kinetics of ionic reactions and testing of the Bronsted relationship (iodide ion is oxidized by persulphate ion). ...
Notebook - Science
... atom: basic unit of a chemical element isotope: each of two or more forms of the same element that contain equal numbers of protons but different numbers of neutrons in their nuclei proton: stable subatomic particle occurring in all atomic nuclei, with positive electric charge to that of an electron ...
... atom: basic unit of a chemical element isotope: each of two or more forms of the same element that contain equal numbers of protons but different numbers of neutrons in their nuclei proton: stable subatomic particle occurring in all atomic nuclei, with positive electric charge to that of an electron ...